IGEM:Hong Kong HKUST/Investigations/Restriction enzyme activities at 37°C vs. 20°C
Authors
AbstractIt is believed that enzymatic activity is highly dependent on temperature. We had performed experiment to compare the efficiency of EcoRI-HF® and SmaI (Restriction Enzymes) in digesting DNA Plasmid of Escherichia coli (pPD95.75 and pBS II KS+) at 37oC and 20oC. The result indicated that the efficiency of restriction enzymes in the above temperatures is similar under addition of 5U restriction enzyme. IntroductionRestriction enzyme is a kind of enzyme that cuts DNA at specific parts of the sequence known as the recognition site or restriction site. During restriction digestion, it cleaves the sugar-phosphate backbone at restriction site of each strand. Like all enzymes, activity of restriction enzyme is also thermo-dependent. Thus, restriction enzyme will lose its biological activity and may even denature under unsuitable temperature. Restriction enzymes such as EcoRI-HF® and SmaI are frequently used in DNA processing, their efficiency is directly related to the experimental result. Therefore, we would like to investigate how efficient restriction enzymes, more specifically EcoRI-HF® and SmaI, are under room temperature (20℃) and 37 ℃. Methods and Materials4 sets of similar experiments were carried out. Set 1 & Set 2 were performed as follow: E. coli (SURE) harboring pPD95.75 or pBS II KS(+) was inoculated into Lysogeny Broth for overnight incubation. Plasmids were then extracted with GTpure™ Plasmid miniprep purification kit. The concentration and quality of purified DNA sample was assessed by NanoDrop 2000 spectrophotometer. Approximately 1μg of pPD95.75 or pBS II KS(+) was digested with 5 units of EcoRI-HF® (NEB, R3101S), in 1X NEB CutSmart™ Buffer (NEB, B7204S). Reactions were incubated at 37°C for 1 hour. Corresponding to each type of DNA molecule, with the enzyme replaced by the same amount of double-distilled water, two negative control groups were also employed. Another identical set of reactions, including the negative controls, was incubated at 20°C for the same amount of time. Approximately 60ng of DNA samples obtained from digestion were analyzed on a 0.8% agarose gel, which was post-stained by 0.5μg/ml ethidium bromide for visualization of DNA fragments. For Set 3 & Set 4, they were similar to Set 1 and Set 2, but with SmaI(NEB, R0141S) as the restriction enzyme instead of EcoRI-HF®. Results and InterpretationsFor Set 1 and Set 2: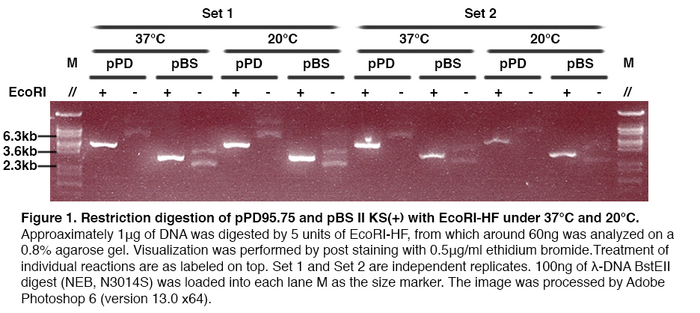 As shown in Figure 1, in both sets of reactions, all negative control groups exhibit spectrum of bands, whose exact pattern is dependent on the type and source of DNA sample. With the addition of EcoRI-HF®, the pattern in samples containing pPD95.75 is replaced by a single sharp band between 3.6kb and 6.3kb while in those containing pBS, a different single sharp band between 2.3kb and 3.6kb is detected. Comparison of each digestion reaction with its corresponding negative control group suggests all digestion reactions are successful. Successful digestion of both types of plasmids indicates that the enzyme used is of proper enzymatic activity. Comparing results from reactions treated at both temperatures, it is clear that under the specific experimental setup, EcoRI-HF® exhibits similar overall digestive activity at both temperatures. For Set 3 and Set 4: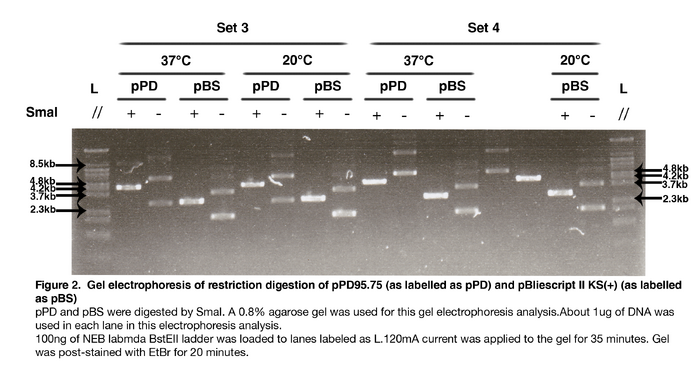 As shown in Figure 2, undigested pPD95.75 sample within set 3 and set 4 yields identical spectrum of bands, which indicates that the undigested form of pPD95.75 used has different conformations; however, a bright band of the size between 3.6k to 4.8k show up in each experimental setups in set 3 and set 4 when SmaI is added, which indicates that linearized pPD95.75 of 4487 base pairs is likely produced, meaning that the pPD95.75 is efficiently digested by the enzyme under both temperature. Similarly, the result of experimental groups of pBS II KS (+) incubated at different temperature also yield pattern of bands different from that of their negative control. A band of size between 2.3k to 3.6k is produced, suggesting that linearized pBS II KS (+) of 2961 base pairs is produced and pBS II KS (+) is digested by SmaI. The two groups of reactions using different DNA serve as positive control for each other and show that SmaI is of proper enzymatic activity. Most digested samples show one single band of the same size, implying the DNA was mostly digested. There is one obscure band produced by pPD95.75 digested with SmaI at 37℃ in set 3, it is probably the undigested pPD95.75. Even though little amount of DNA was undigested, most DNA has been linearized, so SmaI maintains most of its activity at 37℃. It is concluded that the restriction enzyme, SmaI, has similar efficiency at 20℃ or 37℃ with our specific experimental conditions. DiscussionIt is shown that the undigested pPD95.75 yields different banding pattern between set 3 and set 4. The former gives 3 bands while the latter gives 2 bands only. Although sources of DNA of set 3 and 4 are the same, it is very likely to have different kinds of conformation of plasmids if they come from independent preparations. DNA used in set 3 and set 4 are prepared with a slightly different procedures. Such difference might have caused the plasmids to form different conformations. Plasmids of set 3 exhibit 3 major types of conformation while that of set 4 have only 2. Types of conformation may affect the migration of plasmids, thus giving distinct banding pattern. In set 4, the first lane to the right of pBS incubated at 37oC without enzyme is planned to be loaded with pPD95.75 incubated with SmaI at 20oC; the second lane to the right of pBS incubated at 37oC without enzyme is planned to be loaded with pPD95.75 incubated without SmaI at 20oC. However, unexpected result is shown. The first lane yields a spectrum of lane while the second lane yield one band only. Also the spectrum of bands in the first lane is similar with pPD95.75 incubated in 37oC without SmaI. And the band on the second lane has similar size with the band yield in pPD95.75 incubated at 37oC with SmaI. One possible explanation of this strange result is that during loading of DNA samples, the two DNA sample is switched. Another possible explanation is enzymes function differently at 37oC and 20oC. Although the latter possible explanation is unlikely, as SmaI function similarly on pBS at both 37oC and 20oC, we cannot be sure the reason responsible for this result. Thus the result of these two lanes is not used to interpret the function of SmaI. According to the New England Biolab (source of restriction enzyme), the optimum temperature of EcoRI-HF® and SmaI are 37oC and 20oC respectively. Theoretically, EcoRI-HF® working at 37oC should be more efficient than at 20oC. However, our results showed that they are having similar efficiency in 37oC and 20oC. We believe that addition of extra amount of restriction enzyme leads to the deviation between experimental and theoretical results. Instead of adding 1U of restriction enzyme, we had used 5U of restriction enzyme in each set of experiment to ensure the sufficiency. Nevertheless, this change may cover the effect of temperature, thus causing the result become similar in them. ConclusionBoth EcoRI-HF and SmaI can perform their digestive function on plasmids, 5U of EcoRI-HF or SmaI function similarly in both 37oC and 20oC in terms of digestive function on 1ug of DNA. |