IGEM:Harvard/2008/Lab Notebooks/DailyBook/Week2/Chemical and Light
Goals for Week 2
- Transform LacI, TetR, cI + their promoters and GFP. Refer [here].
- Cut Lac promoter -> GFP (P20), and Tet promoter -> GFP (P15), with Xba and PstI and cut SB3K3 (P5) with Xba and PstI and ligate.
- Transform cI promoter. (Refer [here].
- Put constitutive promoters on TetR, LacI, cI.
- Put terminators and RBS on TetR, LacI, and cI.
- Add promoters.
- Make primers that have BioBricks prefix and suffix to flank the origin on Duet Vectors so we can take it out.
- PCR out mtrA and mtrB from Shewie. Also put in with repressors.
- Test anaerobic growth further with birnesite.
Ongoing Experiments
Sequencing and PCRs
- 6/30: Primers to PCR out the CDF origin of replication (with BioBrick ends) were ordered.
- 7/1: PCR reaction set up: 45μL PCR supermix, 1μL S1P13 (435ng/μL), 1μL CDF-F primer (20μM), 1μL CDF-R primer (20μM), 2μL water.
Conditions: 5min @ 94°C → 35x[45s @ 94°C → 45s @ 58°C → 1m45s @72°C] → 5min @ 72°C → ∞ @ 4°C, heated lid
- 7/1: PCR reaction set up: 45μL PCR supermix, 1μL S1P13 (435ng/μL), 1μL CDF-F primer (20μM), 1μL CDF-R primer (20μM), 2μL water.
- 6/30: Most of the minipreps of BioBricks that have been done have been prepared to be sent for sequencing. For longer sequences, both the BBsfx and the BBpfx primers were used. For shorter sequences, only the BBsfx primer was used (this primer is longer and therefore gives more leeway for garbled bases at the beginning of a read.)
- 6/30: 5mL LB-Kan culture of P1 is growing (from glycerol stock)- the plasmid can be digested to yield pSB3K3
The Plan
Goal: High/ Low Constitutive Promoter + RBS + Repressor Coding Region for cI Lambda/ TetR/ LacI + Terminator(s) + pLambda/pTet/pLac Promoter + RBS + GFP + Term in Vector with p15a ori
Scoreboard: Keeping track of what we have and don't have
RBS + Repressor Coding Region
| Plasmid Number | Plasmid Description | Components Digested? | Components Purified? | Ligated? | Transformed? | Miniprepped? | Direct from Registry? |
| P56 | RBS + TetR (P40+P43) | Yes (7/1) | Yes (7/2) | Yes (7/2) | Yes, in E1 (7/2) | ||
| P57 | RBS + cI lambda (P40+P44) | Yes (7/1) | Yes (7/2) | Yes (7/2) | Yes, E1 (7/2) | ||
| RBS + LacI |
High/ Low Constitutive Promoters + RBS + Repressor Coding Region
| Plasmid Number | Plasmid Description | Components Digested? | Components Purified? | Ligated? | Transformed? | Miniprepped? | Direct from Registry? |
| P66 | High + RBS + TetR (P56 +P38) | ||||||
| P67 | Low + RBS + TetR (P56 + P39) | ||||||
| P68 | High + RBS + cI Lambda (P57 + P38) | ||||||
| P69 | Low + RBS + cI Lambda (P57 + P39) | ||||||
| High + RBS + LacI | |||||||
| Low + RBS + LacI |
High/ Low Constitutive Promoters + RBS + Repressor Coding Region + Terminator(s)
| Plasmid Number | Plasmid Description | Components Digested? | Components Purified? | Ligated? | Transformed? | Miniprepped? | Direct from Registry? |
| P70 | High + RBS + TetR + Term (P66 + ) | ||||||
| P71 | Low + RBS + TetR + Term (P67 + P39) | ||||||
| P72 | High + RBS + cI Lambda + Term (P68 + P38) | ||||||
| P73 | Low + RBS + cI Lambda + Term (P69 + P39) | ||||||
| High + RBS + LacI + Term | |||||||
| Low + RBS + LacI + Term |
RBS + GFP + Term
| Plasmid Number | Plasmid Description | Components Digested? | Components Purified? | Ligated? | Transformed? | Miniprepped? | Direct from Registry? |
| P45 | GFP only w/ RBS & terminator | N/A | N/A | N/A | N/A | Yes (7/1) | Yes |
p-lambda/pTet/pLac Promoter + RBS + GFP + Term
| Plasmid Number | Plasmid Description | Components Digested? | Components Purified? | Ligated? | Transformed? | Miniprepped? | Direct from Registry? |
| P15 | pTet + RBS + GFP + Term | N/A | N/A | N/A | N/A | Yes | Yes |
| P20 | pLac + RBS + GFP + Term | N/A | N/A | N/A | N/A | Yes | Yes |
| P74 | pLambda + RBS + GFP + Term (P18 + P45) |
pLambda/pTet/pLac Promoter + RBS + GFP + Term in Vector with p15a ori
| Plasmid Number | Plasmid Description | Components Digested? | Components Purified? | Ligated? | Transformed? | Miniprepped? | Direct from Registry? |
| P58 | pTet + RBS + GFP + Term in p15a ori (P1 + P15) | Yes (7/1) | Yes (7/2) | Yes (7/2) | Yes (7/2) | ||
| P59 | pLac + RBS + GFP + Term in p15a ori (P1 + P20) | Yes (7/1) | Yes (7/2) | Yes (7/2) | Yes (7/2) | ||
| P75 | pLambda + RBS + GFP + Term in p15a ori (P1 + P74) |
High/ Low Constitutive Promoters + RBS + Repressor Coding Region + Terminator(s) + pLambda/pTet/pLac Promoter + RBS + GFP + Term in Vector with p15a ori
| Plasmid Number | Plasmid Description | Components Digested? | Components Purified? | Ligated? | Transformed? | Miniprepped? | Direct from Registry? |
| High + RBS + TetR + Term + pTet + RBS + GFP + Term in p15a ori (P70 + P58) | |||||||
| Low + RBS + TetR + Term + pTet + RBS + GFP + Term in p15a ori (P71 + P58) | |||||||
| High + RBS + cI Lambda + Term + pLambda + RBS + GFP + Term in p15a ori (P72 + P75) | |||||||
| Low + RBS + cI Lambda + Term + pLambda + RBS + GFP + Term in p15a ori (P73 + P75) | |||||||
| High + RBS + LacI + Term + pLac + RBS + GFP + Term in p15a ori (+ P59) | |||||||
| Low + RBS + LacI + Term + pLac + RBS + GFP + Term in p15a ori (+ P59) |
Making a new CDF vector
CDF ori as an insert
| Plasmid Number | Plasmid Description | Components Digested? | Components Purified? | Ligated? | Transformed? | Miniprepped? | Direct from Registry? |
| P76 | modified P13 + P1 (CDF ori + p15a vector) | P1- 7/2, modified P13- 7/3 |
CDF ori + Resistance Cassettes as inserts
| Plasmid Number | Plasmid Description | Components Digested? | Components Purified? | Ligated? | Transformed? | Miniprepped? | Direct from Registry? |
| P79 | P76 + P48 (CDF ori on p15a vector + Cm Resistance) | P48- 7/3 | |||||
| P80 | P76 + P49 (CDF ori on p15a vector + Amp Resistance) | P49- 7/3 |
New Vector w/ CDF ori + Resistance Marker
| Plasmid Number | Plasmid Description | Components Digested? | Components Purified? | Ligated? | Transformed? | Miniprepped? | Direct from Registry? |
| P81 | P79 + P5 (CDF ori + Cm resistance on pSB3K3 vector) | ||||||
| P82 | P80 + P5 (CDF ori + Amp resistance on pSB3K3 vector) |
Transformations from the Registry
Re-Transformation of BioBrick Parts in E1 and E3 (6/27-7/1)
Miniprepped and made glycerol stocks of Biobrick Parts transformed on Friday.
| Name | Registry Name | Description | Origin | Size | Marker | Transformed? | Miniprepped? | Culture in Glycerol Stock? | Verified via sequencing? |
| P5 | pSB3K3 | Low-Medium copy vector (w/ death gene) | p15a | 2750 | Kan | in E3 | Yes | Yes (B- done 6/30. A-done 7/1) | |
| P37 | BBa_I722007 | Constructive expression LacI w/ pTetR promoter | Amp | Failed (liquid cultures didn\'t grow) | |||||
| P38 | BBa_J23114 | High constitutive promoter | 35 | Amp | 2007 in E1 (DH5a), 2008 Failed | Yes. | 2007 (B- done 6/30. A- done 7/1) | ||
| P39 | BBa_J23113 | Low constitutive promoter | 35 | Amp | 2008 (A grew), 2007 (A grew) | Yes. | 2008 (A- done 6/30) 2007 (A-done 6/30) | ||
| P40 | BBa_B0032 | Ribosome Binding Site | 13 | Amp | 2008 (A grew), 2007 (B grew) | Yes. | 2007 (B -done 6/30) 2008 (A-done 6/30) | ||
| P41 | BBa_B0010 | Transcriptional Terminator | Amp | Failed (liquid cultures didn\'t grow) | |||||
| P42 | BBa_C0012 | LacI coding region | Amp | Failed (liquid cultures didn\'t grow) | |||||
| P43 | BBa_C0040 | TetR coding region | 660 | Amp | 2007 (A grew) | Yes | 2007 (A-done 6/30) | ||
| P44 | BBa_C0051 | cI lambda | 750 | Amp | 2007 (A grew) | Yes | 2007 (A-done 6/30) | ||
| P45 | BBa_E0240 | GFP only w/ RBS & terminator | Amp | 2007 (A and B grew) | Yes | 2007 (A and B -done 6/30) | |||
| P46 | BBa_I51020 | Base Vector | Amp | A and B grew. | Yes | A and B -done 6/30 | |||
| P47 | BBa_P1003 | Kan Resistance Cassette | 967 | Kan | 2007 (A and B grew, but spilled) | Yes | 2007 (A and B -done 7/1) | ||
| P48 | BBa_P1004 | Cm Resistance Cassette | 769 | Cm | 2007 (A and B grew, but spilled) | Yes | 2007 (A and B -done 7/1) | ||
| P49 | BBa_P1002 | Amp Resistance Cassette | 943 | Amp | 2008 (A grew), 2007 (A and B grew) | Yes | 2008 (A-done 6/30) 2007 (A and B -done 6/30) | ||
| P50 | BBa_P1005 | Tet Resistance Cassette | 1283 | Tet | 2007 (A and B grew) | Yes | 2007 (A and B- done 7/1) |
Transformation of Inverters and Repressors (7/1-)
| Name | Registry Name | Description | Origin | Size | Marker | Transformed? (7/2) | Miniprepped? | Culture in Glycerol Stock? | Verified via sequencing? |
| P51 | BBa_Q04121 | LacI QPI w/ RBS & promoter | 1370 | Kan | Failed in E1. | ||||
| P52 | BBa_Q04510 | cI QPI w/ RBS & promoter | 987 | Kan | Failed in E1. | ||||
| P53 | BBa_P0440 | PoPS --> TetR | 840 | Amp | Yes, in E1. | ||||
| P54 | BBa_P0412 | PoPS --> LacI | 1308 | Amp | Yes, in E1. | ||||
| P55 | BBa_P0455 | RBS.Lambda.cI. LVA.Terminator | 896 | A/C | Yes, in E1. |
Transformation of Terminators and LacI coding regions (7/2)
| Name | Registry Name | Description | Origin | Size | Marker | Transformed? (7/2) | Miniprepped? | Culture in Glycerol Stock? | Verified via sequencing? |
| P41 | BBa_B0010 | Transcriptional Terminator | Amp | in E1. | |||||
| P61 | BBa_B0011 | Terminator | in E1. | ||||||
| P62 | BBa_B0012 | Terminator | in E1. | ||||||
| P63 | BBa_B0014 | Double terminator (P62 and P61) | in E1. | ||||||
| P64 | BBa_B0015 | Double terminator (P60 and P62) | in E1. | ||||||
| P65 | BBa_B0025 | Double terminator (P62 and P60) | in E1. | ||||||
| P77 | BBa_P0312 | RBS + LacI + terminators | in E1. | ||||||
| P78 | BBa_P0112 | RBS + LacI + terminators | in E1. |
First Round
Putting Lac/Tet + GFP with P15A, and RBS with TetR and cI lambda coding regions.
In this round:
| Vector Name | Vector Description | Insert Name | Insert Description | Projected Resulting Plasmid |
| P1 | P15a ORI | P15 | GFP w/ pTet | P58 |
| P1 | P15a ORI | P20 | GFP with pLac | P59 |
| P40 | Vector with RBS | P43 | TetR coding region | P56 |
| P40 | Vector with RBS | P44 | cI lambda coding region | P57 |
Also cutting P38, P39, and P45 in anticipation of future ligations.
Restriction Enzyme Digestions (7/1)
Vectors (digested with SpeI and PstI)
- RBS (P40, P45)
- cI regulated lambda promoter (P18)
- promoters (P38, P39)
Inserts (digested with XbaI and PstI)
- repressor coding regions (P43, P44)
- GFP under Tet promoter (P15)
- GFP under Lac promoter (P20)
- vector with p15A origin (P1)
Reaction mixture
- 15 μl DNA
- 6.25 μl H2O
- 2.5 μl 10X NEB buffer (buffer 3 for XP, buffer 2 for SP)
- 0.5 μl 50X BSA
- 0.5 μl each RE
Reactions:
| ' | P1 (vector) | P15 (insert) | P20 (insert) | P38 (vector) | P39 (vector) | P40 (vector) | P43 (insert) | P44 (insert) | P45 (vector) |
| DNA | 15 uL | 15 uL | 15 uL | 15 uL | 15 uL | 15 uL | 15 uL | 15 uL | 15 uL |
| 10X Buffer (Volume & #) | 2.5 uL of 3 | 2.5 uL of 3 | 2.5 uL of 3 | 2.5 uL of 2 | 2.5 uL of 2 | 2.5 uL of 2 | 2.5 uL of 3 | 2.5 uL of 3 | 2.5 uL of 2 |
| 50X BSA | 0.5 uL | 0.5 uL | 0.5 uL | 0.5 uL | 0.5 uL | 0.5 uL | 0.5 uL | 0.5 uL | 0.5 uL |
| Restriction Enzyme 1 | 0.5 uL XbaI | 0.5 uL XbaI | 0.5 uL XbaI | 0.5 uL SpeI | 0.5 uL SpeI | 0.5 uL SpeI | 0.5 uL XbaI | 0.5 uL XbaI | 0.5 uL SpeI |
| Restriction Enzyme 2 | 0.5 uL PstI | 0.5 uL PstI | 0.5 uL PstI | 0.5 uL PstI | 0.5 uL PstI | 0.5 uL PstI | 0.5 uL PstI | 0.5 uL PstI | 0.5 uL PstI |
| Water | 6.25 uL | 6.25 uL | 6.25 uL | 6.25 uL | 6.25 uL | 6.25 uL | 6.25 uL | 6.25 uL | 6.25 uL |
| Total Volume | 25.25 uL | 25.25 uL | 25.25 uL | 25.25 uL | 25.25 uL | 25.25 uL | 25.25 uL | 25.25 uL | 25.25 uL |
Incubate overnight at 37 °C
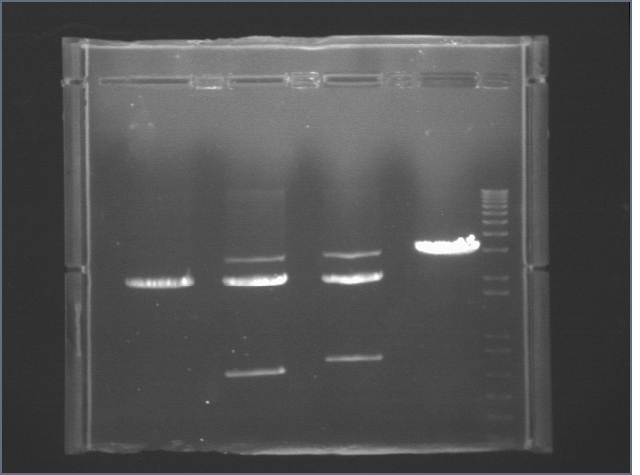
Gel Extraction and Purification of First Round Digestions (7/1)
Gel 1
| Well | Sample |
| 1 | P1 |
| 2 | P15 |
| 3 | P20 |
| 4 | 1 kB Ladder |
Gel 2
| Well | Sample |
| 1 | P40 |
| 2 | P43 |
| 3 | P44 |
| 4 | P45 |
| 5 | 1 kB Ladder |
Ligation and Transformation (7/1)
We are ligated the repressor coding regions (P43 and P44) to the RBS (P40). We are also going to put P15 and 20 into a p15A vector (P1). After sequencing P18 (cI regulated lambda promoter) we will add it to P45 (RBS with GFP).
We transformed DH5α cells with each of these new plasmids (P40+43, P40+44, P1+15, P1+20).
Picking Colonies (7/2)
All four transformations were successful, with many colonies on each plate. Two colonies were picked from each plate.
Minipreps (7/3)
Transformation of P58 and P59, and pET-Duet-1 into Shewanella via Electroporation (7/3)
Second Round
- Adding hi/low constitutive promoters to repressors, OmpR-P promoter to p15a, and p-lambda to (RBS + GFP)
| Vector Name | Vector Description | Insert Name | Insert Description | Projected Resulting Plasmid |
| P1 | P15a ORI | P26 | Mutated promoter activated by OmpR-P with the reporter GFP | P60 |
| P45 | GFP only w/ RBS & terminator | P18 | pLambda (cI regulated) | P74 |
| P56 | RBS + TetR (P40+P43) | P38 | High constitutive promoter | P66 |
| P56 | RBS + TetR (P40+P43) | P39 | Low constitutive promoter | P67 |
| P57 | RBS + cI lambda (P40+P44) | P38 | High constitutive promoter | P68 |
| P57 | RBS + cI lambda (P40+P44) | P39 | Low constitutive promoter | P69 |
| P1 | P15a ORI | modified P13 | pCDF-Duet 1 (origin PCR) | P76 |
(Projected Third Round)
| Vector Name | Vector Description | Insert Name | Insert Description | Projected Resulting Plasmid |
| P76 | CDF ori in p15a vector | P48 | Cm Resistance Cassette | P79 p15a vector w/ CDF ori + Cm resistance |
| P76 | CDF ori in p15a vector | P49 | Amp Resistance Cassette | P80 p15a vector with CDF ori + Amp resistance |
(Projected Fourth Round)
| Vector Name | Vector Description | Insert Name | Insert Description | Projected Resulting Plasmid |
| P5 | Vector w/ NheI sites flanking ori and resistance marker | P79 | CDF ori + Cm resistance | P81 (Vector with CDF ori, Cm resistance, and death gene) |
| P5 | Vector w/ NheI sites flanking ori and resistance marker | P80 | CDF ori + Amp resistance | P82 (Vector with CDF ori, Amp resistance, and death gene) |
Digestions (7/3)
Digestion Reactions:
First Batch
| ' | P26A (insert) | P48B (07) (insert) | P49A (08) (insert) | pCDF (origin PCR) (insert) |
| DNA | 5 uL | 5 uL | 5 uL | 5 uL |
| 10X Buffer (Volume & #) | 2.5 uL of 3 | 2.5 uL of 3 | 2.5 uL of 3 | 2.5 uL of 3 |
| 50X BSA | 0.5 uL | 0.5 uL | 0.5 uL | 0.5 uL |
| Restriction Enzyme 1 | 1 uL XbaI | 1 uL XbaI | 1 uL XbaI | 1 uL XbaI |
| Restriction Enzyme 2 | 1 uL PstI | 1uL PstI | 1 uL PstI | 1 uL PstI |
| Water | 15 uL | 15 uL | 15 uL | 15 uL |
| Total Volume | 25 uL | 25 uL | 25 uL | 25 uL |
Second Batch (We did some of the digestions in the First Batch incorrectly. Only the correct ones are now listed under First Batch.)
| ' | P39A (Vector - Prefix) | P38A (Vector - Prefix) | P57B (Insert - Suffix) | P56B (Insert - Suffix) | P18D (Vector - Prefix) | P45B (Insert - Suffix |
| DNA | 5 uL | 8 uL | 10 uL | 10 uL | 10 uL | 5 uL |
| 10X Buffer (Volume & #) | 2.5 uL of 2 | 2.5 uL of 2 | 2.5 uL of 3 | 2.5 uL of 3 | 2.5 uL of 2 | 2.5 uL of 3 |
| 50X BSA | 0.5 uL | 0.5 uL | 0.5 uL | 0.5 uL | 0.5 uL | 0.5 uL |
| Restriction Enzyme 1 | 1 uL SpeI | 1 uL SpeI | 1 uL XbaI | 1 uL XbaI | 1 uL SpeI | 1 uL XbaI |
| Restriction Enzyme 2 | 1 uL PstI | 1 uL PstI | 1 uL PstI | 1 uL PstI | 1 uL PstI | 1 uL PstI |
| Water | 15 uL | 12 uL | 10 uL | 10 uL | 10 uL | 15 uL |
| Total Volume | 25 uL | 25 uL | 30 uL | 25 uL | 25 uL | 25 uL |
All of these parts were run on a gel and purified
Transformation of Parts into S1
Transformation of Lac/Tet + GFP into S1(7/3)
25 mL culture of S1 was grown for electroporation and transformation.
| Plasmid Name | uL DNA transformed | Plates grew? | Picked colonies? | Miniprepped? | Glycerol Stocks? |
| P58B | 8uL | many small colonies, no GFP | |||
| P59B | 8uL | 5 medium-sized orangish-pinkish colonies, GFP + | |||
| P12 (from Christina) | 5 uL | ||||
| P27 | 2uL | 9 small/medium orangish colonies, also many very small white-ish colonies | |||
| P28 | 2uL | many tiny colonies, no GFP | |||
| P29 | 5uL | many tiny colonies, no Venus fluor | |||
| P30 | 5uL | 2 small pinkish colonies, YFP+ | |||
| P31 | 8uL | ||||
| P32 | 5uL | many (>50) medium orangish-pinkish colonies, no YFP |