IGEM:MIT/2008/Brainstorming
This is the new brainstorming page for our iGEM project. Please edit this page if you have suggestions or ideas.
The previous brainstorming page is at IGEM:MIT/2008/Brainstorming Old
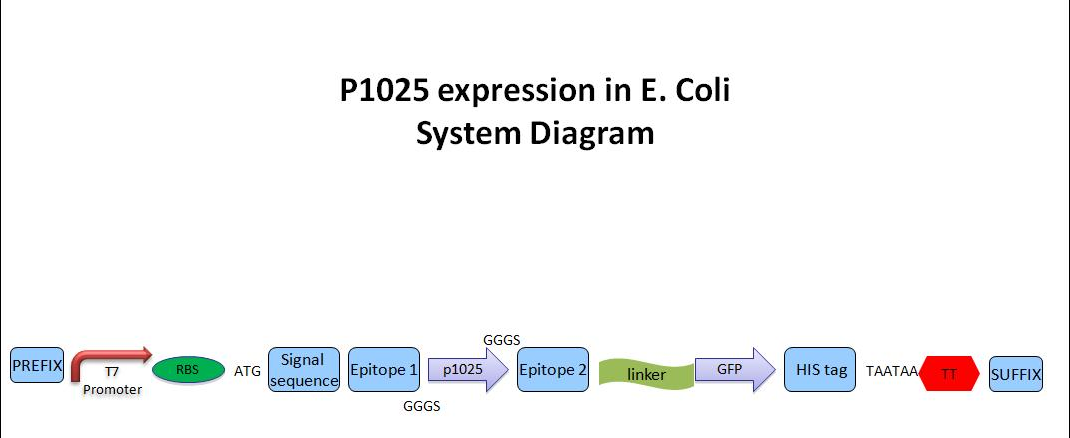
Expression of a p1025-containing peptide in E. coli
References
PMID 9920267
A synthetic peptide adhesion epitope as a novel antimicrobial agent.
PMID 12536251
Overview of tag protein fusions: from molecular and biochemical fundamentals to commercial systems.
Materials
- The entire transcription unit will include
- T7 promoter for over-expression (IPTG inducible)
- ribosomal binding site
- secretion signal (~30AAs) for L. bulgaricus; whether or not it works in E. coli is not critical
- 1st epitope with spacer(~10AAs)
- p1025 peptide (20AAs)
- spacer and 2nd epitope (~10AAs)
- a protease cleavage site (target sequence of thrombin or TEV) plus a Gly and Ser rich flexible linker (~20AAs total).
- GFP (~230AAs)
- 6xHis tag for affinity purification and on-column digestion with the protease, if necessary
- double STOP codon
- transcription terminator
Adhesion of Streptococcus mutans to hydroxyapatite (HA)
Reference
PMID 9062560
Inhibition of Streptococcus mutans adsorption to hydroxyapatite by low-molecular-weight chitosans.
The materials and methods section describes an indirect plating assay to quantify binding of S. mutans to HA. Results in Table 1 indicates that this method can be used to quantify inhibition of binding by a competitor molecule. In their study, the molecules are sugar polymers.
Materials
- HA beads (from Fluka, 80-200 micro-meter grain size)
- saliva as source of the glyco-protein (one vendor is found through Google)
- S. mutans and handling protocol (Chia is contacting Daniel Smith and Martin Taubman of the Forsyth Institute to acquire a suitable strain. Materials and methods section in the reference has a brief description on how to grow S. mutans.)
- synthetic p1025 and p1125 as positive and negative controls?
Secretion of a p1025-containing peptide from Lactobacillus bulgaricus/lactis
References
PMID 12788739 (a secretion signal sequence described; can use the promoter, RBS and secretion signal for this protein for p1025)
Determination of the domain of the Lactobacillus delbrueckii subsp. bulgaricus cell surface proteinase PrtB involved in attachment to the cell wall after heterologous expression of the prtB gene in Lactococcus lactis.
PMID 11772607
Electrotransformation of Lactobacillus delbrueckii subsp. bulgaricus and L. delbrueckii subsp. lactis with various plasmids.
Materials
- Lactobacillus bulgaricus or lactis (check with Tom Knight first. Otherwise request from another lab or purchase from ATCC)
- growth medium: Ligia recommends the MRS medium. It can be purchased from Difco or Oxoid http://www.oxoid.com/uk/blue/index.asp
- Gas Pak to create an anaerobic/micro-aerobic environment. (Chia's lab can lend a few boxes and bags as anaerobic chambers. We can then purchase the disposable chemical envelopes known as "Gas Paks" that reduce oxygen content in a closed system.)
- Expression vector: BBa_I742103 in registry is a shuttle vector for E. coli and Lactobacillus. Let's look up this vector to see whether people have used it for protein expression. Alternatively we can get some of the plasmids described in PMID 11772607.
Project road map
Stage 1
- Construct parts for E. coli for production of the p1025 fusion peptide. Obtain antibodies against epitopes and maybe p1025 itself.
- Get the hydroxyapatite(HA)-S. mutans binding assay to work in our hands.
- Practice growing and transformation of Lactobacillus. Design parts for Lactobacillus.
Stage 2
- Make E. coli produce the p1025 fusion peptide.
- (GOAL #1) See if the p1025 fusion peptide from E. coli can inhibit binding of S. mutants to HA beads. Remove GFP if necessary.
- Engineer Lactobacillus
Stage 3
- (GOAL #2) Verify production and secretion of p1025 fusion peptide by Lactobacillus by Western (and/or microscopy if we decide to have a GFP-containing part in parallel).
- (GOAL #3) Can the yogurt reduce binding of S. mutans?
Fundrai$ing!
Drew has said that he can get in touch with some MIT alumni and the MIT development office. We can send them the brochure and a letter to explain our goal, project and budget.