IGEM:Harvard/2006/Adaptamers/Notebook/2006-8-11
8/11
Magnetic beads came today.
- Bead assay
We will try to detect the linkage of streptavidin and thrombin beads. The linkage should be visible since the streptavidin beads are a murky brown and the agarose thrombin beads are translucent white. The problem is concentrations of proteins: while the agarose beads have a binding capacity of 15-30 ug biotin/mL [| (link)], the magnetic beads have a capacity of 1 ng/mL, a seemingly bizarre difference. As a secondary assay, we'll attempt to detect whatever is linked to the streptavidin beads by cleaving with Proteinase K. This should hopefully cleave thrombin and be visible on the gel. Unfortunately this will still have the problem from the 8/9 assay of interference with cleaved streptavidin bands.
400 uL magnetic strepavidin beads were concentrated to 4X in Bittker Buffer and 400 pmol A50 or S0 were added to a total volume of 140 uL. Beads and DNA were incubated for 30 minutes. 150 uL thrombin beads (washed and resuspended in Bittker Buffer) were added and solution was incubated for another hour. Magnet was then applied as shown below.
Results:
In both conditions, thrombin beads came intertwined with streptavidin beads, making their separation impossible. A50 seemed to take longer to float to the magnet, which hopefully is related to it having bulkier particles, but certainly there is nothing conclusive.
- 8/9 experiment one more time..
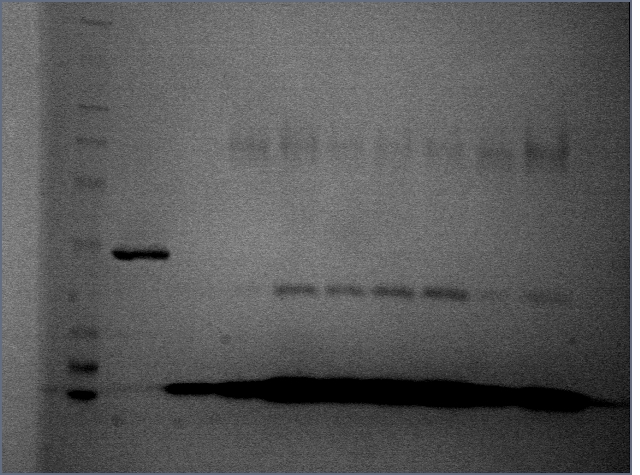
The band where thrombin should be in the Silver staining experiment is pretty unclear thanks to streptavidin staining. I decided to try denaturing the proteins in hopes of seeing more distinct bands.
12% SDS PAGE gel:
Many of the lanes have bands that are unaccounted for by both streptavidin and thrombin. Since the streptavidin bands from previous native gels are consistent with the bands seen in the 8/9 native gels, these are likely streptavidin. Perhaps some of the sample was lost while denaturing it.