IGEM:Peking/2007/Count-Conjugation-Notebook/2007-8-10
From OpenWetWare
Jump to navigationJump to search
Tandem Ori_T by Qu Mingzhi
Double digesting test for F-OriT_J23066_OriT_pSB1A2 & R0010
- use Double digesting test for gel extraction.
- F-OriT_J23066_OriT_pSB1A2 digestion system contains:
4 µl 10*M buffer 1 µl EcoRI 1 µl XbaI 20 µl Plasmid 14 µl dH20 -------------------------- 40 µl Total
- R0010 digestion system contains:
4 µl 10*H buffer 1 µl EcoRI 1 µl SpeI 20 µl Plasmid 14 µl dH20 -------------------------- 40 µl Total
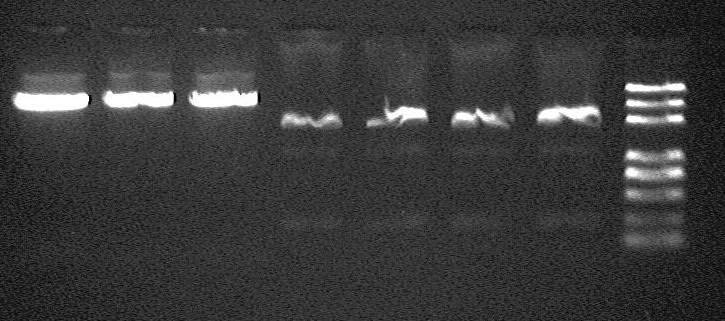
electrophoresis result before gel extraction
- from left to right:
- F-OriT_J23066_OriT_pSB1A2 -1 @ EcoRI/XbaI
- F-OriT_J23066_OriT_pSB1A2 -2 @ EcoRI/XbaI
- F-OriT_J23066_OriT_pSB1A2 -3 @ EcoRI/XbaI
- R0010 -1 @ EcoRI/SpeI
- R0010 -2 @ EcoRI/SpeI
- R0010 -3 @ EcoRI/SpeI
- marker (DL2000 plus)
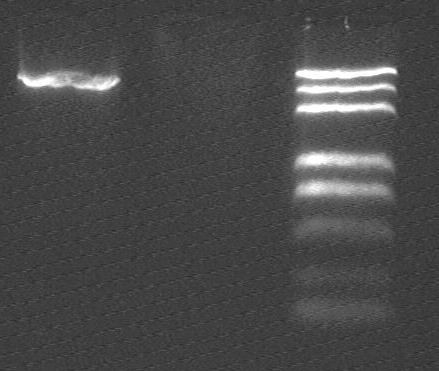
electrophoresis result after gel extraction
- from left to right:
1-3 F-OriT_pSB1A2_OriT_pSB1A2 vector @ EcoRI/XbaI
4-7 R0010 fragment @ EcoRI/SpeI
8 marker (DL2000 plus)
- Problems with R0010 gel extraction ,we will try ligation, we will do another copy of R0010
Ligation: R0010 -> F-OriT_J23066_OriT_pSB1A2
- Ligate the R0010 fragment and F-OriT_J23066_OriT_pSB1A2 vector
- use super fast T4 DNA ligase
- super fast T4 DNA ligase Ligation system contains:
1 µl F-OriT_J23066_OriT_pSB1A2 vector 7 µl R0010 fragment 0.5 µl ''super fast T4 DNA ligase'' 1 µl 10X T4 ligase buffer 0.5 µl dH20 -------------------------- 10 µl Total
- 16℃ for 20 min!
Amplification Culture of PlacI_F-OriT_J23066_OriT_pSB1A2 (fast T4 ligase)
- control plate received clones. this ligation experiment failed.
- Just for test:I will select a PlacI_F-OriT_J23066_OriT_pSB1A2 Colonies from Plate, Culture in liquid LB,waiting for mini-prep.
result:failed
- after mini-prep & digestion
- from left to right:
- PlacI-I741051 FAILED -1 @ EcoRI/PstI
- PlacI-I741051 FAILED -2 @ EcoRI/PstI
- I741051 @ EcoRI/PstI
- pSB1A2 fragment(seems there's some problem with this fragment)
- Maker (DL2000 plus)
Lock & Key By Yu Tao
Sequencing
- Send the precultured R0010<-J23066 to Invitrogen for sequencing.
- Use vf2 primer.
Key1 & Lock1 Efficiency Test
Rough Result of Key1 & Lock1 Efficiency Test
- Overnight incubation.
- Tip a drop of overnight liquid culture of each sample on a slide.
- From top to bottom:
- I7100-DH5a
- R0010.J23066(Key)-DH5a
- R0010.J23066(Key) and R0040.J23078.E0040.B0015(Lock)-DH5a induced by IPTG
- R0010.J23066(Key) and R0040.J23078.E0040.B0015(Lock)-DH5a not induced by IPTG
- DH5a negative control.
- Conclusion: It seems that most of the results conform to the expectation. The lock can really 'lock' the EGFP, and the key can unravel it. However, IPTG appears to be usefulless, or even has negative effect on the efficiency of key. Nevertheless, a more accurate test will be carried out at the end of August.
Prepare Competent Cells
- There are only 20 or so tubes of competent cells left.
- Newly prepare about 50 tubes of competent cells, 100uL/tube.
- Trasform 1uL I7100 minipreped plasmids to see the efficiency.
PCR J01010 and J01008
- The primers of new key(J01008) and new lock(J01010) have arrived.
- We are going to zip the primers together in order to produce a longer integral part.
- All primers: stored as 100uM.
- PCR system contains:
For J01010: For J01008:
1 µL Primer 1
0.5 µL Primer 1 1 µL Primer 2
0.5 µL Primer 2 1 µL Primer 3
4 µL dNTP 4 µL dNTP
0.5 µL Taq 0.5 µL Taq
5µL 10 X buffer 5µL 10 X buffer
38.5µL dH20 37.5µL dH20
-------------------------- --------------------------
50 µl Total 50 µl Total
- Primer final concentration 1uM.
- Add a drop of liquid paraffin to each system.
- PCR program setting:
For J01010 For J01008 Step1 94℃ 5min Step1 94℃ 5min Step2 94℃ 30s Step2 94℃ 30s Step3 57℃ 30s Step3 55℃ 30s Step4 70℃ 30s Step4 70℃ 30s Step5 Go to step 2 for 4 times Step5 Go to step 2 for 3 times Step6 72℃ 10min Step6 72℃ 10min End End
Electrophorsis Result
- from left to right:
- J01010 PCR product
- J01008 PCR product
- marker
J01010 & J01008 PCR Product Purification
- Use Transgen EasyPure PCR Purification Kit.
- 50uL per tube after purification.
Double Digestion: J01010 & J01008
- Digesting J01010 and J01008 with PstI and XbaI.
- Each digestion system contains:
4 µl 10*M 1 µl XbaI 1 µl PstI 10 µl PCR product 4 µl BSA 20 µl ddH20 -------------------------- 40 µl Total
- 37℃ culutre overnight (at least 12 hours).
Sequecing
Sequecing Result of Sample #1
- The report says it fails to sequece Sample #1, damn it...