IGEM:Harvard/2006/Adaptamers/Notebook/2006-8-16
- Yesterday's gel stained with SYBR gold
Following up on yesterday's gel, we restained the gel with SYBR gold. This only caused a more diffuse signal; meanwhile T0 remained unstained.
| Lane | Condition |
| 1 | 10bp ladder |
| 2 | Thrombin beads + T0 wash |
| 3 | Thrombin beads + T0 elution |
| 4 | Thrombin beads + S0 wash |
| 5 | Thrombin beads + S0 elution |
| 6 | Blank beads + T0 wash |
| 7 | Blank beads + T0 elution |
- Yesterday's gel stained with SYBR gold one more time
It may not be possible to stain with SYBR gold following EtBr staining; therefore we concentrated what remained of yesterday's samples using vacuum filtration until there was approximately 10 uL left in the elutions; these samples were run on a 4-20% PA gel once again for 30 minutes, resulting in the gel below:
| Lane | Condition |
| 1 | 10bp ladder |
| 2 | Thrombin beads + T0 wash |
| 3 | Thrombin beads + T0 elution |
| 4 | Thrombin beads + S0 wash |
| 5 | Thrombin beads + S0 elution |
| 6 | Blank beads + T0 wash |
| 7 | Blank beads + T0 elution |
This time we were able to see binding of T0. T0 preferentially binds the thrombin beads and does so at a greater rate than S0. Unfortunately, these results were only achieved after vacuum filtration concentration and high DNA concentrations (although on second thought, these high DNA concentrations were not really necessary). Coupling thrombin to the beads may still be a good option since it is relatively easy to bind large amounts of thrombin to beads, and the efficiency of the procedure will likely improve if we follow protocols made specifically for coupling proteins, which involves a few more reagents.
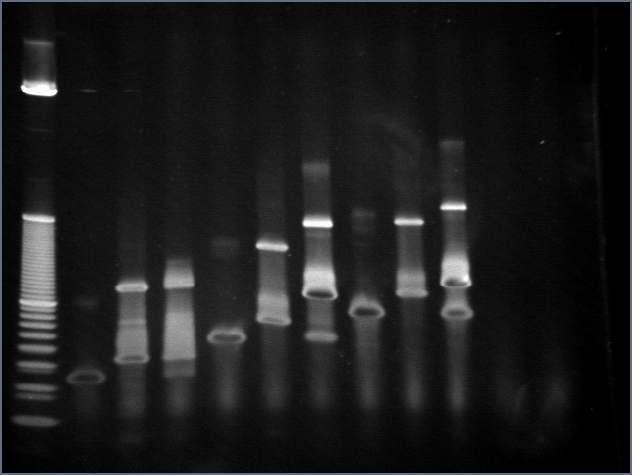
- Basepairing of new adaptamers
From here on, all adaptamers and linkaptamers refer to v.2 unless otherwise stated.
We wanted to make sure that the v.2 linkaptamers came together to form adaptamers. Therefore, we incubated linkaptamers together and ran a 4-20% PA gel in TBE of the samples. Gels were stained in SYBR gold for 30 minutes. Here are the results:
| Lane | Condition |
| 1 | 10bp ladder |
| 2 | 20 pmol T20 |
| 3 | 20 pmol S20 |
| 4 | 20 pmol A20 |
| 5 | 20 pmol T35 |
| 6 | 20 pmol S35 |
| 7 | 20 pmol A35 |
| 8 | 20 pmol T50 |
| 9 | 20 pmol S50 |
| 10 | 20 pmol A50 |
Prior to being run on gels, samples were denatured at 95 degrees and allowed to slowly cool for 15 minutes. This had the potential of some problems since denaturation was carried out in a PCR machine and although the machine was set to avoid evaporation, the small volume (1uL) of sample could have led to loss of samples. Hence we were pleased to see that the signal was still quite strong (in part thanks to SYBR gold staining). The adaptamers appear to be pairing quite well. The S- linkaptamers appear to be the limiting reagent by quite a bit, as marked by the total disappearance of the original S bands and retention of strong T-linkaptamer bands in the lanes with adaptamers. In the future, it may be worth trying different ratios of T and S in order to get the purest adaptamer population possible.
A35 and A50 are forming well, while it is somewhat unclear for A20. While a shift occurs between linkaptamer and adaptamers, it isn't as dramatic as the A35 or A50 shifts, and furthermore, bands of both original species seem to appear (it may just be that there is not much more of one linkaptamer than the other as may be the case for A35 and A50). It is difficult to compare this picture with the same experiment for the v.1 adaptamers since they were run on different gels; the one here provides much higher resolution of the oligos. While I am confident that the A20 adaptamers are forming correctly, we may use A35 adaptamers in initial experiments just to be safe.
- Streptavidin magnetic beads
We'll test the ability of our magnetic beads to bind streptavidin aptamer. The magnetic beads are able to bind ~976 ng biotin/ mL vs. 15-30 ug biotin/mL for the agarose beads. Thus we should probably use greater quantities of magnetic beads to actually see aptamer binding. At the same time, our supplies of magnetic beads are rather limited, so we'll attempt to conserve some. It will likely be possible to conserve by concentrating beads into smaller volumes.
Setup:
Magnetic beads were first washed in Bittker buffer 3X.
50, 100, or 200 uL (.4 % solid) magnetic beads concentrated to 10 uL were mixed with 40 pmol streptavidin aptamer (total of 12 uL) or 40 pmol thrombin aptamer; additionally 8 uL 10% solution was diluted to 10 uL and mixed with streptavidin aptamer as another control. Samples were then shaken for 30 minutes.
Following initial incubation, samples were washed 3X in 1 mL Bittker buffer; initial supernatant was used as wash sample in gels.
Samples were then eluted by incubating with 13 uL 1 mg/mL streptavidin.
Washes and elutions were then run on a 4-20% PA gel at 200 V for 30 minutes in TBE buffer (Novex pre-cast gel).
| Lane | Condition |
| 1 | 10bp ladder |
| 2 | 50 uL beads sample wash |
| 3 | 50 uL beads sample elution |
| 4 | 100 uL beads sample wash |
| 5 | 100 uL beads sample elution |
| 6 | 200 uL beads sample wash |
| 7 | 200 uL beads sample elution |
| 8 | 200 uL beads + T35 sample wash |
| 9 | 200 uL beads +T35 sample elution |
| 10 | 200 uL blank beads sample wash |
| 11 | 200 uL blank beads sample elution |
The magnetic beads worked unexpectedly well. This was likely due to the small volumes and the greater ratio of streptavidin used to elute the aptamers. Only a very small amount of beads and aptamer is really necessary to detect binding, which will be useful for future experiments.