IGEM:Harvard/2006/Adaptamers/Notebook/2006-7-21
7/21
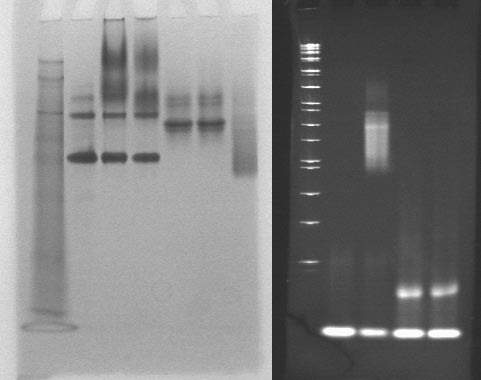
Following continued negative results obtained by the nanostructure group, we repeated most of 7/18's gel at extremely high concentrations with more attention to the positive control of streptavidin and biotin binding. The rationale was that if binding did not occur at these concentrations, then there was something quite wrong with either our materials or procedure. Aiding our confidence in this experiment, more highly-purified samples of the longer aptamers arrived this week (in particular, S5). Incubations were done in total volumes of 9uL.
1: SeeBlue Plus 2 ladder
2: BSA
3: thrombin (100 pmol)
4: thrombin (100 pmol) + T5 (200 pmol)
5: streptavidin (94.5 pmol)
6: streptavidin (94.5 pmol) + S5 (200 pmol)
7: streptavidin (94.5 pmol) + biotinylated oligos (200 pmol)
8: 1kb dna ladder
9: biotinylated oligos (200 pmol)
10: biotinylated oligos (200 pmol) + streptavidin (94.5 pmol)
11: S5 (200 pmol)
12: S5 (200 pmol) + streptavidin (94.5 pmol)
We finally saw clear bands in all conditions. Unfortunately, BSA was responsible for the primary bands of lanes with thrombin. The smear for thrombin may be due to thrombin glycosylation. Because the fuzzy signal, it is difficult to tell if there is a shift when T5 is added to thrombin. Streptavidin does not appear to be binding S5. The fuzzy signal may be due to excessive protein concentrations; we shall try everything again at 40 pmol DNA + 20 pmol protein.
This time around, we could see new bands for S5. So what's the band consistent across the DNA lanes?
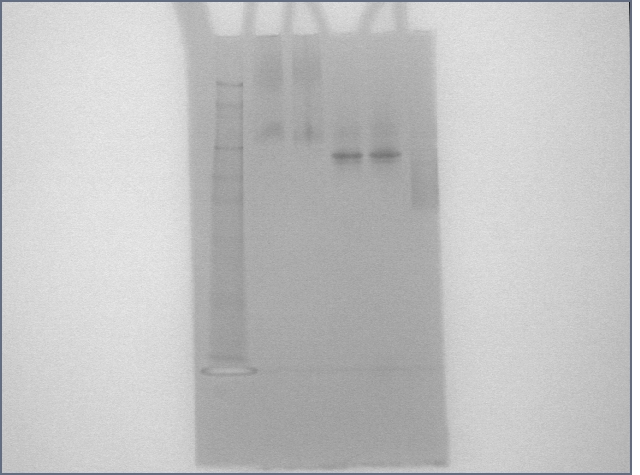
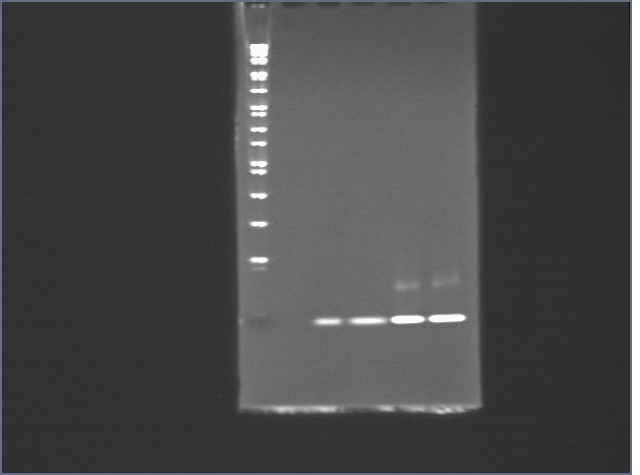
Lower concentration gel
We reconstituted Bovine thrombin in PBS this time around. Lanes are as follows:
Protein
1: SeeBlue Plus 2 ladder
2: thrombin (20 pmol)
3: thrombin (20 pmol) + T5 (40 pmol)
4: streptavidin (20 pmol)
5: streptavidin (20 pmol) + S5 (40 pmol)
6: streptavidin (20 pmol) + biotinylated oligos (40 pmol)
7: 1kb dna ladder
8: nothing + dye
9: T5 (40 pmol)
10: T5 (40 pmol) + thrombin (20 pmol)
11: S5 (40 pmol)
12: S5 (40 pmol) + streptavidin (20 pmol)
Conclusions:
1) Weird DNA band is not due to dye.
2) streptavidin-thrombin binding not detected.
3) Thrombin binding not detected.
Next: Primary: 1) Denature DNA before incubation.
2) Use human thrombin instead of bovine thrombin. 85% homology isn't enough.
Contingency plan: 1) Try a more sensitive binding assay.
2) Try S0, T0 instead of S5, T5
3) Try human thrombin.
4) Run a gel a shorter time to see if T5, biotinylated oligos are resolved separately from mystery band.