Genentech
Genentech Inc.


Genentech Inc. is a biotechnology corporation headquartered in South San Francisco, California. Since 2009, Genentech has been a subsidiary of the Roche Group, a Swiss healthcare company. [1] Genentech boasts a staggering $17.3 billion revenue worldwide [2] and employs nearly 14,800 people across the globe. The corporation holds approximately 11,300 patents and has 38 medicines currently out in the market with an additional 46 molecules in clinical trials or currently in their pipeline. Ian Clark is the chief executive officer of Genentech and was appointed to his role in January of 2010. The biotech giant has 785,000 square feet solely devoted to research at its headquarters in San Francisco and at their additional manufacturing facilities in Vacaville, California, Oceanside, California, and Hillsboro, Oregon. [3] These manufacturing plants also have the capacity to produce approximately 330,000 liters of products at a time, based on the size of their bioreactors.[4]
The branches of research that Genentech currently studies includes oncology, immunology, tissue growth and repair, ophthalmology, metabolism, neuroscience, and infectious diseases. Genentech's main goal is to discover, develop, manufacture, and commercialize innovative medicines that are helping treat the most serious health problems humans face today. The company follows three important Roche values of passion, courage, and integrity.[5] Genentech currently has just under 10,500 publications (10,449 exactly) as of April 24th, 2017.[6] They also garner the three best-selling cancer drugs in the United States of Rituxan, Avastin, and Herceptin [7] and are constantly continuing to research new ways to solve these health crises with two new FDA approvals as recent as April 17th, 2017.
History

Genentech was co-founded on April 7th, 1976 by biochemist Herbert Boyer and venture capitalist Robert Swanson. Boyer, at the time, was considered to be one of the pioneers in biotechnology and he demonstrated this with his breakthrough work with restriction enzymes in 1973 [2]. Boyer and his Stanford colleague, Stanley Cohen showed that restriction enzymes could be used to cut DNA fragments and then inserted into a similar plasmid vector via ligation. This process became its own scientific field called recombinant DNA technology, thus giving Boyer a household name. Robert Swanson became intrigued by Boyer and requested a meeting with him. The two hit it off with Swanson truly believing that Boyer's recombinant DNA technology could be a major commercial selling platform and with that the biotechnology giant, Genentech was born. In 1977, Genentech had its first real breakthrough as a company. The company's scientists and researchers had successfully produced Somatostatin, the first human protein, by inserting the gene into an E.coli plasmid. A year later, Genentech was able to clone synthetic human insulin, which gave the company its second massive innovation. Genentech, then took over Wall Street when it went public in 1980, by raising $35 million with an offering, in which leapt its $35 a share to a high of $88 a share within hours, which is one the stock markets largest rise ups, still to date [9].
Other historical timelines [9]

1982: Synthetic Human Insulin (Humulin) was marketed with the help of Eli Lilly and Company
1985: Received approval from FDA to market first product, Protropin. Protropin is a growth hormone for children with growth deficiency. This was the first recombinant pharmaceutical product to be manufactured and marketed by a biotechnology company.
1987: Genentech's second product, Activase gained FDA approval.
1990: Roche Holding Ltl from Switzerland, takes a majority holding of Genentech Inc.
1992: The Founders Research Center was opened, in honor of Boyer and Swanson, aimed to recognize their visions in biotechnology.
1993: Received FDA approval for the drugs Pulmozyme and Nutropin.
1995: Roche takes over Genentech's European and Canadian sales
1997: Partnered with IDEC Pharmaceuticals (now Biogen Idec Inc.), received FDA approval to market Rituxan.
1998: Humanized antibody Herceptin received FDA approval, opens new $250 million manufacturing facility
1999: Settled a patent dispute relating Protropin, human growth hormone, with University of California for $200 million.
2000: Received FDA approval for TNKase.
2003: Received FDA approval for the drugs Xolair and Raptiva.
2004: Received approval to market Avastin. Avastin is the first FDA approved therapy to tackle angiogenesis.
2005: Purchased Biogen Idec’s biologics manufacturing facility in Oceanside, California.
2007: Finalized acquisition of Tanox, a pharmaceutical company based in Houston, Texas, which allowed Genentech to improve the business of Xolair.
2009: Roche acquires all of Genentech's stock shares.
2010: Received FDA approval for Tarceva as a maintenance treatment for patients with locally advanced or metastatic non-small lung cancer (NSCLC).
2012: Erivedge was approved for the treatment of adults with a type of skin cancer, called basal cell carcinoma (BCC), Tamiflu was FDA approved.
2013: Gazyva and Kadcyla are approved.
2015: Alecensa and Cotellic are FDA approved.
2016: Venclexta and Tecentriq are FDA approved.
List of All Products[10]
Genentech currently has 38 different medicines that are on the market with FDA approval. The products name and its chemical name are as follows:
Actemra (tocilizumab)
Activase (alteplase)
Alecensa (alectinib)
Avastin (bevacizumab)
Boniva Injection (ibandronate sodium)
Boniva Tablets (ibandronate sodium)
Cathflo Activase (alteplase)
CellCept (mycophenolate mofetil)
Copegus (ribavirin)
Cotellic (cobimetinib)
Cytovene (ganciclovir)
Erivedge (vismodegib)
Esbriet (pirfenidone)
Fuzeon (enfuvirtide)
Gazyva (obinutuzumab)
Herceptin (trastuzumab)
Invirase (saquinavir mesylate)
Kadcyla (ado-trastuzumab emtansine)
Klonopin (clonazepam)
Lucentis (ranibizumab injection)
Nutropin AQ (somatropin for injection)
Ocrevus (ocrelizumab)
Pegasys (peginterferon alfa-2a)
Perjeta (pertuzumab)
Pulmozyme (dornase alfa)
Rituxan (rituximab (oncology))
Rituxan (rituximab (immunology))
Tamiflu (oseltamivir phosphate)
Tarceva (erlotinib)
Tecentriq (atezolizumab)
TNKase (tenecteplase)
Valcyte (valganciclovir hydrochloride)
Valium (diazepam)
Venclexta (venetoclax)
Xeloda (capecitabine)
Xenical (orlistat)
Xolair (omalizumab)
Zelboraf (vemurafenib)
Genentech's Major Products
Rituxan (rituximab)
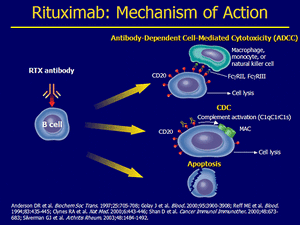
Rituxan (rituximab) is a chimeric monoclonal antibody therapy that can be used alone or with chemotherapy to treat for the cancers of Non-Hodgkin's lymphoma and chronic lymphocytic leukemia that was approved by the FDA in 1997. Rituxan achieves this goal by targeting and attaching to the CD20 protein that are found on cancerous blood cells and as well as some healthy blood cells. Rituxan disrupts blood cells by aiding the immune system to destroy cancer cells, destroying the cells combined with chemotherapy or destroying the cancer cells on its own. [13] This downregulates blood cell receptors and induces apoptosis of CD20+ cells. The combined effect of downregulation and apoptosis results in the removal of blood cells, allowing healthy blood cells to develop from lymphoid stem cells. [14]
Rituxan is used for different purposes when treating for either Non-Hodgkin's lymphoma or chronic lymphocytic leukemia. Non-Hodgkin's lymphoma is the cancer that occurs in lymphocytes, a type of white blood cells, that are crucial to a person's immune system to function properly and fight disease. A patient with NHL starts to build up too many cancerous white blood cells in bone marrow, lymph nodes, blood, and spleen which can limit function in those parts of the body. Rituxan can treat CD20+ Non Hodgkin's lymphoma as a first-time, single agent to the low-grade and follicular forms the cancer, it can be mixed with chemotherapy for the follicular form, or can be used after CVP chemotherapy was not successful for low-grade forms. Chronic lymphocytic leukemia is a type of blood cancer that also affects lymphocyte cells. CLL forces cancerous cells to overwhelm the healthy cells in the blood which leads to fewer white blood cells, fewer red blood cells, and fewer platelets. Rituxan treats chronic lymphocytic leukemia that is CD20+ by combining it with FC chemotherapy as an initial treatment for the cancer.
Rituxan also has various side effects. The most common side effects of Rituxan are infections, chills, fatigue, infusion reactions, low white blood cells, and body aches. More serious side effects include painful sores, hives, ulcers, hepatitis B, and a disease called Progressive Multifocal Leukoencephalopathy, which is a serious brain infection that can lead to death.[13] Genentech classifies Rituxan as a specialty drug. Rituxan is also the best selling cancer drug on the market based on 2016 statistics, accumulating $3 billion in sales during the calendar year.[7] Rituxan has been called the biggest breakthrough in lymphoma treatment in the past decade.[15]
Avastin (bevacizumab)
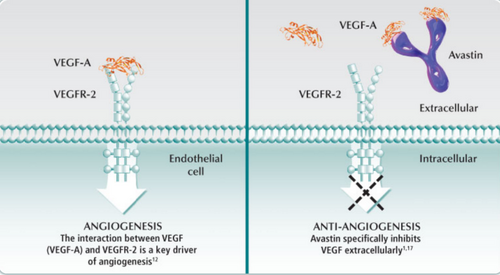
Avastin (bevacizumab) is a recombinant humanized monoclonal antibody that is used as an angiogenesis inhibitor to tumor cells by blocking new blood vessels from forming at the tumor site. Avastin, which was approved by the FDA in 2004, is designed to be used to fight many different cancers: metastatic colorectal cancer, advanced cervical cancer, advanced nonsquamous non-small cell lung cancer, metastatic kidney cancer, and recurrent ovarian cancer. Avastin is different from chemotherapy by instead of attacking the tumor, Avastin starves the tumor from blood vessels, thus stunting its growth. The drug binds to the vascular endothelial growth factor (VEGF), which promotes blood flow to the tumor, and in many cancer cells is overexpressed. By binding to the VEGF, Avastin prevents VEGF from reaching the tumor site, which blocks angiogenesis and makes the tumor unable to grow. Avastin is administered to a patient via intravenous infusion.
Avastin, like many other cancer drugs, can only be used in certain circumstances for the cancers it treats. For metastatic colorectal cancer, Avastin must be combined with 5-fluorouracil-based chemotherapy in either first line or second line treatment to be effective, while in advanced nonsquamous non-small cell lung cancer, the drug must be combined with carboplatin and paciltaxel in people who haven't received chemotherapy. Similar combinations are made in advanced cervical cancer with Avastin injected with paciltaxel and either cisplatin or topotecan and metastatic kidney cancer must have interferon alfa with it. Recurrent ovarian cancer also must be combined with paciltaxel and either topotecan or pegylated liposomal doxorubicin. Minor side effects of Avastin are high blood pressure, recal bleeding, nosebleeds, too much pressure in urine, back pain, head ache or inflamed skin on the face. The major side effects are wounds that don't heal, serious bleeding that includes throwing up blood, and gastrointestinal perforation, which puts a hole in patient's stomach or intestine.[17] Avastin also used to be used for breast cancer treatment until November 2011, when the FDA revoked approval, stating that the drug was not helpful towards breast cancer treatment.[18] Avastin is the second best-selling drug for Genentech, by $2.66 billion in sales in 2016.[7]
Herceptin (trastuzumab)
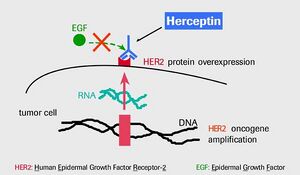
Herceptin (trastuzumab) is a humanized monoclonal antibody that targets and inhibits Human Epidermal growth factor Receptor 2-positive (HER2+), which is commonly found in certain forms of breast cancer. Herceptin was developed by Genentech in partner with UCLA, and approved by the FDA in 1998. The drug is used to treat two different forms of HER2+ breast cancers: adjuvant breast cancer, and metastatic breast cancer. In both forms of these cancers, the HER2+ gene is overexpressed to form HER2+ receptors, which send signals to breast cells to proliferate and divide. This overexpression of receptors, in turn causes cancerous cells to form. Herceptin does its job as a target therapy by attacking these receptors and binding to them, thus preventing them from being able to send more signals to cancerous breast cells to grow and divide.
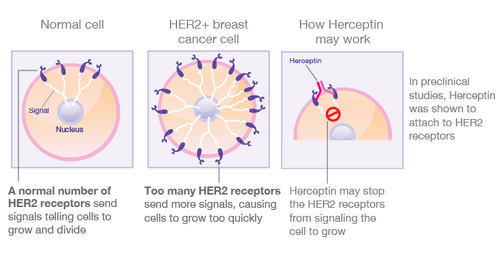
Herceptin is usually combined with a chemotherapy drug so that Herceptin blocks the HER2+ signals from producing new cancerous cells while the chemotherapy kills the cancer cells already established. Herceptin has three approved treatments for adjuvant breast cancer. The first treatment approved is Herceptin mixed with the chemotherapy drugs of doxorubin, cyclophosphamide, and either docetaxel or paciltaxel which is known as AC→TH. The second treatment is a mixture of Herceptin with the drugs of carboplatin and docetaxel, known as TCH. The last approved treatment is do it alone after chemotherapy has already been conducted. As for treatments for patients with metastatic breast cancer, the options are more limited for Herceptin use. Herceptin can only be used in this form of cancer in either a first line treatment with paciltaxel or by itself as a second-line treatment after chemotherapy has been used.
Herceptin is given to patients through intravenous infusion. Herceptin, like every other drug, has its share of side effects. The most common side effects of Herceptin are fever, nausea, vomiting, diarrhea, headache, fatigue, shortness of breath, rash, infections, increased cough, low white blood cell count, and muscle pain. More serious side effects include infusion infections like fever, chills, nausea, and vomiting, heart problems like congestive heart failure, and severe lung problems like fluid leakage into the lungs, lung swelling, and lack of oxygen.[19] Herceptin is the third best selling cancer drug in the United States, grossing $1.66 billion in 2016.[7]
Recent Innovations and Research
Tecentriq (atezolizumab)
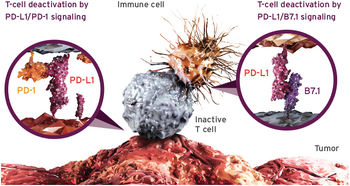
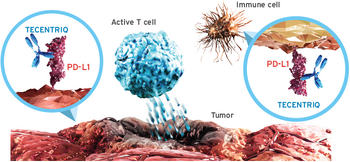
In 2016, Genentech's product of Tecentriq (atezolizumab) was approved by the FDA to treat for locally advanced or metastatic urothelial carcinoma and metastatic non-small cell lung cancer. Tecentriq, is a a programmed death ligand that targets the death of the protein of PD-L1 found in cancer cells. The protein PDL1 is found on cancer cells and can make the cancer cells hide themselves from the immune system antibodies. Thus, when Tecentriq enters the system, it kills the PDL1 protein by blocking it and gives the body's immune system (creation of T cells) a better chance to eliminate the cancer cells. Tecentriq is administered to a patient via intravenous infusion for 60 minutes every three weeks with 1200 mg given in each dose. The drug is specific to only certain restrictions when either having urothelial carcinoma or non-small lung cancer. For urothelial carcinoma, a form of bladder cancer, Tecentriq can only be used when the cancer has spread and cannot be removed by surgery, and the patient has tried chemotherapy containing platinum, which was unsuccessful.[21] As for non-small cell lung cancer, Tecentriq is used when after platinum chemotherapy is completed and the cancer is still, as studies have shown that the Tecentriq is much more successful than a second round of chemotherapy.[22]
On April 17, 2017, Tecentriq gained accelerated approval by the FDA to be used for patients with metastatic urothelial carcinoma who could not use cisplatin chemotherapy as a treatment option.[23] More than half of people who are diagnosed with advanced urothelial carcinoma are unable to use cisplatin as a chemotherapy to help cure the cancer. With this early approval by the FDA, from a Stage II trial that was quite successful, many patients now have an option to treat the urothelial carcinoma. Even though Tecentriq received approval from Stage II by having more than 50% of the patients in the study respond to the drug, Genentech is currently still conducting a Stage III trial to confirm the results that the drug is effective for patients that can not use cisplatin chemotherapy.[24]
There are many side effects to Tecentriq. The biggest and most important side effect to Tecentriq is the fact that it can possibly effect the organs in the patients body that have nothing wrong with them. Tecentriq can create pneumonitis in the lungs, hepatitis in the liver, colitis in the intestines, stop function in the hormone glands, neuropathy or meningitis in the nervous system, or cause serious eye inflammation. The most common side effects, however are slightly different when treating for metastatic urothelial carcinoma or non-small cell lung cancer. In urothelial carcinoma, the most common side effects are fatigue, lost of appetite, fever, urinary tract infection, constipation, and nausea. In non-small cell lung cancer, the side effects are fatigue, lost of appetite, nausea, cough, shortness of breath, and constipation.[21]
Lucentis (ranibizumab injection) for Diabetic Retinopathy
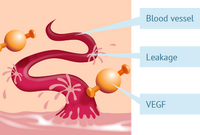
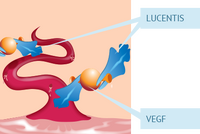
Lucentis was first approved by the FDA in 2006 to treat patients suffering from wet age-related macular degeneration (wet AMD) and in 2012 to treat for diabetic macular edema (DME), both of which cause blindness to the eye.[24] On April 17, 2017, Lucentis was proven with FDA approval to be able to treat for all forms of diabetic retinopathy, which directly damages the retina. Diabetic retinopathy is when poorly controlled blood sugar counts stemming from diabetes, causes the blood vessels in tissue behind the eyes (retina) to leak, thus effecting a person's vision with the excess fluid. The leaking of the blood vessels in the retina is caused by the overexpression of the VEGF protein (vascular endothelial growth factor), which is essential in vessels. Diabetic retinopathy is the leading cause of vision loss in working class U.S. adults between 20-74 and effects 7.7 million Americans, thus making a solution a necessity. Lucentis is therefore designed to inhibit VEGF-A by binding to it and eliminating its angiogenesis and hyperpermeability, which has shown in a most recent study to greatly relieve diabetic retinopathy. [26]
The process of receiving Lucentis to the body is done through a direct injection to the infected eye. The process was done every month with 0.3 mg of Lucentis injected each time. There are some minor and some serious side effects to Lucentis. The minor side effects include eye pain, increased eye pressure, redness of the eye, and small specks in a person's vision. The major side effects of Lucentis are cataracts, detached retinas, and serious infections inside the eye.[25]
Career Opportunities [27]
There are currently 318 job openings posted on the Genentech website (https://www.gene.com/careers/find-a-job) as of April 23rd, 2017. The majority of available positions look for chemistry, biochemistry, mechanical engineering, and chemical engineering majors although there are also jobs in marketing and business analysis. Experience level varies by the job description, but Genentech offers jobs to student internships, regular employees, special programs and temporary workers at all of their business and manufacturing sites. Entry level bachelors are encouraged to apply to the rotation programs. Process development rotation program associates will experience four six month assignments over the course of two years in different areas within US Biologics Technical Development. Genentech hires approximately 3500 people a year in a four step process listed as Discovery, Evaluation, Offer, and Welcome.
Awards & Recognition [28]

“100 Best Companies to Work For” awarded for the 19th consecutive year, 6th overall on list – Fortune (2017)
"Top Twenty Employers"- Science Magazine (2016)
"Top 50 Corporate Philanthropy Awards"- Silicon Valley Business Journal (2016)
“Best Places to Work for LGBT Equality”- Corporate Equality Index (2016)
"Top 50 Companies for Diversity"- Diversity Inc (2016)
“100 Best Companies for Working Mothers” – Working Mother Magazine (2016)
“Healthiest Companies to Work For in America” – Greatist.com (2014)
“The World’s 50 Most Innovative Companies” – Fast Company (2012)
References
[1] Smith Hughes, S. (2011). Genentech: The Beginnings of Biotech. [1]
[2] Godar, M. (2015). History of Biotech: How Modern Biotechnology Started-Up 39 Years Ago. [2]
[3] Genentech Inc. (2017). Innovative Clinical Science. Improved Patient Care. Genentech Inc. [3]
[4] Genentech Inc. (2017). Genentech Manufacturing. Genentech Inc. [4]
[5] Genentech Inc. (2017). About Us. Genentech Inc. [5]
[6] Genentech Inc. (2017). Publications Genentech Inc.. [6]
[7] Staton, T. (2017). Top 10 Best-selling Cancer Drugs. Questex. [7]
[8] Glassdoor. (2017). Glassdoor Inc. [8]
[9] Genentech Inc. (2017). A History of Firsts. Genentech Inc. [9]
[10] Genentech Inc. (2017). Our Medicines. Genentech Inc. [10]
[11] Tansey, B. (2005). Cancer drugs drive up Genentech profit. San Francisco Gate. [11]
[12] InterMune. (2014). SEC. [12]
[13] Genentech Inc & Biogen Inc. (2017). What is Rituxan. RITUXAN®. [13]
[14] Weiner GJ. (2010). Rituximab: mechanism of action. Seminars in hematology.115-123. [14]
[15] Dotan, E, Aggarwal, C, Smith, M. (2010). Impact of Rituximab (Rituxan) on the Treatment of B-Cell Non-Hodgkin’s Lymphoma. Pharmacy and Therapeutics35(3), 148-157. [15]
[16] Pinterest. (2017) [16]
[17] Genentech Inc. (2017). Avastin® (Bevacizumab). [17]
[18] Genentech Inc. (2011). Genentech Inc. [18]
[19] Genentech Inc. (2017). Fight HER2+ Breast Cancer With Herceptin. Herceptin® (Trastuzumab). [19]
[20] Hudis, C. (2007). Trastuzumab — Mechanism Of Action And Use In Clinical Practice. New England Journal of Medicine (357.1), 39-51. [20]
[21] Genentech Inc. (2017). What is Tecentriq. Tecentriq® (atezolizumab). [21]
[22] Vansteenkiste, J, Wauters, E, Park, K, Rittmeyer, A, Sandler, A, Spira, A. (2017). Prospects and progress of atezolizumab in non-small cell lung cancer. Expert Opinion on Biological Therapy. [22]
[23] Genentech Inc. (2017). FDA Grants Genentech’s Tecentriq (Atezolizumab) Accelerated Approval as Initial Treatment for Certain People with Advanced Bladder Cancer. Genentech Inc. [23]
[24] Balar, A.V, Galsky, M.D, Rosenberg, J.E, Powles, T, Petrylak, D.P, Bellmunt, J, Loriot, Y, Necchi, A, Hoffman-Censits, J, Perez-Gracia, J.L, Dawson, N.A, van der Heijden, M.S, Dreicer, R, Srinivas, S, Retz, M.M, Joseph, R.W, Drakaki, A, Vaishampayan, U.N, Sridhar, S.S, Quinn, D.I, Durán, I, Shaffer, D.R, Eigl, B.J, Grivas, P.D, Yu, E.Y, Li, S, Kadel, E.E, Boyd, Z, Bourgon, R, Hegde, P.S, Mariathasan, S, Thåström, A, Abidoye, O.O, Fine, G.D, Bajorin, D.F. (2017). Atezolizumab as first-line treatment in cisplatin-ineligible patients with locally advanced and metastatic urothelial carcinoma: a single-arm, multicentre, phase 2 trial. The Lancet389(10064), 67-76. [24]
[25] Genentech Inc. (2017). What is Lucentis. Lucentis® (ranibizumab injection). [25]
[26] Genentech Inc. (2017). FDA Approves Genentech’s Lucentis (Ranibizumab Injection) for Diabetic Retinopathy, the Leading Cause of Blindness Among Working Age Adults in the United States. Genentech Inc. [26]
[27] Genentech Inc. (2017). Job Search. Genentech Inc. [27]
[28] Genentech Inc. (2017). Awards and Recognition. Genentech Inc. [28]
