Godin:Protocols/Piranha
From OpenWetWare
Jump to navigationJump to search
|
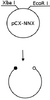
It has been requested that this page be removed with restriction endonucleases. Other articles for deletion are listed here. |

|
It has been requested that this page be removed with restriction endonucleases. Other articles for deletion are listed here. |