IGEM:IMPERIAL/2007/Projects/brain dump
On Diffusion Through The Biofilm
Effective diffusion depend on solute type and density of biofilm. Mean relative effective diffusive permeability (De/$aq) of different solutes:
- inorganic anions or cations - 0.56
- nonpolar solutes (with molecular weights of 44 or less) - 0.43
- organic solutes (molecular weight greater than 44) - 0.29
Effective diffusive permeabilities decrease sharply with increasing biomass volume fraction suggesting a serial resistance
model of diffusion in biofilms. Large solutes are effectively excluded from microbial cells, small solutes partition into and diffuse within cells, and ionic solutes are excluded from cells but exhibit increased diffusive permeability (but decreased effective diffusion coefficients)due to sorption to the biofilm matrix. (21)
When substances diffuse into biofilms, they are not distributed evenly, due to the clusters of cells within the biofilm. This provides protective pockets for biofilms, from which regrowth can occur. (22)
Notes
The metabolism of cells is different at different depths within the biofilm. At the deepest level of a mature biofilm, anaerobic respiration may predominate.
Clusters of nitrite oxidizers crowd around distinct clusters of ammonia oxidizers (20, 29) (see references above). Thus, is the metabolic waste product of the ammonia oxidizers, nitrite, made available to the bacteria that can use it as a substrate for oxidation. The activities of these commingled species lead to the consumption of ammonia and oxygen near the biofilm surface and the simultaneous production and consumption of nitrite slightly below the biofilm surface.
Initial ideas
- be able to detect that it is in the presence of a biofilm on
- contact lenses
- catheters (which type?)
- surgical instruments
- kitchen utensils
- pipes
- food samples
- IUDs
- be able to adsorb to, and penetrate, the biofilm
- survive long enough in the biofilm in order to disperse it (probably using DspB)
- be able to disperse 99% of the biofilm material
- Parameters to quantify
- maximum time for biofilm dispersal
- minimum amount of biofilm to be dispersed
- environmental conditions for system to work in (temperature, pH, pressure)
- any others..?
On Biofilm Formation
Biofilm Formation
There are three stages of biofilm formation:
- The initial attachments of microorganisms to a surface.
- After a certain density is reached a 'slime capsule' is built up. This capsule is composed of exopolysacchorides that are secreted from the cell.
- The final stage is the growth of the biofilm to form the distinct architecture that is associated with biofilms. Biofilms are composed of layers, much like our own skin, these layers has very specific architecture.
Toggle Switch Summary
Toggle Switch is interfaced with a transgenic Quorum signaling pathway that detects AHL and transcribes a gene which inhibits the growth of biofilm when cell population reaches a threshold density. We seek to use this for keeping the amount of biofilm at a maximum level. In addition to stopping growth the toggle switch will also turn the biofilm green when the threshold is reached.
Adhesion Problems
Factors Affecting Bacterial Adhesion:
- Biological:
- Osmolarity & Growth Phase: OmpZ/OmpA
- Membrane Stress : Cpx pathway & Rcs Pathway
- Physical:
- Hydrophobicity of surface
- Roughness of surface
- [PVC is easily bound to - Harvard1998]
Destroying biofilms
The process of biofilm investigation carried us through a host of different-natured biofilms, including those produced by MRSA and Pseudomonas Aeruginosa. The scope of implementing a bio-film detector, and possibly disperser ("saboteur"), proved too ambitious to undertake over a limited project period of 10 weeks. It was determined, therefore, that the well-studied E-Coli biofilm would be investigated further, and that the Detector would in fact be applied to this biofilm platform. More detailed information concerning the previously investigated Biofilms can be found here: Future Extensions of Biofilm Detector.This is important to consider in future applications of the "Biofilm Detector".
Recently, a paper[1] was published describing the use of an engineered bacteriophage that would also produce DspB, an enzyme that breaks down one of the major structural components of biofilm. However, it was pointed out that bacteriophages target very specific cells, which may not be present in the biofilm under consideration. Furthermore, bacteria often evolve defences against phages. Thus, although the phage technique has proven that the principle of infecting a biofilm is a good strategy to disperse it, it is far from becoming usable outside the tightly controlled lab environments.
This project proposes an alternative method of biofilm dispersal. In short, a bacteria will be modified to express DspB as a response to three input signals - quorum sensing, biofilm presence, and anaerobic conditions. These bacteria can then be sprayed over a biofilm, which they infiltrate, and disperse.
There are three major assumptions that may prove problematic in this proposal:
- That the saboteur bacteria will be able to penetrate the biofilm
- That the saboteur bacteria will survive in the biofilm
- That the quorum sensing mechanism will work
A list of references can be found at the bottom, that may be able to help addressing these concerns.
The Hrp system may be used with its three inputs: biofilm detection, quorum sensing, and anaerobic conditions sensing (inverted in V input). By inverting the anaerobic conditions sensing, a basal level (the 10% leakage of the V-inhibition) of DspB will be produced. As soon as anaerobic conditions are reached, V-expression is inhibited (the inversion) which kicks the Hrp system into full swing.
On Escherichia coli Bio-Films
E. coli is ubiquitous in human intestines, and is normally benign. However, a few strains can cause infections, of three types: urinary tract infection (UTI), neonatal meningitis, and intestinal diseases (gastroenteritis). Of these, urinary tract infection is the most common nosocomial infection, and is caused by E. coli in abou 80% of all cases. Further, the UTI infective strains are well documented[1].
Once again, creating a system that can detect the presence of the particular infectious strains would serve two useful purposes: one, it could help in the surveillance and prevention of e.coli infections. Two, and perhaps more importantly, it could serve as a proof of concept that synthetic biology can be used to produce cheap, reliable, and fast sensors for particular pathogens. Also, E.coli is usually used as an indicator for human fecal contamination of water.
One question is that, if e.coli is used as the chassis for the detection system, how would one avoid having the system activate itself? A method for separating the target e.coli from the system e.coli must be devised.
Other nosocomial infections
Apparently, according to WikiPedia, urinary tract infections account for 40% of nosocomial infections (note, reference not found). Also, the most common cause of urinary tract infections is e.coli (again, note the lacking references - they are mostly about acupuncture, and not directly related to statistics on infections).
Defining the Project
After having several people spend many hour researching the possibility of detecting S. aureus biofilms, it looks like this will be difficult to achieve within the time restrictions of the iGEM project. However, a proof of concept may be achievable instead: we might be able to demonstrate that we can build a system that detects the presence of minimal e.coli biofilms, and then reports back quickly and strongly. The pathway would involve detection, amplification, and reporting. Proving that the concept works with e.coli might then inspire further work into a similar solution for MRSA. At this point, we still have several options going forward: try it with Bacillus Subtilis, change target to Pseudomonae, or change target to e.coli.
Biofilm formation on catheters
- AHL is produced by the clinical biofilms http://aem.asm.org/cgi/reprint/64/9/3486
Detection Targets
- After looking through the literature tried to identify possible targets for detection of biofilms.
- Can generally split them down into three catagories;
- Signaling molecules - This really concerns quorum sensing.
- Exopolysacchorides
- Attachment molecules
- more?
Quorum Sensing
- Two possible targets for quorum sensing are the:
- Las pathway
- Rhl pathway
- Lux pathway
At the outset of this investigation into biofilms we looked at specific target biofilms that we could target. We tried to consider biofilms that have importance within medicine and in particular, are a problem in terms of disease and detection.
Staphylococcus aureus
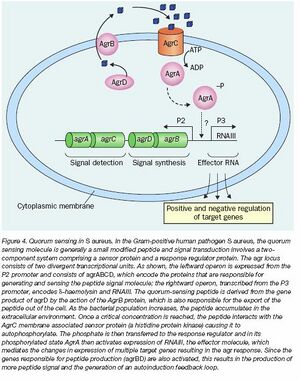
- We have identified a pathway that is unique to Staphylococcus aureus this system is called the Arg system. We investigated this and the potential target of this pathway. A mechanism of this pathway is shown to the right.
- However, upon further investigation we came across several problems:
- This type of quorum sensing is not with soluble 'signal inducers' but small peptides. The small peptides are cannot freely diffuse in and out of cells and require specialized membrane equipment in order for the cells to uptake the peptides. This is a problem because Staphylococcus aureus is a gram positive bacterium and the model organism that we would ideally use is E.coli which is a gram negative bacterium. The difference in membrane structures posses a problem and in previous studies, attempts to apply the gram postive equipment in E.coli has failed.
- In addition we failed to find a way to specifically targeted Staphylococcus aureus biofilm signaling or indeed the adhesion and exopolysacchorides of the biofilm.
- Cámara M, Williams P, and Hardman A. Controlling infection by tuning in and turning down the volume of bacterial small-talk. Lancet Infect Dis. 2002 Nov;2(11):667-76. DOI:10.1016/s1473-3099(02)00447-4 |
A biofilm is a organizations of microorganisms on various biotic and abiotic surfaces. They are naturally occurring and are key for many organisms survival, in addition to being key for many organisms infectious ability. There are a variety of fields concerned with biofilms, notably medical and industrial. Medical interests in biofilms are in controlling the growth harmful biofilms forming. In addition, there has been interest for industrial applications, for example, biofilms can grow and block pipes.