Hydrophobic Valves - Thomas Falcucci
Motivation
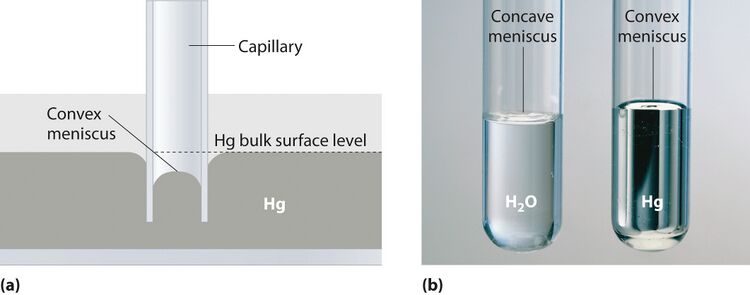
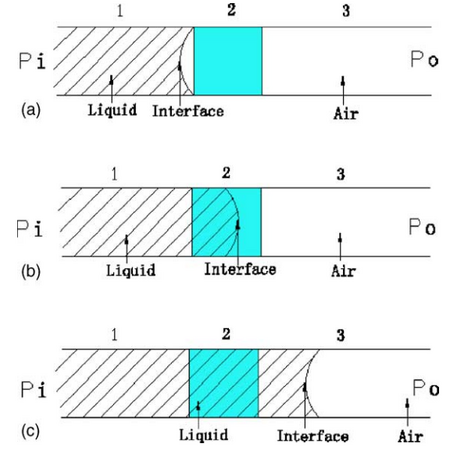
In the world of microfluidics, many of the forces that are considered insignificant in the macroscale play a determining factor in the microscale systems. On the microscale, the fluid-surface interactions--the hydrophilic and hydrophobic interaction between the channel surface and the liquid solution--play a dominant role in the transportation of the liquid throughout the device.1 Fig. 1 shows an example of this surface-fluid interaction: the meniscus that forms on the walls of a test tube. Based off this knowledge, hydrophobic valves employ the same science to precisely control the flow of a liquid throughout a microchannel. While many valves require external or extra components to be added to the channel for the valve to function, hydrophobic valves require few extra components, all of which are simple to integrate. The hydrophobic valve is a passive valve that requires no actuation or movement to function.2 In conjunction with the lack of actuation and movement, the few additional materials make the hydrophobic valve a reliable, cheap, and adaptable valving method.
Current Methods
All the current methods utilize the opposing surface chemistry of hydrophilic glass, Poly(Dimethyl siloxane)PDMS, or Silica and hydrophobic Self-Assembled monolayers (SAMs) or Poly(methyl methacrylate) or PMMA, or polyelectrolyte to create the valves. While the preparation methods differ for the varying surfaces and polymers, the general idea of the deposition of a thin hydrophobic film on a microfluidic device remains constant. As research continues on this valving mechanism, more methods are developed to overcome specific hurdles. The current techniques, while all different in appearance and production, all exploit the same principles of surface energy. They all exploit the pressure phenomena that occurs at the surface-liquid interface, which is expressed by Eqn.1.3 The larger the meniscus angle (the more hydrophobic the surface) the larger the pressure difference is across the meniscus. Thus, the fluid will stop at the hydrophobic surface and require an external force electrical or pressure) to overcome the pressure difference and force the liquid through the valve.
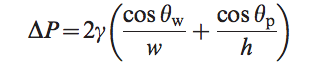
Where γ is the surface energy, w is the width of the channel, h is the height of the channel, θw is the wetting angle on the walls, and θp is the wetting angle on the top and bottom surfaces. This is the basic idea of the hydrophobic valve: use the surface-liquid interaction to limit and control fluid flow through a channel. Each technique exploits a different property to control this pressure difference.
Geometry
The geometry of a hydrophobic valve plays a significant role in controlling the fluid movements through the channel. The pressure difference above is a function of not only the wetting angle (θw in Eqn.1 or θc in Fig. 2) but also of the channel geometry w and h, width and height respectively. In geometric hydrophobic valves, the cross-sectional area determines the valve capabilities. If the material properties of the channel and therefore, the contact angle θc, are kept constant, the difference in pressure between the interface is positive when θc > 90◦ and the liquid flows from channel 1 with larger cross-section area to channel 2 with smaller cross-section area. This means that additional pressure should be applied to force liquid from channel 1 to channel 2. Therefore, the narrowing of channel with hydrophobic surface (θc > 90◦) can function as a stop valve. From Eq. (5), the pressure difference, capillary restriction, have no relationship with the distance the liquid flows through and the flow velocity.

Materials
These types of valves are usually limited to the more sturdy device materials like glass, silicon wafers, or molded plastics due to the forces involved in channel dimension changes. The materials are also generally those that are compatible with wet etching and photoresists. Hydrophobic materials like SAMs or PMMA.
Challenges
The challenges below are associated with materials like glass or silicon. Molded plastic resolves most of these problems, but fails to match the resolution of the other materials.
- Multistep and multicomponent assembly
- Methods confined to a relatively narrow range of materials
- More expensive materials
- The device making process requires more time and chemicals due to the materials used
- Limited to specific fluid-surface pairings beyond water
Patterning
Unlike the geometric valves, which employ channels with a fully coated hydrophobic surface, systematic patterning of the hydrophobic patches can be used to valve. Often only one wall of the channel is coated not only saving costs for materials but also making assembly much easier. Patterning also allows for customizable structures for the valves that are not limited by the valve geometry.
Printing

A simple concept, printing directly onto the device surface, like printing a document on paper, provides extreme flexibility and customizability for a low cost. Printing onto surfaces has the same parameters as printing on paper The most useful and unique ability is the ability to control the gray-scale settings which control the amount of pressure required to overcome valve.3 Ink-jet printers, the same printers used in homes, are used to precisely control the toner deposition as shown in Fig.3. This method is easy for mass production due to its low cost and ease of use. Combined with the ability to tune the pressure difference required to overcome the surface forces based on the gray-scale makes printed valves very unique and useful.
Materials
Ink-jet printer. Many toners can be used, but some are more effective than others (the best results were achieved by an HP Laserjet printer).3 An channel surface can be used so long as the toner can bond to the surface.
Challenges
- There are variations in the patterning due to the printing methods
- Thus far toner has only been tested as hydrophobic material
- Limited to specific fluid-surface pairings
- Results vary with different toners
- Resolution limited to the printer resolution, so fine detail is more difficult to achieve than with other methods.
Micropatterning
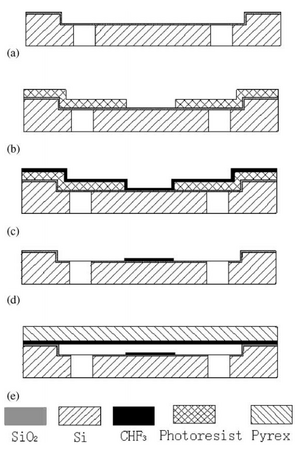
Photolithography is used to create hydrophobic patches on the channel surface on the top of any surface as seen in Fig.4. While this is more versatile because photolithography can be applied to any of the bases, the processing is much more costly and time consuming. This of use of photolithography also expands the material used for the hydrophobic surfaces beyond toner ink, thus widening the range for the working fluid of the microfluidic device.1 Microcontact printing can be employed to overcome the time requirements and to ensure repeatability, but this method is limited to the multiple layering and the alignment of the pieces.
Materials
Challenges
- Multistep and multicomponent assembly
- More expensive processing materials
- The device making process requires more time and chemicals due to the materials used
- Aligning the two sides of the device are difficult
Tunable Hydrophobic Valves
These valves are created through self assembling hydrophobic molecules on a treated surface. These molecules act, such that, when a voltage is applied the bonds connecting them to the surface is broken, destroy the hydrophobic aspect and allowing the fluid to proceed through. This is much more accurate than the tunable gray-scale printing method, but once the bond between the hydrophobic molecules and the surfaces is broken, they can not be reset. This means the valve is for one time use only. This limits the uses to things such as systematic reactant addition. 5
For more information see Electrowetting Valves - Davis Miller
Challenges
Once the monolayer is destroyed there is no current method to reconstruct it; the valve is for one time use. Requires the addition of an external voltage source and thus the design must account for anodes. There has been a study that uses carbon nanotubes and certain alloys as SAMs to create reversible electrowetting valves but they have been limited to specific solvents and their application has not been tested in working lab-on-a-chip devices.6
References
1. Feng, Yanying, Zhaoying Zhou, Xiongying Ye, and Jijun Xiong. "Passive valves based on hydrophobic microfluidics." Sensors and Actuators A: Physical 108, no. 1 (2003): 138-143. http://dx.doi.org/0.1016/S0924-4247(03)00363-7
2. Oh, Kwang W., and Chong H. Ahn. "A review of microvalves." Journal of micromechanics and microengineering 16, no. 5 (2006): R13. http://dx.doi.org/10.1088/0960-1317/16/5/R01
3. Ouyang, Yiwen, Shibo Wang, Jingyi Li, Paul S. Riehl, Matthew Begley, and James P. Landers. "Rapid patterning of ‘tunable’hydrophobic valves on disposable microchips by laser printer lithography." Lab on a Chip 13, no. 9 (2013): 1762-1771. http://dx.doi.org/10.1039/c3lc41275j
4. Tsougeni, Katerina, Dimitris Papageorgiou, Angeliki Tserepi, and Evangelos Gogolides. "“Smart” polymeric microfluidics fabricated by plasma processing: controlled wetting, capillary filling and hydrophobic valving." Lab on a Chip 10, no. 4 (2010): 462-469. http://dx.doi.org/10.1039/b916566e
5. Biswas, Gokul Chandra, Takahiro Watanabe, Edwin T. Carlen, Masatoshi Yokokawa, and Hiroaki Suzuki. "Switchable hydrophobic valve for controlled microfluidic processing." ChemPhysChem (2016). http://dx.doi.org/10.1002/cphc.201501015
6. Manjeet S. Dhindsa,Neil R. Smith, and, Jason Heikenfeld*, Philip D. Rack, Jason D. Fowlkes,Mitchel J. Doktycz,Anatoli V. Melechko, and, and Michael L. Simpson Langmuir 2006 22 (21), "Reversible Electrowetting of Vertically Aligned Superhydrophobic Carbon Nanofibers " (2006) 9030-9034 http://dx.doi.org/10.1021/la061139b