IGEM:Peking/2007/Count-Conjugation-Notebook/2007-8-13
From OpenWetWare
Jump to navigationJump to search
Tandem Ori-T by Mingzhi Qu, Ze Ren
Trimethoprim(TLC) antibiotic Efficiency Test
- new antibiotic,test efficiency, use Dh5α+pSB1A2(Amp+) as control.
- Preservative:1mg/mL in 2.5% acetic acid.
TLC(working solution): 0.5ug/mL 1ug/mL 2ug/mL 3ug/mL 4ug/mL 5ug/mL LB- amp+ R751(TLC+): ++§ ++ ++ ++ ++ ++ + / Dh5α+pSB1A2(Amp+): +¢ <+ <<+ <<<+ <<<<+ <<<<<+ + + empty: - / / / / - - /
- §:grown
- ¢:can be seen by centrifugal.
- <+ means less than + by centrifugal.
(8.15-8.16) Trimethoprim(TLC) antibiotic Efficiency Test(II)
- culture in solid LB.
TLC(working solution): 4ug/mL 8ug/mL 12ug/mL 15ug/mL 20ug/mL LB- Amp+ Tc+ R751(TLC+): ++§ ++ ++ ++ ++ ++ - - Dh5α+pSB1A2(Amp+): +¢ <+ <<+ <<<<+ - ++ ++ - pSC101(Tc+) +¢ <+ <<+ <<<<+ - ++ - ++
- §:grown
- ¢:can be seen by centrifugal.
- <+ means less than "+",still can be seen by centrifugal.
colony PCR Test for pSC101 Conjugation Test
- Conjugation Test
- Test Lb- R751 plate, Lb- pSC101 have the correct plasmid.
- Test plate:LB- R751, Lb- pSB101, R751-pSC101 X Dh5α-R0040, R751 X Dh5α-R0040
- primer :R751 OriT primer, pSC101 primer.
- according to <Colony PCR STANDARD PROTOCOL>
- PCR system contains(each well):
0.5 µl Primer 1(100uM) 0.5 µl Primer 2 2 µl dNTP(2.5uM) 2.5 µl 10X Taq Buffer 0.25 µl Taq 19 µl dH20 1 µl template -------------------------- ~25 µl Total
- PCR program condition 1: 94℃ 5min, 94℃ 30s, 51℃ 30s, 72℃ 45s, Go to step 2 for 29 times, 72℃ 10min, 4℃ end.
- PCR program condition 2: 94℃ 5min, 94℃ 30s, 55℃ 30s, 72℃ 45s, Go to step 2 for 29 times, 72℃ 10min, 4℃ end .
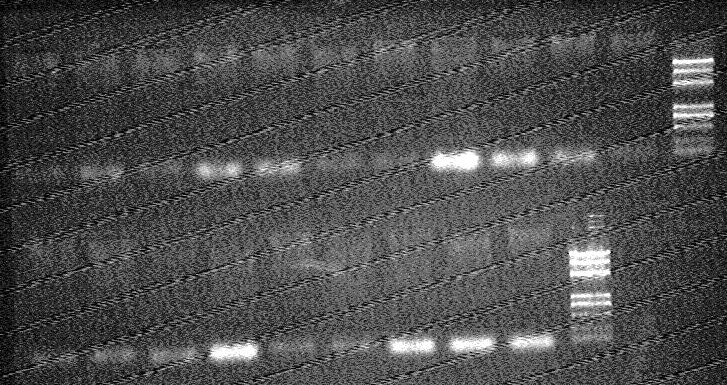
electrophorsis result
- top:from left to right
- 1-5 R751-pSC101 X Dh5α-R0040
- 6-10 R751 X Dh5α-R0040
- 11 R751
- 13 Maker(DL2000 plus)
- botton:from left to right
- 1-4 R751
- 5-10 psc101
- 11 Maker(DL2000 plus)
Lock & Key By Yu Tao
Efficiency Test
Transformation Result: Competent Cells II
- To our superise, there are colonies both in the experimental plate and the negative control plate.
- Select some colonies from the negative plate, culture them in liquid LB overnight for mini-prep.
- I decided to redo the efficiency test, with all possible controls. I use the good competent cells and DH5a as control groups, and culture each group on Amp+, Kan+ and Empty LB plate.
Recheck Test Result
| Competent Cells II | Good Competent Cells | DH5a -------------------------------------------------------------------- Amp+ | + | - | - -------------------------------------------------------------------- Kan+ | - | - | - -------------------------------------------------------------------- LB- | + | + | +
- Conclusion: I need to reprepare the competent cells again. ... -_-:
Mini-prep: R0010<-J01008 and R0040<-J01010
- Using Transgen mini plasmid purification kit.
- 50 uL per tube after purification, 2 tubes per type of plasmids.
Mini-prep Double Digesting Test Result
- Digesting all the newly minipreped plasmids with EcoRI/PstI.
- Each digestion system contains:
1 µl 10*H buffer 0.25 µl EcoRI 0.25 µl PstI 5 µl Plasmid 3.5 µl ddH20 -------------------------- 10 µl Total
- 37℃ culutre for 3 hours.
- from left to right:
- R0040<-J01010-1 @ EcoRI/PstI
- R0040<-J01010-2 @ EcoRI/PstI
- R0010<-J01008-1 @ EcoRI/PstI
- R0010<-J01008-2 @ EcoRI/PstI
- R0040 negative ligation control @ EcoRI/PstI
- R0010 negative ligation control @ EcoRI/PstI
- J01008 PCR product
- J01010 PCR product
- marker (DL2000 Plus)
Ligation and Transformation: J01010 and J01008 PCR product -> pEASY-T3 cloning vector
- For sequencing.
- Use Transgen pEASY-T3 vector.
- Transform the ligation product, result to be seen tomorrow.