IGEM:Harvard/2009/Lab Notebook/Week1
Monday: June 15, 2009
Tuesday: June 16, 2009
Since we are still unable to obtain PhyA, PhyB, and PIF3 cDNAs from any of the sources we have enquired with (although there is a possible lead from the Mathews group), we have decided to pursue extraction of mRNA from Arabidopsis directly. We obtained two small Colombian Arabidopsis plants from the Pierce lab, courtesy of one of their graduate students, and will use the Qiagen Plant RNeasy kit for mRNA extraction. Per their instructions, we should flash freeze the leaves with liquid nitrogen, grind them with a frozen mortor and pestal under liquid nitrogen, and then use the kit for the extraction.
As of the moment we are still waiting to hear back from the Mathews group about a cDNA library or PhyA and PhyC cDNAs; a phone call was placed to them today. Hopefully they should get back to us tomorrow.
-AG
Today we did a Midiprep on the GFP overnight cultures The midiprep procedure we used could not be found on the Qiagen website, so instead Anu googled up a midiprep protocol which has now been uploaded in the Protocols section of the Wiki but here is a summary:
- Spin the large conical tubes that contained the culture down in a centrifuge under 4000 rpm for 10 minutes
- Resuspend the pellet in 4mL of Buffer P1 (chilled, RNAase added - the reason it needs to be chilled is to delay the activity of the enzyme until it is added to our pellet suspension)
- Add 4mL buffer P2, invert 4-6 times and incubate at room temperature for 5 minutes
- Add 4mL chilled Buffer P3, mix and invert 4-6 times. Incubate on ice for 15 minutes
- Equilibrate (synonymous to 'run through') a Qiagen Tip 100 (this can be found in the Midiprep kit box) by applying 4mL of Buffer RBT and let the column empty by gravity flow (ie. wait for the solution to completely drip from the Tip 100 thing into the tube
- Apply the supernatant to the Qiagen tip and allow it to enter the resin by gravity flow
- Wash the Qiagen tip with 2x 10mL Buffer QC, we run two separate 10mL QCs
- Elute DNA with 5mL Buffer QE
- Add isopropanol directly to the solution (not through column) to precipitate DNA. Add 3.5mL (0.7 volume) isopropanol. Mix and centrifuge immediately at 15,000g for 30min at 4°C
- Decant supernatant. The white pellet we see is DNA.
- Wash DNA pellet with 2mL 70% EtOH at room temperature and centrifuge at 15,000 rpm for 10 minutes
- Precipitate with EtOH and let tube dry so we can see the DNA
This midiprep method seemed to have taken a while and the EtOH precipitation did not yield any visible pellets. So additionally we are gonig to do a backup "quick and dirty" plasmid prep from a the GFP agar plate. This procedure is just like a regular miniprep except we did not do the overnight culture and instead scraped off all the colonies from the plate to get off as much bacteria as possible to extract the maximum amount of DNA
Here are our spec results:
- Midiprep 1 sample: ~3.8mg/μL
- Midiprep 2 sample: ~2.2mg/μL
- Miniprep sample: ~15.6mg/μL
We then followed up by doing digestions
GFP Vector Digest (quantities are in μL with a total volume of 40μL)
- 30 DNA
- 4 NEB4
- 4 BSA
- 1 Xba1
- 1 Spe1
Low Promoter Digest (quantities are in μL with a total volume of 65μL)
- 50 NA
- 6.5 NEB4
- 6.5 BSA
- 1 Xba1
- 1 Spe1
These samples were subsequently incubated at 37°C for 1 hour. We now have 3 low promoter samples and 1 GFP sample.
After the one hour incubation the samples were purified using the PCR purification kit. This removes the salts and unwanted residue, etc... and we used the Qiagen Nucleic Acid Clean up protocol
Spec results
- Low promoter insert 1: 7.6mg/μL
- Low promoter insert 2: 14.8mg/μL
- Low promoter insert 3: 6.5mg/μL
- GFP vector: 2.7mg/μL
Wednesday: June 17, 2009
Today Ivan and Gosia started out by pouring 2% gels (they made one gel, cut up half of it and used the other half) and ran the digested GFP and low promoter (3 of the latter) samples from yesterday.
Amy and Neena then carried out minipreps of the overnight GFP culture. We used up all the overnight culture by filling 1.5mL of culture into eppendorf tubes, spinning down the sample, and then repeating this procedure 3x so we have ~5mL of culture per tube. We ended up with 8 tubes of 3x1.5mL of overnight GFP culture.
Spec results of miniprepped DNA
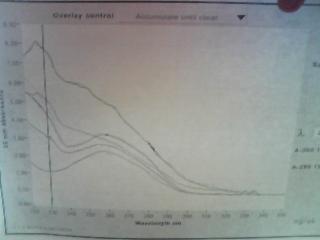
We wanted to pick the best stock, some samples seemed to have relatively high alcohol contamination so we observed the graph of the specs and for overlay control clicked Accumulate Until Clear to get a single graph with accumulated specs. A peak at ~220-230nm corresponded to possible alcohol contamination so based on the concentration and graph results GFP sample 4 seemed to be the best sample.
Trial 1/Trial 2 (quantities in ng/μL)
- GFP 1: 143.4 / 145.5
- GFP 2: 164.9 / 174.15
- GFP 3: 215.9 / 169.71
- GFP 4: 113.9 / 120.92
- GFP 5: 149.8 / 255/ 257
- GFP 6: 107.0 / 153.90
- GFP 7: 115.0 / 117.4
- GFP 8: 164.2 / 141.2
Basically we do not want to see a graph that is completely downward sloping, we do not want to see any peaks in the ~220-230nm range. What is ideal is a single peak in the ~260nm range (towards the center of the graph). These ideally peaked samples correspond to samples GFP 4 and GFP 7.
We then digested our miniprepped DNA (We ran digestions only on GFP 4 and GFP 7 based on the spec results) using the following protocol
- 30μL DNA
- 4μL NEB Buffer
- 4μL BSA
- 2μL Spe1
- 1μL Xba1
=40μL total volume
Now that we are done with the digestions and incubated the digested product at 37°C for 70 minutes. We will now culture components of the yeast two-hybrid that Amrita transformed and plated from last night.
Plate Number / Yeast 2-hybrid component / Antibiotic
- Plate 1: HO1 (the biosynthetic pathway) / Kan
- Plate 2: PhyA (labeled PycA) / Kan
- Plate 3: PhyB / Amp
- Plate 4: Pif3 / Amp
We picked 3 colonies per plate and labeled them 1a, 1b, 1c, etc... with antibiotic and the date
We added the colonies to 5mL of LB and 5μL antibiotic (either Kan or Amp). The colonies from the plate were very small so we used the Fisherbran white needles to pick the colonies. We then incubate at 37°C overnight. We then sealed the plates with parafilm and put them back in the iGEM 2009 4°C fridge.
Now that we have picked the colonies we will do gel purifications of the vector backbone to separate the backbone from the GFP. We ran the samples on the leftover 2% gel that we cut in half earlier today.
We added 4μL of loading dye per sample volume and ran the gel first for 20 minutes (since it is a 2% gel)
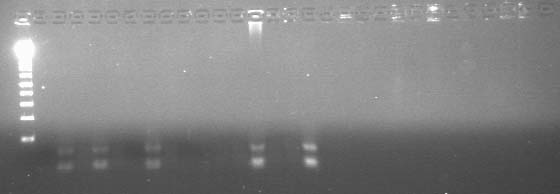
Here is an annotation of the gel
- Well 1: 10μL DNA ladder
- Well 2: Empty
- Well 3: Empty
- Well 4: Empty
- Well 5: GFP
- Well 6: Empty
- Well 7: Empty
- Well 8: GFP 7
We then gel purified the samples using David's filter paper method, our yields were as followed:
- GFP 4: 0.3ng/μL
- GFP 7: 3.7ng/μL
We then followed up by putting the samples in the spinvac (Eppendorf Vacufuge, which can be found on top of the 4°C iGEM 2009 fridge in the main lab were we do our minipreps) to increase the relative concentration of DNA for our ligations. We then let this run for ~10 minutes to reduce our volume to 20mL.
Thursday: June 18, 2009
Plan:
- Minprep on overnightst
- Diagnostic digest and e-gel
- Ligation/transformation
We also nanodropped the speedvaccumed samples of GFP 4 and GFP 7 from yesterday:
GFP 4 had a concentration of around 18 ng/μL
GFP 7 had a concentration of around 59.5ng/μL