Extraction of PCB from Spirulina
This protocol was received from the Quail Lab at UC-Berkely
Day 1
1. Suspend 50g of Spirulina powder in 1.5 L of H2O and stir for 10 minutes. Divide into 6 250 mL centrifuge bottles and centrifuge at 8000 RPM, GS3 motor, 4°C for 1 hour.
2. Decant the supernatant back into a 2 L flask and add 15 g of TCA. Stir for 1 hour in the dark (covered in aluminum foil), and then centrifuge at 8000 RPM, GS3 motor, 4°C for 10 minutes. Discard supernatant.
3. Add 25 mL of MeOH to each bottle, resuspending the green pellets completely (can use vortex), and then add MeOH to bottle until a final volume of 250 mL is reached. Centrifuge at 8000 RPM, GS3 motor, 4°C for 10 minutes and then discard the supernatant.
4. Repeat step 3 four times or until the MeOH wash is colorless.
5. Store pellets at -20°C, wrapped in foil overnight.
It is critical that all steps from here on are performed under green 'safelight' conditions in a dark room (Light cannot emit a wavelength of 600-750nm), or that all samples are covered in foil. The free chromophore can become photobleached.
Day 2 - Methanolysis
6. Resuspend and pool pellets in a final volume of 500 mL.
7. Heat suspension in a 1-liter flask in a water bath at 70 – 75 ºC with a condensing coil cooled with tap water (see below) for 5-8 hrs (very slow boil).
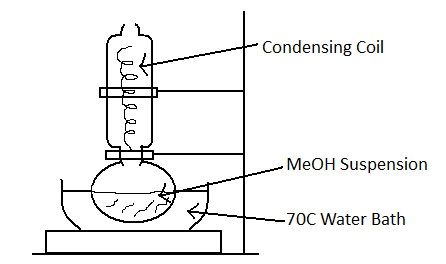
8. Transfer suspension to two 250 mL bottles and centrifuge 8000 RPM,GS3 rotor, 4º C for 20 min.
9.Decant and filter supernatant through miracloth or cheesecloth directly into a 1-liter side-armed flask and store, foil-wrapped at –20 ºC (1st methanolysis supernatant).
10. Repeat steps 6-9 with pellet overnight.
Day 3
11.Remove 1st methanolysis supernatant (step 9) from freezer and thaw to room temperature and connect to another side-armed flask which is attached to a vacuum pump and sitting in a dry ice / EtOH bath (cold trap collection flask). Monitor volume of MeOH in collection flask until it reaches 400 mL. This process will take 9-10 hrs depending on vacuum. Store concentrated PCB/MeOH remaining in the original flask(~50 mL) foil-wrapped, at -20 ºC. See below for setup.
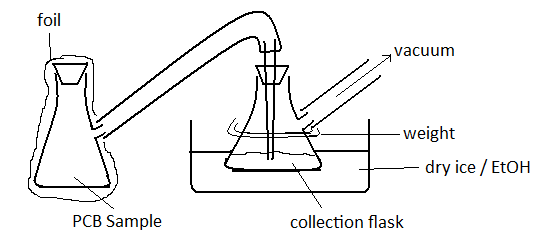
Day 4
12. In a large separatory funnel, mix concentrated PCB/MeOH (~50 mL) with 50 mL chloroform and 100 mL H2O. Shake and let stand for 5 min.
13. Collect chloroform (bottom phase) in a beaker. This contains the PCB.
14. Add another 50 mL chloroform to remaining aqueous phase and repeat step 13. Continue with chloroform extraction until solution is almost colorless (usually 2-3X).
15. Pool these several chloroform/PCB fractions and transfer to a 1 L round bottom flask. Evaporate chloroform with a stream of nitrogen gas or air.
16. Dissolve dried PCB in 4 X 0.75 mL DMSO – Final volume ~3 mL. This is the stock solution. Remove samples to determine PCB concentration (see below), then aliquot remaining DMSO/PCB and store, foil-wrapped, at –80 ºC.
17 – Repeat steps 11-16 on 2nd methanolysis supernatant.
Determination of PCB Concentration
1. Dilute DMSO/PCB stock (1:100, 1:1000) into 1 mL MeOH:HCl, 95%:5% solution for the determination of [PCB] in a spectrophotometer (HCl is 37.5%)
2. Aliquot DMSO/PCB stock and store, foil-wrapped, at –80º C
3. Read Absorbance at 680 nm.