Herpes Simplex Virus (HSV I) as Cancer Targeting Agent
Created by Eva Klinman and Jackie Goldstein
Brief Project Overview
We will attempt to target the Herpes virus against specific cancer cells, using a modified form of Herpes which will do one of two things:
- A modified form of Herpes, G207, which selectively targets and replicates in cancer cells, already exists.
- We will obtain G207 from another lab, and seek to further direct its targeting, such that it attacks a specific cancer, rather than non-human cells in general.
- Our modification will focus on the heparan sulfate (HS) binding mediated by glycoproteins B and C (gB and gC).
- By selecting such that gB or gC (we will test both in parallel) optimally bind to a surface protein expressed by the cancer of choice, in this case cervical cancer, we will create a targeted cancer-killing virus.
- Importantly, G207 has already been shown to have very very little toxic effects on normal cells and creatures, even in animals selectively sensitive to HSV I.
Background Info
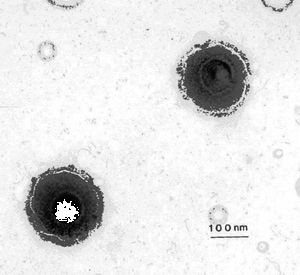
The Herpes virus naturally attacks and kills quickly dividing cells, such as the ones present around the mouth, which results in cold sores. Currently, Herpes is being used to target fast growing cancers in areas of little cell growth, such as the brain. However, researchers and doctors have no guarantees of success with this treatment method, and no assurance that the virus will stop after it has destroyed the cancer, and instead turn on the normal human body cells.
Function of Herpes in nature
- Find natural function of Herpes (cold sores, what they do)
- Reproductive cycle
- How Herpes inserts itself into living cells (host)
- Heldwein et. al "Entry of herpesviruses into mammalian cells"
- What glycoproteins binds to
- gD:[B5, heparan sulfate, HVEM, nectin-1]
- gB:[heparan sulfate]
- gC:[heparan sulfate]
- Order of binding for entry
- What glycoproteins binds to
- Heldwein et. al "Entry of herpesviruses into mammalian cells"
- gC binds to heparan sulfate (not essential)
- Promoter/activator sequence in Herpes
Signaling of Cancer
- Cervical cancer is notoriously hard to target and find, because as opposed to many cancer types, cervical cancer cells display less, rather than more, cell surface proteins. Many screening techniques rely on comparisons between healthy cells and possibly cancerous cells, and determine the cancerous nature of the test cells based on significant LOWERING of cell surface proteins.
- However, a few surface proteins show elevated levels of expression in cervical cancer, including pp17b.
- Cervical cancer is a slow growing by highly invasive form of cancer, that is very hard to detect unless the woman gets frequent pap smears. Its stages of growth are divided into 5 parts, numbered 0 through IV.
- Past the later part of stage I or early stage II, the patient has a much higher risk of death, as the cancer has spread to other regions of the body. Even though it is still localized in the pelvic area, it is much harder to track down.
Papers
- Ebright MI et al. Replication-competent herpes virus NV1020 as direct treatment of pleural cancer in a rat model. J Thorac Cardiovasc Surg 2002;124:123-129.
- Showed that a specific herpes virus is effective at killing a human lung cancer line both in vitro and in vivo (in mice)
- Stojdl DR et al. VSV strains with defects in their ability to shutdown innate immunity are potent systemic anti-cancer agents. Cancer Cell. 2003 Oct;4(4):263-75.
- Demonstrated how an oncolytic virus is capable of killing human ovarian cancer (in vitro) metastatic colon cancer (in vivo- mice)
- Thorne SH et al. Rational strain selection and engineering creates a broad-spectrum, systemically effective oncolytic poxvirus, JX-963. J. Clin. Invest. 117(11): 3350-3358 (2007)
- An oncolytic virus is capable of both targeting and killing cancer in vivo (in mice and rabbits)
- Varghese S, Rabkin SD. Oncolytic herpes simplex virus vectors for cancer virotherapy. Cancer Gene Therapy. 2002 Dec; 9(12):967-978
- Summarizes the types and effects of mutations in HSV I to date, including G207.
- Wolfe D et all. Engineering Herpes Simplex Virus Vectors for CNS Applications. Experimental Neurology 159, 34–46 (1999)
- Overview of funtion and mutations of Herpes and what they do. Includes possible further steps, and failed past experiments.
- Than NG et al. Cloning and sequence analysis of cDNAs encoding human placental tissue protein 17 (PP17) variants. Eur. J. Biochem. 1998. 258:752-757
- DNA and amino acid sequence of pp17 (a and b versions)
Statement of Research Problem
Anti-cancer drugs are not specific enough, and cause long-term harm in humans who undergo cancer treatment.
The Herpes virus provides a way of selectively targeting cancer cells in vivo. Herpes has already been used to kill fast-growing cancers in areas of minimal normal cell growth, such as in the brain.
We plan to alter the Herpes recognition and duplication cycle such that it bind to specific cell surface proteins presented by cancer cells. This way, the Herpes virus should invade and kill the cancer cells, but leave the surrounding cells untouched, even after the cancer is destroyed.
Research Goals
- Identify over-expressed cell surface protein in cervical cancer: pp17b
- pp17 is expressed five fold more in cancerous cells than in normal cells, thus will act as a useful target.
- Modify the Herpes virus (HSV I) such that it can target the selected protein: mutate gB and gC (separately)
- gB and gC bind to HS naturally. Creating a fusion gB or gC such that it binds to pp17b will increase the likelihood of invading the cancerous cell.
- Use a variant of HSV I that will not hurt normal human body cells: G207
- G207 has already been proven to selectively kill cancer cells. It is not toxic even when over injected into herpes-sensitive creatures such as young BALB/c mice and New World owl monkeys.
Test Conditions
Test the Herpes in three conditions:
- Will it kill the chosen cancer if injected into the tumor
- Will it selectively kill the cancer if placed in the tumor growing on normal tissue
- Will it deactivate when the cancer is gone/if no cancer is present
Ultimately, if our research goal is met, we plan to make Herpes which target many types of cancer, and is financially affordable.
Project Details and Methods
- Using the sequence for pp17b (known), generate complementary DNA that will selectively bind to an extracellular matrix section of the pp17b protein.
- One of two methods to get pp17b sequence:
- Acquire pp17b and selectively amplify only the region we want using directed primers
- Create/order DNA of the correct sequence and PCR amplify it
- One of two methods to get pp17b sequence:
- Selectively replace the coding region of either gB or gC with the pp17b sequence, to create a fusion protein gB|pp17b or gC|pp17b.
- Possibly use restriction enzyme insertion methods.
- Insert the fusion protein into functional G207 HSV I, such that it takes the place of the wildtype gB or gC proteins.
DNA/Protein sequences
- gC sequence (UL44)
- gB sequence (UL27)
- other glycoprotein sequences in HSV envelope
- pp17b protein sequence
Creating gC|pp17b Plasmids
- We're going to follow the same experimental setup as listed here:Laquerre et. al. "Herpes Simplex Virus Type 1 Engineered for Targeted Binding to Erythropoietin Receptor Bearing Cells." Journal of Virology, 1998
- They created plasmids containing the gC|EPO fusion gene (note - only pgCEPO2 worked so we'll use the same locations that previously worked for changing gC and gB to bind to pp17b)
- obtained plasmid containing gC gene (UL44) from KOS (we'll use G207)
- used restriction digests of gC gene (BglII and KpnI) to delete signal peptide and 82 N-terminal residues
- replaced deleted sequence with signal peptide of EPO (from an EPO plasmid)by ligating the backbone and fragment together
from Laquerre et. al, Journal of Virology, 1998:
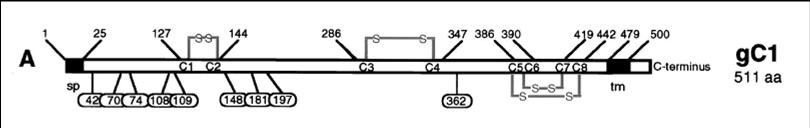
Wild Type gC: TM - transmembrane region, BD - heparin sulfate binding domain (136-152), SP - signalling peptide
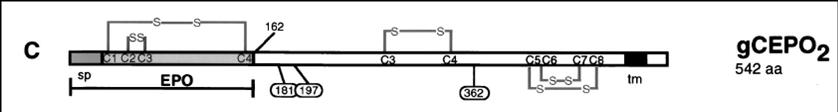
gC|EPO fusion: TM - transmembrane region, BD - heparin sulfate binding domain (136-152), SP - signalling peptide
Transfecting gC|pp17b into G207 viruses
- Standard marker transfer method
- transfect viral DNA with linearized plasmid DNA
- the mutated part of gC needs to be flanked by sequences homologous to viral DNA in order for homologous recombination to occur
- only 0.1-5% recombination frequencies
- would be more difficult to select for viruses that actually have the fusion protein inserted
- transfect viral DNA with linearized plasmid DNA
- Method developed by [Krisky et. al]
- 19-65% efficient at obtaining mutated viruses
- transfect viral DNA with a lacZ reporter construct (the lacZ reporter construct has homology to the gC locus so homologous recombination occurs and in the presence of X-gal, the viral plaques turn blue)
- select for blue plaques
- digest blue plaques with PacI (restriction site only present in lacZ cassette) to release lacZ cassette
- transfect gC|pp17b fusion gene into viruses - homologous recombination will occur to add the fusion gene into the viral genome
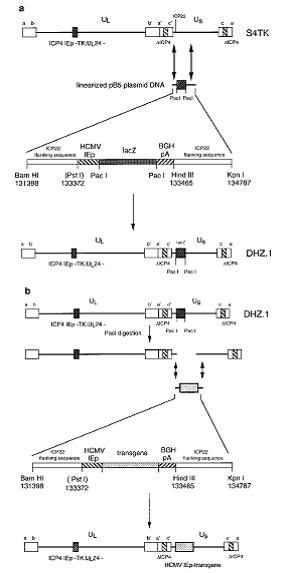
Assays to confirm gC|pp17b inserted into G207 virus
- Diagnostic gels
- Southern Blot
- DNA sequencing
Assays to quantify gC|pp17b G207 specificity to cervical cancer cells
Predicted Outcomes
There are two possible outcomes:
- The modified virus works
- The modified virus fails
Herpes effectively targets cancer cells
- The cancer cells will die, having been selectively targeted by the modified Herpes virus
- Remaining Herpes would remain in the bloodstream and body for a short period of time, and then die, or lie quiet (not infect normal cells)
- If this occurs, we would continue research:
- Publish.
- Apply for the ability to test the Herpes in actual cancer patients in a clinical trial
- Attempt to target other types of cancer (expand the research)
Herpes does not effectively target cancer cells
- The virus was not selective enough (was toxic to human cells as well)
- The virus did not function (its ability to destroy cells was demolished)
- The virus remained active even after the cancer was gone
- If any of the above occurs, we would change our experiment, and propose a new method of approach, perhaps including:
- Returning to using Herpes virus to kill cancer only in regions of slow normal cell growth
- Testing other viruses for ease of modification
- Looking for different possible sites of mutation/regulation within Herpes.
Resources (Material)
- G207
- pp17b
- PCR materials
- restriction enzymes
- buffers
- Human tissue cells
- Human cancer cells
- Mice (immuno-competent)
...