Fast Lithographic Activation of Sheets (FLASH) - Louis Colaruotolo
Summary
Fast Lithographic Acitivation of Sheets (FLASH) is an exceedingly simple method of photolithography that employs the most basic of methods to create detailed, well structured, and accurately dimensional microfluidic devices.

FLASH employs the simple principals of photolithography using a medium of paper rather than silicon as a base for the Point of Diagnostic (POD) surface. This technique radically reduces the cost of creating a microfluidic device, and is especially useful for prototyping devices as the method is quick, and very user friendly. Fairly recent in the world of microfluidics, the Martinez Lab (Figure 1) based out of Cal Poly has contributed the majority of literature for FLASH by pioneering the field of method development, check out their website for more information.
Materials
The novel nature of FLASH resides in the small amount of materials needed for the creation of microfluidic devices, and the omission of the need for laboratory grade equipment. Five sets of materials are needed:
- Paper (Figure 2)

Figure 2.Whatman chromatography paper NO. 1. - Photoresist (Figure 3)

Figure 3. Various volatile chemical solvents are used with mixed photoresist molecules. - Printer (Figure 4)

Figure 4. Classic desktop printers are a easy option for design. - Light source (Figure 5)

Figure 5. Handheld or free standing lamps all work equally well. - Hot plate (Figure 6)

Figure 6. Any source of heat will do, but a hot plate offers a flat surface for polymer curing.
This list of five basic materials can be tailored to fit the best purpose of your project's specifications and demand for accuracy.
Paper based microfluidic devices are becoming more mainstream in recent years as their cheap production values allow for mass production with little cost. Many medical testing devices are fabricated using paper fluidics including tuberculosis tests[7], malaria diagnosis[2], and other various portable medical tools.[12] Many different types of papers are used ranging from copy paper to chromatography paper. As a proof of concept, microfluidic devices can even be built on composite papers like toilet paper
The photoresist will allow for certain areas of the paper to be resistant to the curing process, which in turn allows for the differentiation of hydrophilic and hydrophobic channels. Many different photoresists can be used for FLASH, some examples range from: epoxy-based photoresists in a propylene glycol monomethyl ether acetate solution,[10] dry film negative photoresists[9], and diazo based positive photoresists[9]. The differences between positive and negative photoresists allow the designer to choose to allow etching or disallow etching dependent on the positive or negative nature, which creates hydrophobic and hydrophilic channels which dictates flow (Figure 7). Procedure of this methodology in FLASH to follow.
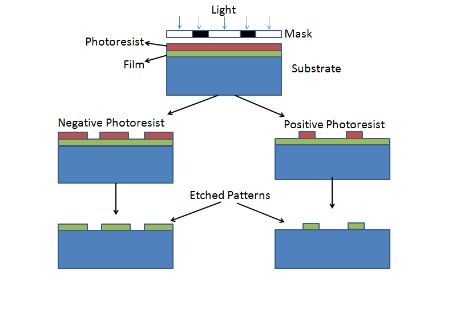
Traditional ink jet printers or laser printers can be used for FLASH due to the high simplicity of the procedure. The ink must be water proof which most traditional laser printing devices currently employ. It has been suggested that instead of the use of a printing device, a water-proof black ink marker can be used to outline the design which reduces the time needed to make a design, but suffers from a decrease of accuracy and precision in the replication process.[10] The depth of the channels are directly related to the depth of the ink in the paper (which is limited by the paper thickness), so without applying stacked layers, FLASH is limited to 2D structures
A light source is needed to provide the UV light for the curing process. dependent on the type of photoresist used, the UV light will etch a pattern. UV lamps can be employed, but to further simplify this procedure, the UV from sunlight is sufficient enough. Using a UV light will decrease the amount of time needed for the curing process, but the wavelengths of light produced by the sun at it's intensity with no additional equipment works just as well (with added processing time).[10]
Lastly, a hot plate is used to thermally cure and cross link the polymers in the the photoresist to create the channels and features in the microfluidic device. A hot plate has all the qualifications needed. If sunlight is used to etch the design and cross link the polymers the heating plate is not needed.[10]
Procedure
Due to the highly simple procedure of FLASH, the steps from start to end can be broken down in eight simple steps.[10]
1. Impregnate paper with a photoresist
2. Dry the paper to remove the solvent
3. Cover one side with adhesive transparent film and the other with black construction paper
4. Print a pattern onto the film (or use a marker) to create a mask
5. Expose to UV
6. Remove the film and construction paper
7. Bake the paper to polymerize the photoresist
8. Wash in propanol and acetone to remove polymerized resist
This procedure is not entirely unique to FLASH, but rather is standard for most photolithography operations. The procedural separation of FLASH from traditional photolithography is how the photoresist is applied. The following describes the methodology (Figure 8) used by the Martinez group in the creation of the first FLASH microfluidic devices:
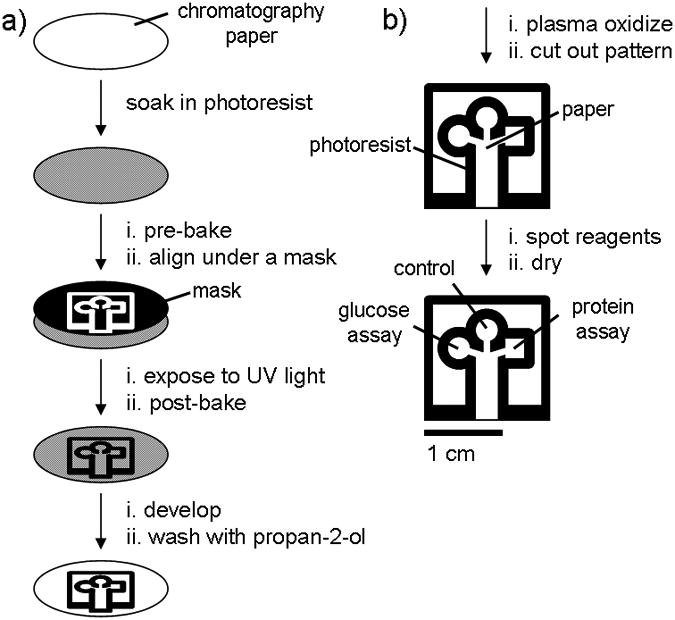
Specific details of the procedure are as follows:
"We patterned chromatography paper with SU-8 2010 photoresist as shown in (Figure 8) and as described below: we soaked a 7.5-cm diameter piece of chromatography paper in 2 mL of SU-8 2010 for 30 s, spun it at 2000 rpm for 30 s, and then baked it at 95 °C for 5 min to remove the cyclopentanone in the SU-8 formula. We then exposed the photoresist and paper to 405 nm UV light (50 mW/cm2) for 10 s through a photo-mask [transparent film] (CAD/Art Services, Inc.) that was aligned using a mask aligner (OL-2 Mask Aligner, AB-M, Inc). After exposure, we baked the paper a second time at 95 °C for 5 min to cross-link the exposed portions of the resist. The unpolymerized photoresist was removed by soaking the paper in propylene glycol monomethyl ether acetate (PGMEA) (5 min), and by washing the pattern with propan-2-ol (3 × 10 mL)" [4].
Note that the paper must be soaked in a thinner than typical photoresist then spin coated rather than simply spin coated. This treatment is semi-unique to the FLASH procedure because of the fragility of the paper medium. It ensures even distribution of the photoresist, and effectively coats both sides of the paper (necessary for this type of paper fluidic device). Not mentioned in the above procedure is the use of black construction paper, this allows for the paper to be resistant to full penetration by the UV light, allowing for the formation of 2D channels with micro dimensions without completely breaking through the paper. The Black paper is peeled off before polymerization in step 6.
Benefits
FLASH shines brightly in three ways: ease of use, low cost, and appreciable patterning resolution
Considering that only 8 steps are needed to go from start to finished product and the lack of expensive equipment needed using the FLASH method for creating a paper based microfluidic device, the benefit of easy use is incredibly clear. This type of project can be carried out in small labs, and even in class room settings. photoresist impregnated sheets can be shipped and stored in light excluding packages, but are sensitive to light (since sunlight can cure them), this of course is a slight limitation, which will require the designers to work in a dark environment. The fact that the designs can be hand drawn using black marker is not only convenient, but a quick and easy way to make concept designs that are ready to test in a matter of minutes rather than days [6]. The most profound of all advantages is that there is no need for a clean room in making a device using FLASH which greatly benefits small labs [11].
The cost for FLASH paper based microfluidic devices is very low due to the uncomplicated nature of the procedure. This allows for the development of simple and greatly inexpensive diagnostic assays in less industrialized countries where the need for these devices is high, but the ability to create silicone based devices is low [4]. Even when more complicated devices are needed that extend the depth of single sheet FLASH, two-ply FLASH double the depth capabilities and design abilities while only increasing the cost of the paper materials while others costs remain steady [1]. Below is a cost and dimensional accuracy analysis (Figure 9) of different paper materials using FLASH.

In a comparison to normal paper fluidics, the material cost is quite comparable. The real cost saving component is not needing highly technical equipment to produce these sheets and masks, and most notably is not needing a clean room to conduct these procedures. This combination of money saving attributes of FLASH make it the ultimate prototyping device, allowing multiple prototypes to be made without the worry of cost, and the ease of subsequent production.
FLASH is certainly limited in the resolution of channels and features on the paper based microfluidic device, but with the combination of a few technologies and methods the resolution of features can increase to a workable level. Jet and flash imprint lithogrpahy (J-FIL) combines the concept of FLASH with jet fluid printing to allow for more precise design allow for the patterning of sub-25nm features [6]. Further dimensional detail can be achieved using FLASH but substituting the paper medium for a cloth based sheet, allowing for more dimensional resolution on the z-axis which allows for deeper more complex designs [3].
Applicability to the real world
FLASH being as simple, cheap, and accessible as it is could be means to revolutionize the way point of care diagnostic tests are made and distributed. Not only does the ease of prototyping help small and large labs in developing new paper microfluidic devices, but this method enables countries with little to no industrial infrastructure to make their own devices. The need for diagnostic testing of AIDS, tuberculosis, and malaria in developing countries is critical due to the infectious nature of these diseases [2].
The development of simple point of diagnostic devices like pregnancy tests all the way to more complicated fully enclosed paper devices that suffer from quick solvent volitility [8] are possible. With the continuing development of paper fluidic products that are used in developing countries, not only benefits the disenfranchised country, but the industry of those who manufacture the product. This allows for smaller less industrialized areas to make their own devices, saving on shipping cost, and empowers the nation by providing a sustainable means of aiding their own need for detection devices [5].
References
[1] Camplisson, C. K.; Schilling, K. M.; Pedrotti, W. L.; Stone, H. A.; Martinez, A. W. Two-ply channels for faster wicking in paper-based microfluidic devices. Lab on a Chip 2015, 15(23), 4461-4466, https://doi.org/10.1039/c5lc01115a
[2)] Lee, W. G.; Kim, Y.; Chung, B. G.; Demirci, U.; Khademhosseini, A. Nano/Microfluidics for diagnosis of infectious diseases in developing countries. Adv. Drug Delivery Rev. 2010, 62(4), 449-457, https://doi.org/10.1016/j.addr.2009.11.016
[3] Li, H.; Liu, C.; Wang, D.; Zhang, C. Programmable fluid transport on photolithographically micropatterned cloth devices: Towards the development of facile, multifunctional colorimetric diagnostic platforms. Sens. Actuators, B. 2018, 255(2), 2416-2430, https://doi.org/10.1016/j.snb.2017.08.215
[4] Martinez, A. W.; Phillips, S. T.; Butte, M. J.; Whitesides, G. M. Patterned paper as a platform for inexpensive, low-volume, portable bioassays. Angew. Chem., Int. Ed. 2007, 46(8), 1318-1320, https://doi.org/10.1002/anie.200603817
[5] Martinez, A. W.; Phillips, S. T.; Whitesides, G. M.; Carrilho, E. Diagnostics for the developing world: Microfluidic paper-based analytical devices. Analy. Chem. 2009, 82, 3-10, https://doi.org/10.1021/ac9013989
[6] Cherala, A.; Schumaker, P.; Mokaberi, B.; Selindis, K.; Choi, B.; Meissi, M. J.; Khusnatdinov, N. N.; LaBrake, D.; Sreenivasan, S.V. Nanoscale magnification and shape control system for precision overlay in jet and flash imprint lithography. IEEE/ASME Transactions on Mechatronics 2015, 20(1), 122-132, https://doi.org/10.1109/TMECH.2013.2297679
[7] Ruan, Y. L.; Tsai, T.; Tsai, T.; Shen, S.; Chen, C.; Cheng, C. Paper-based tuberculosis diagnostic devices with colorimetric gold nanoparticles. Sci. Technol. Adv. Mater. 2013, 14(4), https://doi.org/10.1088/1468-6996/14/4/044404
[8] Schilling, K. M.; Lepore, A. L.; Kurian, J. A.; Martinez, A. W. Fully enclosed microfluidic paper-based analytical devices. Analy. Chem. 2012, 84(3), 1579-1585, https://doi.org/10.1021/ac202837s
[9] Shaw, J. M.; Gelorme, J. D. (1997). Negative photoresists for optical lithography. IBM J. Res. Dev. '1997, 41(1), 81-94, https://doi.org/10.1147/rd.411.0081
[10] Wiley, B.; Phillips; S.; Martinez, A. W.; Phillips, S. T.; Wiley, B. J.; Gupta, M.; Whitesides, G. M. FLASH: A rapid method for prototyping paper-based microfluidic devices. Lab on a Chip 2008, 12(8), 2146-2150, https://doi.org/10.1039/B811135A
[11] Xia, Y.; Si, J.; Li, Z. Fabrication techniques for microfluidic paper-based analytical devices and their applications for biological testing: A review. Biosens. Bioelectron. 2016, 77, 774-789, https://doi.org/10.1016/j.bios.2015.10.032
[12] Zhang, Y.; Ma, H.; Wu, D.; Li, Y.; Du, B.; Wei, Q. Label-free immunosensor based on nanoparticles/magnetic chitosan matrix for sensitive determination of ractopamine. J. Electroanal. Chem. 2015, 741, 14-19, https://doi.org/10.1016/j.jelechem.2015.01.007
