IGEM:IMPERIAL/2006/LabCalendar/2006-8-16
From OpenWetWare
Jump to navigationJump to search
12D->2H
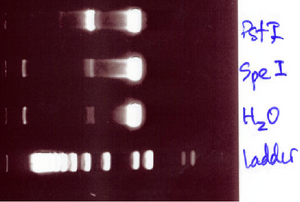
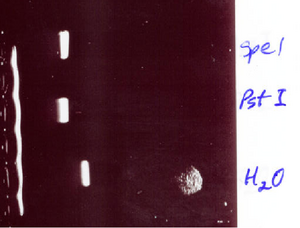
- The gel that was run yesterday was inconclusive as to whether the DNA has the problem or the enzyme.
- We will run 7A using the same procedure to see if it is cutting the plamid at all. We are again using the same enzymes with yellow buffer
- SpeI & PstI were able to successfully cut 7A, so it is not a problem with the enzyme!
- We will try re-electroporating part 2H again using the DNA from the parts registry to see if we can get it to work tomorrow
Reculture J37016
- Plates were quite good and we managed to isolate single colonies
- We took a total of 4 cultures to miniprep tomorrow or later this afternoon to see if we get what we expect.
Ligation of 6B+RS+12D+24A
- The insert and vector were removed from the overnight gel
- 20 uL ligation performed
Electroporations
- J37015RS (6B->RS->12D->24A)
- 1M B0021 removed from iGEM plate
- 4G S01656 removed from iGEM plate 2, grown on kanamycin plate
- 2H I13033 re-electroporated to see if the SpeI site has mutated on the gene
- Didn't bother with adding LB, since we only have LB Amp and no Kanamycin. So just plated the electrocompetent cells straight onto plate. Hopefully this doesn't affect growth.
Set up Maxipreps
- 6B F2620 (ran out of maxiprep)
- 1I B0015 (remaxiprep, since whereabouts of previous maxiprep unknown)
To do tomorrow
- Maxiprep parts above
- Reculture the electoporations into liquid media
- Ligate 1I and 9G to make part 2H, in case 2H fails yet again
- Miniprep the parts from electroporation, if possible
- Miniprep J37016 which is currently in shaker since 11 am Wednesday