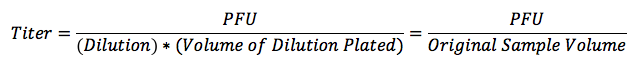IGEM:Virginia 2012/Protocols/Plaque Assay
Overview
Performing a plaque assay is one method of determining the concentration of a virus (the virus titer) in a sample. The first step is a series of ten-fold dilutions of the original virus sample. Then the virus is plated along with bacteria. After incubation, clear circular areas called plaques will form where the virus has infected and lysed the bacterial cells. The virus titer is calculated by counting the number of plaques on a countable plate (i.e., a plate that has 30-300 plaques, so that the number is statistically significant but still easy to count manually) and dividing that by the volume of the original sample which was added to that plate. Each of the plaques represents one plaque-forming unit (PFU).
Materials
Note: be sure to use filter pipette tips so that the pipettes do not become contaminated with phage.
- Phage suspension
- Bacterial culture
- Top agar
- Solid agar plates (one for each dilution + one for the control)
- Dilution tubes
- Microtubes for bacteria (one for each dilution + one for the control)
- Motorized pipette filler
- Tubes for top agar
Procedure
- Liquify top agar in a hot water bath. Turn the heating plate up to 400°C initially. Once the agar is liquid, turn the heating plate down to 130°C and add some more water.
- Label agar plates (type of bacteria, phage dilution or "Control", your initials, and the date) and place in 35°C incubator. Make sure to have a plate for each of the phage dilutions as well as one for only bacteria-- this is the control.
- Pipette 90 μl liquid LB medium into each of the dilution tubes.
- Mix the phage suspension by gently tapping the side of the tube with your fingers, and pipette 10 μl into the first dilution tube-- this is the 10-1 dilution.
- Mix the 10-1 dilution tube by tapping it with your fingers, and then pipette 10 μl from that dilution tube into the next dilution tube-- this is the 10-2 dilution.
- Continue this process until you have created all of the required phage dilutions. Taking it out to 10-8 should be sufficient.
- Mix the bacterial culture by gently tapping the side of the tube with your fingers, and transfer 30 μl into each of the bacteria microtubes.
- Mix the suspension in the first dilution tube, and then pipette it into one of the microtubes which contains bacteria. Repeat this process for each of the dilution tubes, making sure to mix the dilutions well before transferring them. There should be one bacteria microtube left over that has no phage added to it to serve as the control.
- Remove the plates from the incubator.
- Using a motorized pipette filler, add 3 mL of top agar to each of the agar tubes. So that the agar does not solidify, only fill three agar tubes at a time.
- Use a pipette to add the entire contents of a bacteria/phage tube to a top of top agar.
- Mix the top agar well, and immediately pour it onto an agar plate. Spread the agar mixture across the solid medium by tilting the plate back and forth.
- Repeat for all tubes.
- Wait for the agar to solidify completely, and then invert the plates and incubate at 35°C for 24-48 hours.
- Examine the control plate for growth and the absence of plaques. If there is not a lawn of growth on the control plate, put all plates back in the incubator until there is. If there are plaques on the control plate, there is contamination, and the assay must be repeated.
- Examine all plates and determine which one is countable. A plate must have 30-300 plaques to be countable. If there is more than one countable plate, then average the titer calculated from each. If there are no countable plates, perform further dilutions.
- Use the number of plaques on the countable plate to calculate phage titer. Phage titer = PFU / (dilution * volume of dilution plated). E.g., if you counted 50 plaques on the 10-8 dilution plate, the phage titer would be 5.0 * 108 PFU/μl = 50 PFU / (10-8 * 10 μl).
Notes
Please feel free to post comments, questions, or improvements to this protocol. Happy to have your input!
References
Contact
- Who has experience with this protocol?
or instead, discuss this protocol.