IGEM:IMPERIAL/2009/Assays Protocols/Colanic Acid
Colonic acid
Qualitative Assay:
Visual analysis and electron microscopy.
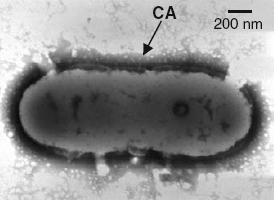
Electron micrograph of colanic acid encapsulated cell.
[1]
Quantitative Assay:
The fastest way to measure volume of colanic acid production will be to use PACKED CELL VOLUME ASSAYS.
This approach was inspired by a recent paper which used this method to quantify colanic acid production. [2]
PCV Assay:
Measure OD at 600nm to determine cell density.
Add to PCV tube and centrifuge.
Measure height of pellet.
PCV tubes can be purchased from:
http://216.15.207.230/cat//prodprice2_Detail.cfm?ID=2221
A full protocol for the procedure can be found here: http://216.15.207.230/userfiles/File/PCV_Tube_protocol_2.pdf
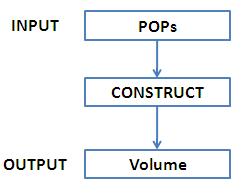
An input of POPs would be converted to an output of packed cell volume:
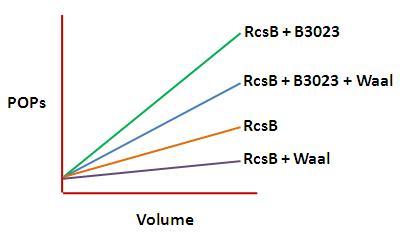
This data could be displated on a graph as shown below:
Quantification of total EPS paper
1. Incubate cells for 12 hours at 37 °C. After this, collect about 60 mg of culture from the surface of LB agar plates.
2. Resuspended cells in 1 ml of sterile distilled water (use vortex).
3. Determine cell density by measuring OD at 600 nm.
4. To inactivate EPS-degrading enzymes and release EPS:
a. Boil resuspended culture for 10 min.
b. Cool to room temperature.
c. Centrifuge at 16000g for 10 min.
d. Save supernatant (for quantification) by freezing at - 20 °C.
Note: Perform all experiments with two independent cultures.
Colonic acid assay
This method is based on the specific difference in absorbance at 396 and 427 nm after reacting fucose with sulfuric acid and cysteine hydrochloride
1. Measure the fucose concentration to determine colonic acid concentration
2. Use a L-fucose (Acros Organics) calibration curve from 10–60 g ml- 1
3. normalise these values by cell turbidity at 600 nm
4. For negative control, glucose was assayed. It should not produce a significant signal.
Note: Perform all experiments with two independent cultures.
Another assay for colonic acid from here
Principle : Measure fucose concentration (actually, we can also use a test kit from sigma
1. Dilute 10 to 100 μl of the colanic acid preparation to 1 ml with distilled water
2. mixed it with 4.5 ml of H2SO4/H2O (6:1; v/v) at room temperature
3. heat at 100 °C for 20 min
4. cooled it down to room temperature
5. For each sample measure absorbance at 396 nm and 427 nm was measured
a. directly (control sample (A-co))
b. After addition of 100 μl of cysteine hydrochloride (cysteine sample (A-cy)).
6. Heat with H2SO4 to yield brown products absorbing between 396 nm and 427 nm.
7. Subtract the absorption due to this unspecific reaction from the total absorption of the sample: A396-co and A427-co were subtracted from A396-cy and A427-cy, respectively, to obtain ΔA396 and ΔA427.
8. Values of (ΔA396–ΔA427) were directly correlated to methylpentose concentration by using a standard curve obtained with a fucose concentration ranging from 5 μg/ml to 100 μg/ml.
- Navasa N, Rodríguez-Aparicio L, Martínez-Blanco H, Arcos M, and Ferrero MA. Temperature has reciprocal effects on colanic acid and polysialic acid biosynthesis in E. coli K92. Appl Microbiol Biotechnol. 2009 Mar;82(4):721-9. DOI:10.1007/s00253-008-1840-4 |
- Ionescu M and Belkin S. Simple quantification of bacterial envelope-associated extracellular materials. J Microbiol Methods. 2009 Sep;78(3):302-6. DOI:10.1016/j.mimet.2009.06.020 |