IGEM:Harvard/2006/DNA nanostructures/Notebook/2006-8-9
From OpenWetWare
Jump to navigationJump to search
Folding reaction
Fold 200 μL each of 3.2.Fo, .Go, .Ho, .Io
Each reaction:
- 45 μL p7308 scaffold (44 nM)
- 80 μL oligo stock (250 nM)
- 20 μL 10x folding buffer (500 mM HEPES pH 7.5, 500 mM NaCl, 100 mM MgCl2)
- 55 μL dH2O
Folding conditions:
- high-volume protocol: start at 80[[:Category:{{{1}}}|{{{1}}}]], decrement 0.5[[:Category:{{{1}}}|{{{1}}}]] every 1 min. for 120 cycles.
Gel analysis
- see below
More Microcon trials
Another protocol, inspired by Millipore's Concentrating and Desalting DNA or RNA with Microcon or Centricon Centrifugal Filters:
- 20 μL folded nanostructures (4.0) into a YM-50 Microcon tube
- 480 μL water added
- centrifuged at 14,000 g for 6 min.
- flow-through discarded
- centrifuged upside-down at 1,300 g for 3 min. to collect retentate
- 53 μL collected
Gel analysis
- ran half of retentate, along with half of starting nanostructure volume for comparison
- also ran 5 μL of each nanostructure folded today to see if they folded properly
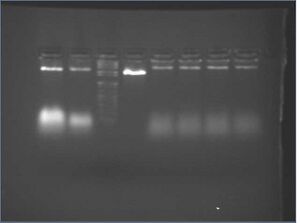
| Lane | Contents |
| 1 | 10 μL nanostructures |
| 2 | 26.5 μL retentate |
| 3 | 1 kb+ ladder |
| 4 | p7308 |
| 5 | 5 μL 3.2.Fo |
| 6 | 5 μL 3.2.Go |
| 7 | 5 μL 3.2.Ho |
| 8 | 5 μL 3.2.Io |
- results/discussion
- Microcon yield is reasonably good, but lots of oligos remain
- in the future: go back to repeating wash four times for 6 min each time (the maximum number of washes, according to Microcon)
- folding reactions appear to have folded properly
Microcon purification of yesterday's folding reactions
- speedvac'd c3.2F, G, H, I, Eb, Fb flowthrough for 50 min. (vol. def much higher than 40 μL)
- ran 2% agarose gel + MgCl2
- 3 flowthroughs is probably not enough - notice pretty clear oligo smears: should probably do 5/6
- don't know if oligo-ligand and latches were added to rxns before microcon: repeat folding of c3.2F, G, H, I and retest
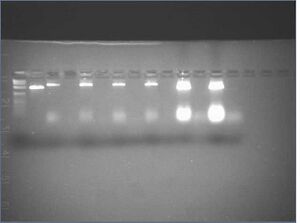
0.5x TBE, 11 mM MgCl2
| Lane | Contents | Loading Buffer |
| 0 | 1kb ladder (10 μL) | - |
| 1 | naked p7308 (7.5 μL) | 5 μL |
| 2 | c3.2F (7.5 μL) | 5 μL |
| 3 | c3.2F flowthrough (35 μL) | 5 μL |
| 4 | c3.2G (7.5 μL) | 5 μL |
| 5 | c3.2G flowthrough (35 μL) | 5 μL |
| 6 | c3.2H (7.5 μL) | 5 μL |
| 7 | c3.2H flowthrough (35 μL) | 5 μL |
| 8 | c3.2I (7.5 μL) | 5 μL |
| 9 | c3.2I flowthrough (35 μL) | 5 μL |
| 10 | c5.0Eb (7.5 μL) | 5 μL |
| 11 | c5.0Eb flowthrough (35 μL) | 5 μL |
| 12 | c5.0Fb (7.5 μL) | 5 μL |
| 13 | c5.0Fb flowthrough (35 μL) | 5 μL |