IGEM:IMPERIAL/2007/Projects/Experimental Design/Improve Methodology/Results3
In Vitro Testing
Aim
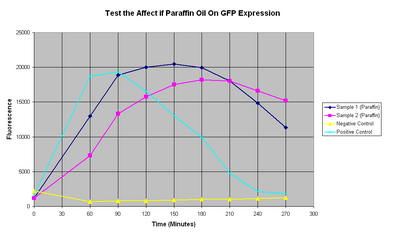
To determine if GFP can express and fold correctly in vitro and with 30ul paraffin oil.
The testing was carried out Tested 12-09-2007
Results
Rate of Expression
Here is a link to the Raw Data
Rate of Evaporation
Measured 17hours after reaction was started
| Well | Well Contents | Paraffin Oil(ul) | Initial Volume(ul) | Final Volume | Loss by Evaporation(ul) | |
|---|---|---|---|---|---|---|
| D5 | 60 ul Cell Extract + DNA | 30 | 90 | 20 | 70 | |
| D7 | 60 ul Cell Extract + DNA | 30 | 90 | 25 | 65 | |
| E6 | 60 ul Cell Extract + DNA | 30 | 90 | 25 | 65 | |
| E9 | 60 ul Cell Extract + DNA | NA | 60 | 0 | 60 | |
| F5 | 60ul water | NA | 60 | 0 | 60 | |
| F6 | 60ul water | NA | 60 | 0 | 60 | |
| F7 | 60ul water | NA | 60 | 0 | 60 |
All that was left at the end of the reaction was oil.
Discussion
Rate of Expression
If we compare these results to previous results for pTet-GFP in vitro at 37 °C then we notice several interesting differences:
- The positive control behaves strangely, not reaching high levels and also it does not maintain steady state.
- The total fluorescence is dramatically reduced in this expreriment compared to earlier ones
- The decay of the steady state appears to be much quicker
Some of the variables between the data previously collected and this set of data are as follows:
- Different batches of cell extract and DNA were used
- For the positive control this exp. no lid was placed on top, allowing increased exporation compared to previous data
- The sampling time was kept at 30 minutes, whereas before it varied from 10 to 30 minutes.
1. Positive Control
Ideally we would compare our positive controls to previous results and see if it reproduces what we have previously seen. However, because the positive control had no lid then we are not able to compare because of the additional evaporation that can affect the fluorescence output.
What is interesting is that the positive control initially has the same rate of GFP production as the 37°C for the first 30mins. After this our experiment decreases in rate until it suddenly starts to stop around 60mins. The reason for this could be
- The difference in DNA or Cell extract batches used which must not only limit total fluorescence output and also the length of expression
- The evaporation from the well that must decrease the total fluorescence and limit the total length of expression
2.Total GFP Output
Because we cannot directly compare back to previous data it is difficult to identify why the results are this way, the two causes that seem most likely are:
- The difference in cell extract batch and DNA prep used have a dramatic affect on the expression of GFP
- The available oxygen is limited by the parafin oil and so limits the number of GFP molecules that have correctly folded chromophores and so expression is unlikely to be affected but the fraction of GFP molecules that we can visualise is limited
We need to test to see which of these is correct. Several tests that we can consider are:
- To test this we need to first test the DNA concentration and then test the cell extract. If it is possible to reach higher levels of expression with greater concentrations of DNA this needs to be investigated.
- To investigate the rate of evaporation
Rate of Evaporation
Following a 17 hour period we measured the volume of the cell extract under mineral oil. The results show that there was significant evaporation for the experiments. When we compare these results to those of the test with water we see very different results, where we saw very little evaporation. There are several reasons for the difference:
- The first exp. with water and oil, the plate was left in the incubator for the duration without moving it, whereas this sample was moved every 30minutes. Although we tried to minimise movement, we may have dirupted the oil layer.
- That the cell extract is denser than the oil and so the oil does not form an efficient layer
- The problems of measuring out oil which were resolved in the second exp, may have made the first exp unreliable.
- Sampling the plate may have cause evaporation that the first exp. with water could not have stopped
To try to resolve these problems we could conduct the following experiments:
- Repeat the evaporation exp with water, however make several key changes. Sample to water to expose it to similar conditions to those of our exp. and also measure out the oil more accuratly. In addition test a greater range of oil volumes
- Test the density of the mineral oil and paraffin oil against the cell extract - we can use home made cell extract to test this.