IGEM:IMPERIAL/2009/M3/Assays/F2620/calib/fluor/expt2
Relating intracellular fluorescence to GFP molecules
Aims
- By relating intracellular [GFP] rather than extracellular [GFP] to fluorescence observed, cells do not have to be lysed. Therefore, fluorescence can be measured at various time points for a particular culture of cells.
Equipment
- Spectrophotometer
- 600nm absorbance filter
- 17mm tubes
- 96 well plates
Reagents
Media
M9 Minimal Media
Disodium Phosphate (12.0g)
Potassium dihydrogen phosphate (6.0g)
Sodium Chloride (1.0g)
Ammonium Chloride (2.0g)
Magnesium Sulphate (0.75g)
Glycerol (5.0g per L)= 54.39uM 0.5%
Glucose (0.5g per L) = 2.78mM 0.05% per 100ml
Others
IPTG (2 g)
Protocol
Day 1 4PM:
Omit this day if there are already available cells in culture and do the minimal media preparation in day 2
Things needed
- Minimal Media constituents
- 17mm tubes
1) M9 Minimal Media Preparation:
- Measure out the following reagents and dissolve them in 1000ml of sterile H20:
Disodium Phosphate = 6.0g
Potassium dihydrogen phosphate = 3.0g
Sodium Chloride = 0.5g
Ammonium Chloride = 1.0g
Glycerol (5.0g per L)= 54.39uM
Glucose (0.5g per L) = 2.78mM
2) Inoculation of cells
- Inoculate single colonies of E. coli cells into 17mm tubes containing 5 ml of the pre-warmed (37°C) normal supplemented M9 medium with kanamycin (20 ug/ml)
- Grow the cultures for O/N with spinning at 70 rpm.
Day 2 AM:
Start from here if there are already available cells in culture
Things needed
- IPTG
- Minimal Media constituents
- 17mm tubes
1) IPTG solution Preparation:
- 1g of IPTG dissolved in 4ml of dH2O (filter sterilize) to get 1M IPTG
2) Growing up of cultures
- Dilute the cultures which are at a high cell density 1:1000 into 20 ml of fresh media in a 100ml flask and grow the cultures at 28°C in 28°C incubator O/N
Things needed
- Spectrophotometer
- 600nm absorbance filter
Monitering of OD and fluorescence
- Now prepare the lysozyme solution by adding 2mg of lysozymes to 1ml of ddH2O, mix thoroughly and store on ice.
- The OD is monitered by transferring 200ul aliquots into a 96 well plate and measuring the absorbance at 600nm.
- After the OD reaches 0.7 (should be immediately), transfer 200 ul aliquots from the culture into the labelled flat-bottomed 96 well plate with IPTG (all except well 1)and mix well.
- Incubate the plate in a multi-well spectrophotometer at 28°C (water bath?) and assay with an automatically repeating protocol of absorbance measurements (600 nm absorbance filter, 0.1 second counting time through 5 mm of fluid) and shaking (3 mm, linear, normal speed, 15 seconds)
- Repeat the absorbance and fluorescence measurement every hour during mid-exponential growth for 6 hours
- Determine background absorbance by measuring control well. This should be subtracted from subsequent absorbance readings.
- For each hourly sample, add 4μl of the lysis solution, to the 200ul culture drop by drop mixing gently in between drops.
- Incubate at 28°C for 30 minutes with occasional mixing - Lysis should be indicated by a change in the viscosity of the culture.
- In the 96 well plate, there will be the following wells:
Well 1: 0 ul of IPTG
Well 2: IPTG for 1hr
Well 3: IPTG for 2hr
Well 4: IPTG for 3hr
Well 5: IPTG for 4hr
Well 6: IPTG for 5hr
Well 7: IPTG for 6hr
Well 8: IPTG for 7hr
Data processing
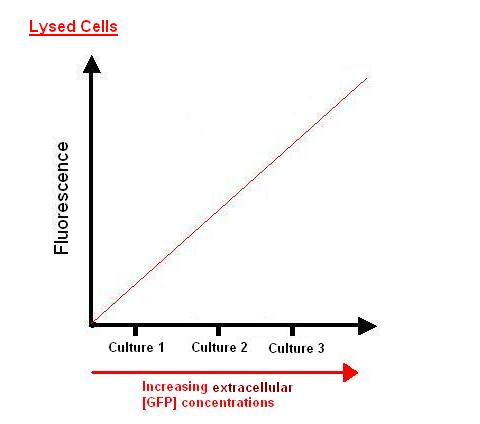
- From the standard curve in experiment 1, we can determine [extracellular GFP] from the normalised fluorescence reading obtained.
- After lysing the cells, we know the [intracellular GFP] for each duration of IPTG induction by relating fluorescence to [lysed extracellular GFP].
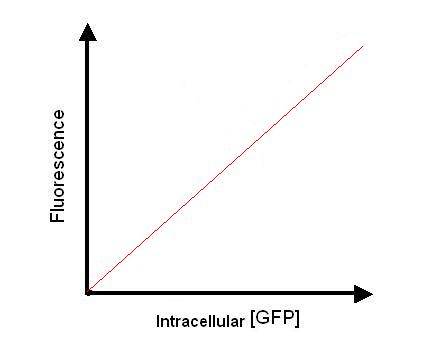
Therefore, now we can plot [intracellular GFP] against fluorescence observed.