IGEM:Imperial/2010/Detection module/2 components systems/ComXadditional
ComX additionalinfo
pre-ComX processing
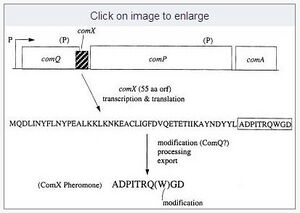
ComX is produced as a 55aa pre-ComX precursor.
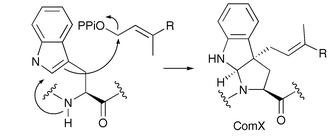
ComQ appears to carry out two enzymatic steps, MODIFICATION and CLEAVAGE (Ansaldi et al 2004) (ComQ and ComX are both necessary and sufficient for production of mature ComX, which is released as isoprenylated peptide).
Ansaldi et al (2004) determined the following:
- both C-terminal and N-terminal sequence motives of ComX are important for interaction with ComQ: "The yield of pheromone activity with the chimeric pre-ComX molecule was intermediate between that obtained with the comQRO-H-1 comXRO-E-2 and comQRO-E-2 comXRO-E-2 combinations, suggesting that ComQ interacts with both the conserved N-terminal domain of pre-ComX and the less conserved C-terminal moiety (Table 3)."
- Only the conserved sequence of the mature ComX part (C-terminal) appears to determine the specific type of isoprenylation of the mature ComX sequence at the central W: "We show that the choice of isoprenyl derivative is determined by the C-terminal (mature) sequence of pre-ComX rather than by the ComQ protein."
Furhter, while the cleavage recognition sites have not been determined, the cleavage sites have been found for several subgroups of ComX:

With this knowledge we might be able to design a secretable peptide in which a C-terminal ComX is still modified by ComQ, but which is not cleaved by the enzyme. A possible approach would be to alter the aa-sequence N-terminal of the determined cleavage sites without altering the rest of the protein. Additionally a Sec-secretion signal-sequence could be added to the N-terminus of the overall protein.
Additionally, structures are avaliable for ComQ and ComP. The structures for ComA are limited to the C-terminal DNA binding domain-region which includes around 100/214 aa. The pathway of modification of ComX is also known.
- ComQ - structure: link
- ComP - structure: link
- ComA - structure: Hobbs et al (2010)