Lateral Flow Assay - Gabrielle Berns, Alex Debreceni and Bryanne Zonghi
Introduction and Motivation
Lateral flow assays are paper based microfluidic devices used to detect the presence of a target analyte in a complex mixture. Detecting specific chemical components in liquid mixtures can be done simply by using these cheap paper devices, without needing any costly equipment. An immunoassay is a general procedure for detecting proteins or other substances through their specific antibody properties. The general idea behind this test is that the antibody or some other molecule can interact with a target molecule and usually it binds to the surface, allowing for a readout. This readout typically scales with the number of molecules bound to the surface. Lateral flow immunoassays are essentially a simple diagnostic device to test the presence or absence of a target analyte, such as a pathogen for example. These paper devices that carry out these tests have dominated rapid diagnostics in the past thirty years due them being compact, light-weight, easy to interpret, and disposable [1]. Lateral flow assay tests are a subtype of these devices used for testing that do not require any training or previous experience and are designed for use in non-laboratory settings. These devices are widely used for a variety of different applications. This technology is commonly used in medical fields and law enforcement to test for bodily fluids such as urine, blood, sweat, and saliva [2]. Biomarkers for organ failure, infection, toxic pathogens, or illicit drugs can be detected with high sensitivity and selectivity due to the fact that each device is specifically designed to interact with the molecule of question [3]. Essentially these devices operate through the flow process called capillary action, which pushes the fluid containing the analyte through various zones, where molecules are bound to interact with and detect only that specific analyte [3]. Lateral flow assay tests are lucrative because there is a large demand for decentralized diagnostic tests that are easy to use, accurate, and provide rapid results [4]. These lateral flow assays also include a control line and a target test line that is used to confirm that the test is actually working properly. This is included in the design of the device to incorporate intuitive user protocol and it does not require any training to operate or understand the device. These control line tests usually can be visually determined and are very simple to understand.
History
The technical ideas of these microfluidic paper devices originated in 1956. The ideas for lateral flow assays derived from Charles M. Plotz and Jaques M. Singer when they first discovered Rheumatoid Factor Test based on latex agglutination assays. During this time, plate-based immunoassays and the first radio-immunoassay (RIA) were also newly developed. The enzyme immunoassay was first developed in the 1960s and replaced radioisotopes due to its faster reaction times and longer shelf-life. During the years following this, the fundamental principles of lateral flow technology continued to develop until the 1980s [4] . During the late 1980s, this technology finally became firmly established within the field. Companies finally began to patent this paper microfluidic technology, specifically in the 1970s with great strides in urine testing for medical diagnosis. Finally, the first lateral flow assay device was put on the market during the 1980s. Since the initial introduction to the market, lateral flow assay products have continued to evolve and become more reliable. As of the mid 2000s, hundreds of companies internationally started creating their own testing formats relating to lateral flow assay technology. This technology has already expanded to areas of agriculture, food, environmental health and safety, biowarfare, and most commonly for pregnancy tests [4] .
Components of the Assay
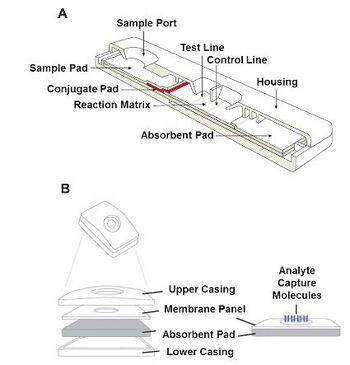
Lateral flow assays are generally carried out on strips, shown to the left in Figure 1, which contain 4 major parts: a sample application pad, conjugate pad, nitrocellulose membrane (labeled as the reaction matrix in Figure 1) and adsorption pad [5]. The application pad, typically made of cellulose or glass fiber, is where the sample is first applied at the start of the test. The application pad treats the sample and modulates any chemical variability. The pad can also be made of cotton linter, glass fibre, rayon, and filtration materials. These pads are pretreated with a pH buffer, surfactants, and blocking reagents to influence the flow rate and viscosity of the sample [3] This pad must have properties that allow for a continuous transport of the sample to the next part of the device, the conjugate pad. The conjugate pad couples the analyte in the sample with the conjugate and releases the pairing. These conjugates are generally antibodies that specifically bind to the target molecule. These antibodies are bound to the pad using colloidal nanoparticles, such as gold [3][6]. This pad is where the labeled molecules for biorecognition are supplied before running the assay [5]. It is vital to have a conjugate that can remain stable throughout the entire assay or the results could be inaccurate or unreadable. Depending on the desired level of sensitivity of the assay and the release of the conjugate, the material of this pad is selected; cellulose, polyesters and glass fiber are most commonly used. In addition to the conjugate pad material, the nitrocellulose membrane is a large contributor to the sensitivity of the lateral flow assay. These membranes contain various evenly distributed pore sizes and work as a capturing mechanism for the conjugate-analyte pair. This part also forms visible bands when the analyte is present. These bands are placed further down the device to improve interaction time. Changing the material and pore size influences the capillary flow time, which influences the reaction time of the analyte in binding area [3]. They are used for test and control lines, and they provide effective binding areas and support that are inexpensive and contain high affinity for many biomolecules and proteins [5]. The last component of the device is the adsorbent, also known as a waste reservoir, which acts as a sink at the end of the assay as shown in figure 1. This component captures the excess sample and prevents wicking towards the reaction membrane [3]. It also helps to maintain a continuous flow rate while preventing any backflow within the system. The five main steps to create a lateral flow consist of (i) developing the antibody used to target the analyte (ii) making the label (iii) choosing and labeling the molecules for recognition (iv) preparing all reagents at their appropriate pad and arranging all aspects of the assay (v) applying the sample onto the device. [5]
Mechanism
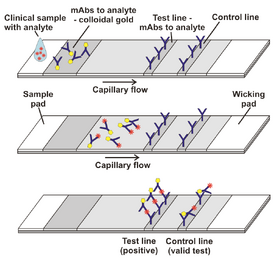
A liquid sample, which contains the analyte of interest is placed on the sample pad. Through capillary action, the fluid travels through the various zones of the device; this is shown in detail in figure 2. When the fluid reaches the conjugate pad, the analyte will bind to the antibodies specific to the molecule. these antibodies are conjugated to colored or fluorescent particles. The most common conjugate particles are colloidal gold and latex microspheres. Figure 2 shows the conjugate-analyte pairing traveling with the fluid to the reaction membrane. This membrane contains antibodies or antigens, immobilized in a nitrocellulose These particles react with the analyte and activates the test line. The control line in the system activates when there is flow through the system. When both lines are present, the analyte is present in the sample. If only the control line is present, there is no analyte in the sample. Once the fluid passes these sections, and absorbing pad at the end of the system collects the excess. This pad also maintains the capillary action throughout the full device [2].
Competitive Assays vs. Sandwich/Direct Assays
Lateral flow assays essentially use capillary action to draw liquid through a paper strip and carry analytes and various reagents to a series of test regions. These lateral flow assays can be divided into two major categories: competitive assays and direct (also known as sandwich) assays. Figure 3 shows the general comparison of the two types of assays. Competitive assays are not suited for high molecular weight compounds. Competitive assays are used for detecting analytes when the antibody pairs are not available, or if the analyte is not large enough for many antibody events. In competitive assays, the test line usually has the analyte molecule and the conjugate pad has the detection antibody and nanoparticle conjugate [7] . The analyte will bind to the conjugate if the target analyte is present. Competitive assays have two different layouts.
The first layout consists of a solution that has the target analyte on the application pad and the conjugate is hydrated and flows with the liquid. The test line has previously immobilized antigens that bind to label conjugates. The control line has previously immobilized secondary antibodies that can bind with labeled antibody conjugates. The previously immobilized antigens bind to the labeled conjugates when the liquid gets to the test line. A competition to bind with the conjugate takes place between the immobilized antigen at the test line strip and the antigen within the sample.
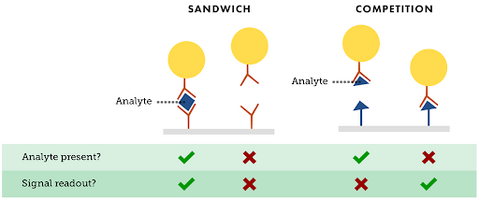
The second layout consists of a labeled analyte conjugate that is dispensed at the conjugate pad and a primary antibody conjugate at the test line. When the analyte solution is applied, there is a competition between the analyte and the labeled analyte to see which can bind with the primary antibody, which occurs at the test line [5]. Competitive assays are specialized for drugs and toxins, and are usually applied to small molecular weight analytes. Figure 3 helps explain that if there is an analyte present for competitive assays, then there is not a signal readout. Similarly, if there is not an analyte present, then there is indeed a signal readout [7]. In competitive assays, the absence of a color at the test line means that the analyte is present. Color at the test line region means the test is negative.
Direct assays, or sandwich assays, are used for larger molecular weight analytes with more than one antigenic sites. This format is normally used to detect larger analytes with at least two binding sites. Typically an antibody to one binding site is conjugated to the nanoparticle and the antibody to the other binding site is used for the assay's test line [7]. These sandwich assays are used to detect larger analytes that have two or more binding sites. An antibody is conjugated to the nanoparticle on one binding site, and another antibody to a different binding site is used for the test line. The analyte will bind to the antibody and the nanoparticle conjugate as well as to the antibody on the test line if the sample contains an analyte. In sandwich assays, the intensity of the signal at the test line is directly proportional to the quantity of analyte present [7]. This kind of test would not work for small molecular weight analytes, only for large molecular weight analytes. Direct assays consist of a label coated antibody, which is usually an enzyme, nanoparticle, or fluorescent dye [5]. The nanoparticles in these assays are a method of detection to simply give the results a visual color. Primary antibodies against target analytes are immobilized over the test line. At the control zone, a secondary antibody is immobilized against the conjugate antibody that is labeled. The sample with the analyte is applied to the application sample and then it migrates to the remaining sections of the strip. The target analyte is then caught by the immobilized antibody and finally the labeled antibody/analyte complex is formed. This labeled antibody/analyte duo then gets to a nitrocellulose membrane and then moves using capillary action [5]. The analyte then migrates in between the labeled antibody and the primary antibody in a sandwich fashion, forming a labeled antibody complex of analyte, conjugate, and the primary antibody. The secondary antibody captures the labeled antibody conjugate in excess at the control zone. The absorption pad captures buffer or any excess solution. The amount of target analyte can be seen through the intensity of the color at the test line. This is measured by using an optical strip reader on the device. Color at the control line is a good indication that the strip is functioning accurately [5]. For sandwich assays, figure 3 helps describe that when there is an analyte present, there is indeed a signal readout [7]. Similarly, when there is not an analyte present, there is not a signal readout [7]. Figure 3 is a helpful visual to summarize the main differences in the two types of lateral flow assays. A colored line means the test is positive, and a negative test comes from the absence of a line there at all. The most common sandwich assay test used commercially is the over the counter pregnancy test.
Advantages as a Paper Microfluidic
Lateral flow assay technology is relatively cheap, simple, and rapid. This specific kind of assay is an attractive technology because it has the potential to give patients an instant diagnosis without any equipment needed. This technology is so useful because it involves only one-step, thus the lateral flow assay technology is a fast process with no previous knowledge or experience needed [2]. The results are qualitative (or semi-qualitative) and entail a very simple test procedure. In these lateral flow assay tests, proteins, haptens, nucleic acids, and amplicons can all be detected. In addition to these tests being rapid and very simple, there is no need to refrigerate them, batches can be prepared ahead of time, and the tests have a long shelf life [2]. Due to long shelf lives and the fact that refrigeration is usually not necessary, lateral flow assay tests can be used in developing countries for a variety of different purposes [2]. Lateral flow assays are also advantageous in resource poor settings because they do not require fancy or expensive equipment to run.
Applications
Pregnancy Tests
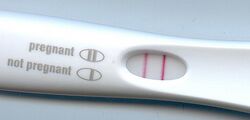
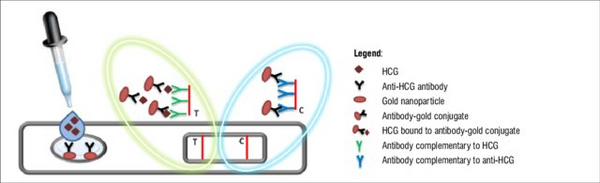
Lateral flow assay devices, specifically sandwich assays, are most commonly used for commercial pregnancy tests. Urine is placed in the sample introduction window and then on the sample pad, as shown in figure 5. When a woman is pregnant, the human chronic gonadotropin (HCG) hormone is present in the urine. Over-the-counter pregnancy tests simply work by detecting HCG, which is made at the beginning of pregnancy. The exposed end of the pregnancy test strip is where the urine is applied. Figure 5 shows that as the fluid travels up the strip, it crosses various separate zones. Antibodies grab onto any HCG as it travels up the strip, with the help of enzymes with the ability to turn on either dye molecules or gold particles. The antibodies used for pregnancy tests are different for each manufacture. Some use antibodies to detect nonnicked hCG and free-beta hCG, while others detect nicked and nonnicked hCG [8]. If HCG is in the urine, then it binds to an antibody attached to a gold particle and results in a positive pregnancy test as shown in figure 4 [3]. Figure 4 shows that when HCG is present in the urine, the result is a positive lateral flow assay pregnancy test. This then flows to meet another antibody that is also complimentary to HCG and results in the test stripe. As the urine continues to flow up the strip, the urine picks up all the AB-1 enzymes and carries them to the test zone of the strip. There are more Y shaped antibodies that will also stick to HCG during this process. The reason this form of lateral assay is called sandwich assay is because if HCG is present, then it gets sandwiched between the two different antibodies. The antibody conjugates that do not bind to HCG flow to a third antibody and results in the control stripe. A negative test happens when the control line appears alone, and a positive test happens when both the test and control line appear [9]. As shown in figure 5, HCG antibodies, gold nanoparticles, antibody gold conjugates, and antibodies complementary to both HCG and anti-HCG are included in these assays. These lateral flow assay devices are commercially available in most drug stores and pharmacies all over the country.
Environmental Testing
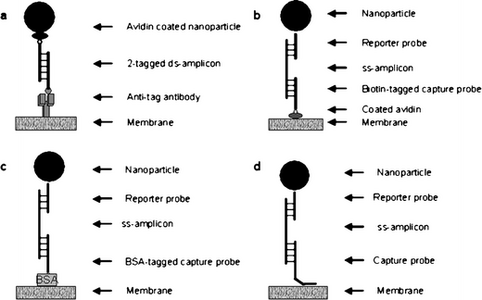
Microfluidic devices such as nucleic acid lateral flow (immuno)assays can test for bacteria or pathogens within the environment. Here, the analyte of interest is a double stranded nucleic acid sequence specific to the organism using primers with two distinct tags. The analyte is recognized because it binds to a tag-specific antibody. When detecting pathogenic bacteria, the nucleic acid was amplified using two tagged primers. At the test line on the device, a solution was sprayed which contained antibodies raised against the tag. One of these tags will bind to specific nanoparticles, and the other tag will bind to the anti-tag antibody, which results in a colored signal. This response is proportional to how much analyte there is in the sample [3]. Figure 6 shows the nucleic acid formats that are possible when using these lateral assay test devices for bacteria and pathogen testing. These nucleic acid formats are available for environmental testing in commercially used kits. As shown in figure 6, there are many possible ways for nucleic acid lateral flow assays to be formatted, with mainly the differences in the detail at the test line. Part a of figure 6 shows the nucleic acid lateral flow assay with nanoparticle-labelled avidin used as reporter with two tagged primers [3]. In this format, biotin binds to avidin and the anti-FITC antibody bind to FITC. Part b of figure 6 is different because of the addition of a single-stranded amplicon (ss-amplicon) with various probes [3]. Part c of figure 6 has the addition of the BSA probe that is immobilized through passive adsorption [3]. Finally, part d of figure 6 shows the capture probe immobilized at the test time by ss-amplicon hybridizing with reporter and capture probes [3]. The importance of figure 6 is that it shows just some of the possible formats for nucleic acid lateral flow assays that test for the presence or absence of pathogens in the environment.
Crop and Animal Health
Pesticides are extensively used in crops in order to keep away insects, diseases and weeds, and they can end up in the bodies of humans and animals which may cause many health issues. There is a lateral flow assay that is useful for simultaneously detecting two common toxins, triazophos and carbofuran, from the pesticides within the water at the crops [5]. This assay uses an immunogold conjugate, and the strip contains the control line and two test lines for each toxin. If these toxins are detected, other studies can be performed to determine the how much is actually present and whether the food could safely be given to humans to consume. There are many lateral flow tests that can detect diseases in various animals. These tests are not only helpful in detecting animal diseases, but they help mitigate human exposure when interacting with these animals. Bisphenol A (BPA) has been found to harm the fertility of fish by reducing their population. A rapid, one step assay was created to detect this contaminant in fish within the water of oceans and rivers where these fish swim. Humans are now able to determine the reproductive status of most dairy animals using a colloidal gold conjugate to detect the analyte, called progesterone, found in their milk. Also, an additive within the food of livestock, ractopamine, is toxic to humans. Ractopamine is now detectable using a lateral flow assay and the urine of pigs [5]. These devices are available for routine crop health management and veterinary diagnostic testing.
Food Testing
Lateral flow assays can also be used for determining and ensuring the safety of some food products. In addition to testing the water on crop land, food products can be tested for pesticides using gold nanoparticles. A recent lateral flow assay was created for detecting Salmonella at lower concentrations using gold nanoparticles and aptamer. A nucleic acid lateral flow assay is able to actually quantify the Salmonella within a food sample [5]. Another novel use for these assays is to prevent the chance of dealing with food poisoning. Staphylococcol exterotoxin B (SEB) is one of the most common toxins affiliated with food poisoning, and lateral flow tests now are capable of detecting SEB in both food and liquids. Food testing using lateral flow assays is an extremely useful, rapid, and easy way to detect a wide range of contaminants that could prevent the body from getting food poisoning or viruses [5]. Food testing lateral flow assay devices are available commercially in test strip forms for foodborne bacterial pathogens and other toxins. The primary application for the microbiology sector of this field is mainly testing in the laboratory, but there are indeed some commercial field test kits for mycotoxins in many samples.
Detection
Lateral flow immunoassay performance can be determined analytically by using nanoparticles. Nanoparticles are used in lateral flow assays to simply give the device a resulting color, so that it is easy to see a positive or negative test visually. Gold nanoparticles specifically are used as a method of detection to improve the analytical sensitivity and detection range of these devices, along with quantifying lateral flow assays. The size of the nanoparticle used is the primary contributor to the lateral flow assay performance. For sandwich lateral flow devices for example, the analyte that flows through the assay is captured by detection antibody gold nanoparticles. These gold nanoparticles are then captured by antibodies on the membrane and eventually the nanoparticles accumulate on the test site. If the test site visually is red for this specific test, then the results are positive and there were enough gold nanoparticles present [10]. Another form of detection, which is slightly less common, is lateral flow assay with a pressure meter readout for disease-associated protein detection. Since lateral flow assays typically require specified instruments to obtain quantitative results, handheld pressure meter readouts can provide rapid detection of disease-related proteins with large sensitivity. There is a noticeable pressure change produced by the catalytic reaction of Pt nanoparticles related to the target concentration. Platinum (Pt) nanoparticles accumulate in the test line and form a band by biomolecular recognition, then eventually convert this into a very sensitive pressure readout to later be analyzed [11] . Finally, the last form of lateral flow assay detection is using enzymes that drive a colorimetric reaction. Some lateral flow assay device have the ability to test for multiple analytes in a single sample by using multi-path lateral flow devices. These multi-flow paths allow for the simultaneous testing of various analytes individually by looking for the appearance of colored test lines in individual channels. The result of a colored test line channel indicates the presence of a certain analyte within that sample. Multiple analyte detection in one lateral flow assay device is thus possible by using colorimetric reactions for each individual channel in the one device. Each channel has a different enzyme that drives a certain colorimetric reaction, resulting in a different color. This makes it possible to carry out these tests for more than one analyte [12]. Colorimetric reaction and nanoparticles are both commonly used for detection methods with lateral flow assays, while a pressure meter readout for disease-associated protein detection is less common. The main reasons for choosing one of these methods over another is for simplicity, and thus the colorimetric reaction detection methods would be recommended. As previously mentioned, this method of detection also easily allows for multi-flow paths for the simultaneous testing of various analytes.
Shortcomings
While these tests are indeed very common and easy to use, lateral flow assay technology is dependent on an accurate sample volume. Small or invalid sample volume results in reduced accuracy. In addition to sample volume, good antibody preparation is required for these tests to confidently work. This technology works very well for fluid samples with no pretreatment needed, but sample pretreatment is indeed a requirement when testing non-fluids, which can be very time consuming [2]. Another limitation to lateral flow assays is that the results obtained are generally qualitative which may prevent specific conclusions to be made, and the reproducibility of each test can vary quite significantly. However, people have indeed tried to make these devices more quantitative by creating multiple test lines, as previously mentioned in the detection section. Multi-flow paths have been implemented in these devices to allow for the simultaneous testing of multiple analytes [12].
Future Work
The main advances in lateral flow assay technology pertains to improving detection sensitivity in order to obtain results for lower concentrations of the analyte that could be found in saliva or sweat. Various novel reagents have been discovered to reduce the detection limits to a minimum of 0.1 ng/ml. To do this, magnetic particles such as nano-gold microspheres can be used to decrease detection limits [2]. Another goal for future work is to find a way to make the assays more reproducible to improve its data processing and quantification capabilities [5].
References
[1] Yetisen, A. K., Akram, M. S., & Lowe, C. R. (2013). Paper-based microfluidic point-of-care diagnostic devices. Lab on a Chip, 13(12), 2210. doi:10.1039/c3lc50169h
[2] Koczula, K. M.; Gallotta, A. Essays In Biochemistry 2016, 60 (1), 111–120. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4986465/
[3] Posthuma-Trumpie, G.A., Korf, J. & van Amerongen, A. Anal Bioanal Chem (2009) 393: 569. https://doi.org/10.1007/s00216-008-2287-2 https://link.springer.com/article/10.1007/s00216-008-2287-2
[4] Wong, R. C.; Tse, H. Y. Lateral Flow Immunoassay 2008, 1–19. http://www.diagnostics1.com/MANUAL/LFIA_Book%5B1%5D.pdf
[5] Sajid, M.; Kawde, A.-N.; Daud, M. Journal of Saudi Chemical Society 2015, 19(6), 689–705. https://www.sciencedirect.com/science/article/pii/S131961031400129X
[6] Martin, J. M., P[Acaron]Ques, M., Tiny A. M. Van Der Velden-De Groot, & Beuvery, E. C. (1990). Characterization of Antibody Labelled Colloidal Gold Particles and Their Applicability in a Sol Particle Immuno Assay (SPIA). Journal of Immunoassay, 11(1), 31-47. doi:10.1080/01971529008053256
[7] Introduction to Lateral Flow Rapid Test Diagnostics. nanoComposix. https://nanocomposix.com/pages/introduction-to-lateral-flow-rapid-test-diagnostics#target (accessed Feb 22, 2019).
[8] Cole, L. A. (1997). Immunoassay of human chorionic gonadotropin, its free subunits, and metabolites. Clinical Chemistry, 43(12), 453. Retrieved February 17, 2017, from https://www.ncbi.nlm.nih.gov/pubmed/9439438.
[9] Smith, S.; Govindasamy, K.; Govender, U.; Land, K. J. South African Journal of Science 2015 111(11/12), 1-10.
[10] Zhan, L.; Guo, S.Z.; Song, F.; Gong, Y.; Xu, F.; Boulware, D.R.; McAlpine, M.C.; Chan, W.C.W.; Bischof, J.C. “The Role of Nanoparticle Design in Determining Analytical Performance of Lateral Flow Immunoassays” U.S National Library of Medicine, National Institute of Health. 2017, 17, 7207-7212.
[11] Lin, B.; Guan, Z.; Song, Y.; Song, E.; Lu, Z.; Liu, D.; An, Y.; Zhu, Z,; Zhou, L.; Yang, C. “Lateral flow assay with pressure meter readout for rapid point-of-care detection of disease-associated protein” Royal Society of Chemistry. 2018, 18, 965-970.
[12] He, P.J.W.; Katis, I.N.; Eason, R.W.; Sones, C.L. “Rapid Multiplexed Detected on Lateral-Flow Devices Using a Laser Direct-Write Technique” U.S National Library of Medicine, National Institute of Health. 2018, 8 (4), 97.
[13] HCG pregnancy strip test. (2016, November 19). Retrieved February 17, 2017, from https://en.wikipedia.org/wiki/HCG_pregnancy_strip_test
[14] Lateral Flow Assay. (2014, March 29). Retrieved February 17, 2017, from http://www.dxdiscovery.com/lateral-flow-assay.html
[15] Tisone, T. C.; O’Farrell, B. Lateral Flow Immunoassay 2008, 1–26. https://pdfs.semanticscholar.org/7f56/785b54d6646d0498c0ee046f66911529df79.pdf