Endochondral ossification
Motivation
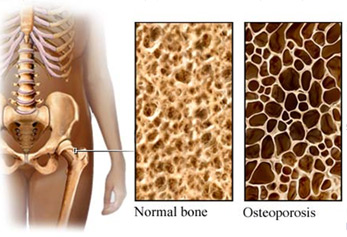
Bone regeneration is a process that is seen in normal fracture healing and remodeling throughout the adult life. Even with this capability, there are conditions where spontaneous bone healing are not ideal. Trauma, skeletal abnormalities, infection, and osteoporosis are all circumstances where current osteogenesis options will not suffice and other measures, such as bone grafting, must be utilized.
Osteoporosis is a polygenetic bone disease, which leads to fragility fractures and results in high risk of fractures in accidents and trauma. According to the International Osteoporosis Foundation, 1 in 3 women over age 50 will experience osteoporotic fractures, as will 1 in 5 men aged over 50, worldwide. Although implant design and quality has been tailored to help heal osteoporotic bone, there is a high incidence of implant failure for patients [1]. The clinical need for bone regeneration applications, systemic or in situ guided bone regeneration and bone tissue engineering continues to increase and will become a challenge for health care systems [1].
Currently, the United States, as well as other countries worldwide, is experiencing an exceedingly high demand for functional bone grafts. Annually in the United States, more than half a million patients receive bone defect repairs, with a cost greater than $2.5 billion and this figure is expected to double by 2020 [2]. Tissue engineered bone constructs have the potential to alleviate the high demand for suitable autograft and allograft materials for augmenting healing of critical-sized fracture defects [3].
Bone Formation
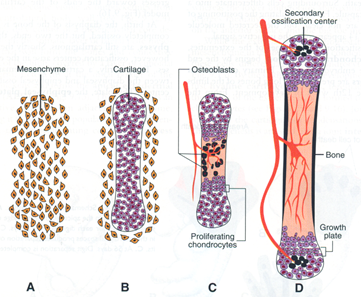
During embryonic skeletal formation, mesenchymal stem cells (MSCs) differentiate into many generative cells. One of those pathways involves MSCs differentiating into chondroblasts, which are responsible for producing the extra-cellular matrix of cartilage. The mature cells, or chondroblasts, are responsible for maintaining the extra-cellular matrix and forming a cartilage model of the skeletal elements where longitudinal growth is essential, such as the limbs, phalanges, and vertebrae. The chondroblasts grow in number and size within the center of the cartilage model. Some of the chondroblasts are caught inside their secreted ECM and become chondrocytes, which function mainly in the diffusion of nutrients and other substances and the repair of the matrix. As the cells die, the matrix is calcified and vascularized. The blood vessels deposit osteoblasts and osteoclasts which continue to replace the cartilage with bone in a process known as endochondral ossification.
When the bone is fractured, the inflammatory response begins immediately and lasts up to 7 days until the cartilage and bone formation begins. A soft callus forms and is stabilized, followed by vascularization. Mesenchymal progenitor cells proliferate and migrate through the callus, differentiating into fibroblasts or chondrocytes, each producing their characteristic extracellular matrix and slowly replacing the hematoma [5]. The fractured ends are connected with the soft callous as the intermembranous bone forms and the soft tissue within the gap undergoes endochondral ossification.
Current Options for Bone Healing and Regrowth

There are many conditions where the normal bone regeneration process is insufficient, requiring an enhanced treatment method. Standard approaches widely used in clinical practice to stimulate or augment bone regeneration include distraction osteogenesis and bone transplant, and the use of a number of different bone-grafting methods, such as autologous grafts, allografts, and bone-graft substitutes with the aid of growth factors [7].
Of the many types of grafts available to patients, each option presents biological and economic advantages and disadvantages. The most commonly used method for bone grafting is autologous, which means harvested from the patient's own body, which is advantageous due of the complete histocompatibility of tissues. However, autologous grafting has its limitations related to the harvesting process including surgical site pain and complications. Allopathic bone grafts are harvested from cadavers and account for 200,000 cases in the U.S. per year [8]. Despite the high costs and risks for viral transmission, allografts are a popular alternative to autologous bone grafts due to the lack of donor site morbidity, general success of outcomes, and decreased surgical times. Lastly, synthetic bone grafts are a more economical option without some of the transmission risks, but are limited in their biological roles. Many bone grafts are accompanied by growth factors, bone morphogenetic proteins, and hormones to assist in repair.
Ceramics and glass replacements have been widely used in the biomedical engineering and bone substitution/regeneration field. However, they are brittle and do not provide enough mechanical stability to be used for longer bones [9]. There are also two types of biodegradable polymers that are ideal for bone tissue engineering.
- Natural polymers utilize tissue sources such as collagen, fibrin, and chitosan and have a low immunogenic response as well as chemical versatility
- Synthetic polymers are more commonly used within the tissue engineering field and can be processed using multiple techniques to generate a range of three-dimensional scaffolds with different porosities and surface characteristics [10]
Developments in the fields of bone biology, materials science, and bioreactor technology will enable us to develop standard operating procedures for ex vivo tissue engineering of bone in the near future. Additionally, recent discoveries in materials science and bioreactor technology will enable researchers to tailor their work for compromised conditions like vitamin D and sex hormone deficiencies, cellular deficits, and high production of regeneration inhibitors, as they are prevalent in osteoporosis and in higher age [1].
Endochondral Ossification Tissue Engineering
The tissue-engineering approach aims to generate new, cell-driven, functional tissues. In essence, bone-tissue engineering combines progenitor cells, such as MSCs (native or expanded) or mature cells (for osteogenesis) seeded in biocompatible scaffolds and ideally in three-dimensional tissue-like structures (for osteoconduction and vascular ingrowth), with appropriate growth factors (for osteoinduction), in order to generate and maintain bone [7].
The conceptual transition from the primary target of engineering tissues to that of engineering processes recapitulating the stages of tissue development (e.g., based on self-organization of the cells, activation of specific morphogenetic pathways, and typical spatial and temporal arrangement) has the potential to instruct tissue regeneration to a higher degree of effectiveness and robustness [13]. Endochondral ossification (EO) could lead to the engineering of a fully functional “bone organ,” defined as an osseous matrix with mature vascularization and including the pivotal component of a hematopoietic BM [13]. In order to reproduce EO, there must be a hypertrophic cartilage template that would contain all necessary signals to initiate bone tissue formation, vascularization, remodeling, and establishment of a functional bone marrow.
There are currently many scaffolds that have shown the ability to support cartilage-like matrix deposition and may be suitable candidates for the development of templates for endochondral bone formation [14]. Listed below are just some of the scaffold material that is derived from humans from [Huang, et. al. 2010].
- Alginate, with or without RGD
- Chitosan
- Collagen types I and II
- Fibrin
- Hyaluronic acid
- Poly(ethylene glycol) (PEG)
- Poly(l-lactic acid) (PLLA) nanofiber scaffolds
- Silk fibroin
MSCs derived from the bone marrow have been the most frequently investigated source of progenitor cells for in vivo endochondral tissue engineering to date, in part due to their potential to differentiate into chondrocytes and subsequently progress towards a hypertrophic phenotype in vitro. [16] Plus, they are more likely to respond favorably to signals associated with tissue damage, inflammation and bone repair. Hypertrophic chondrocytes can secrete angiogenic and osteogenic factors that play pivotal roles in both the vascularization of constructs in vivo and the deposition of a mineralized extra-cellular matrix with resulting bone formation. The onset of hypertrophy has been shown to progress implant remodeling and vascularization similar to what has been observed in the growth plate and during bone repair through callus formation [13].
One of the major challenges to EO-based strategies for bone formation is identifying the optimal in vitro cultivation period and conditions prior to implantation. Despite numerous studies carried out to date, no consensus exists in terms of in vitro incubation periods in order to facilitate MSC-derived chondrocytic maturation and hypertrophy prior to implantation [14]. Therefore the cartilage maturity and hypertrophy is depends on the culture regime and incubation period. Modulation of the host's foreign body response is a challenge and if not addressed results in fibrous tissue encapsulation, preventing further growth. Through the incorporation of growth factors into the scaffold, genetic modification of cells that release increased Vascular Endothelial Growth Factor, or an anti-inflammatory coated scaffold are a few methods being researched to help with these problems [2]. The most significant problem with EO bone tissue engineering is vascularization. The greatest amount of new bone formation occurs in the most vascularized areas, whereas inadequate vascularization at bone defect sites is associated with decreased bone tissue repair and regeneration, and has been identified as the major pitfall to successful bone tissue engineering [2]. There are many factors to consider when optimizing vascularization including, scaffold design, growth factors, and pre-vascularization (either in vitro or in vivo). With all of these ideas there is still no clear answer which method will provide the best results.
Exposure to growth factors that play crucial roles in cartilage and endochondral bone formation, such as bone morphogenetic proteins, transforming and insulin-like growth factors, thyroid hormones, trans-retinoic acid, and pro-inflammatory cytokines, promote differentiation, proliferation, and hypertrophy of chondrocytes [14].
Host immunological responses are well known to play a key role in remodeling and neotissue formation following biomaterial implantation. There are two master regulators of the inflammatory process during fracture repair, IL-1β and TNF-α. Studies show that introducing IL-1β in vitro, before implantation, demonstrated a physiological response of the system to inflammatory signals and accelerated resorption of the large mass of hypertrophic cartilage [13].
The majority of studies published thus far have utilized subcutaneous models to determine the in vivo efficacy of their constructs for endochondral bone formation because offers a reproducible, easily accessible location for in vivo validation [15]. From a translational perspective, this approach may also be adopted in the clinic, that is, using an ectopic environment as an in vivo bioreactor to allow maturation of an engineered tissue, with functional vasculature and marrow components which can then be implanted into an orthotopic defect site [16].
Future of Endochondral Ossification for Bone Tissue Engineering

There is more interest than ever looking at the combination of biomaterials with cell therapy for bone tissue engineering. Potentially using mesenchymal stem cells in the regeneration of bone has been researched for the last 30 years. There have been many successes in animal models using MSCs from the bone marrow and enriched biomaterials to regerate bone tissue, but at this point most of the animal trials report ectopic applications and have not addressed the higher defect sizes and lack of vascular supply of humans.
There have been bioreactor environments created to induce the body's natural healing mechanisms to generate cartilage and new tissue. These bioreactors can provide all the necessary cells and factors in the correct temporal and biochemical sequence to support bone generation and growth with the organization and mechanical properties of native bone [10].
One example of this bioreactor system is in use by EpiBone, a biomedical engineering company in Brooklyn, NY. They take a CT scan to get the 3D structure of a patient’s bone and use a high-precision machine to carve a decellularised bovine bone into the required shape. They take fat tissue from a patient and extract stem cells from it. They combine the stem cells with the piece of carved bone and put it into a bioreactor in their lab and after three weeks in the bioreactor, they have a piece of bone ready for implantation. They are still currently doing work in pigs, but hope to start human trials by next year. By 2022, EpiBone hopes to be able to personally regrow bones from an individual's own tissues right in their lab.
References
[1] Jakoba F, Eberta R, Ignatiusc A, Matsushitad T, Watanabed T, Grolla J, Wallesa H. "Bone tissue engineering in osteoporosis." Maturitas 75 (2013) 118–124.
[2] Amini, Ami R, Laurencin, Cato T, Nukavarapu, Syam P. "Bone Tissue Engineering: Recent Advances and Challenges." Crit Rev Biomed Eng. 2012; 40(5): 363–408.
[3] Fröhlich M, Grayson W, Wan LQ, Marolt D, Drobnic M, Vunjak-Novakovic G. "Tissue Engineered Bone Grafts: Biological Requirements, Tissue Culture and Clinical Relevance." Current Stem Cell Res Ther. 2009 Nov 5. Dec; 3(4): 254–264.
[4] Healthy Bones vs. Osteoporotic Bones. Digital image. Optimorthopedics. http://www.theosteoporosisclinic.com/, n.d. Web. Jan. 2017.
[5] Sarmiento A, Latta LL. “Functional Fracture Bracing.” Berlin Heidelberg New York: Springer-Verlag.
[6] Kozhemyakina, Elena, Lassar, Andrew B., Zelzer, Elazar. "A pathway to bone: signaling molecules and transcription factors involved in chondrocyte development and maturation." Development 2015. Web. Jan. 2017.
[7] Dimitriou R, Jones E, McGonagle D, Giannoudis PV. "Bone regeneration: current concepts and future directions." BMC Medicine 2011. Web. Jan. 2017.
[8] Roberts TT, Rosenbaum AJ. "Bone grafts, bone substitutes and orthobiologics The bridge between basic science and clinical advancements in fracture healing." Organogenesis. 2012 Oct 1; 8(4): 114–124.
[9] Salgado A, Coutinho O, Reis RL. "Tissue Engineering: State of the Art and Future Trends." Macromolecular Bioscience 2004. 4(8):743-65.
[10] Stevens M. "Biomaterials for bone tissue engineering." Materials Today. Volume 11, Issue 5, May 2008, Pages 18–25.
[11] Mitra J, Tripathi G, Sharmaa A, Basu B. “Scaffolds for bone tissue engineering: role of surface patterning on osteoblast response” Materials. 2015, 8(9), 5744-5794.
[12] Sridharan R, Cameron A, Kelly D, O'Brien F. "Biomaterial based modulation of macrophage polarization: A review and suggested design principles." Materials Today. Feb 2015.
[13] Scotti Celeste, Piccinini Elia, Takizawa Hitoshi, Todorov Atanas, Bourgine Paul, Papadimitropoulos Adam, Barbero Andrea, Manz Marcus G, Martina Ivan. "Engineering of a functional bone organ through endochondral ossification." Natl Acad Sci U S A. 2013 Mar 5; 110(10): 3997–4002
[14] Scotti C, Tonnarelli B, Papadimitropoulos A, Scherberich A, Schaeren S, Schauerte A, Lopez-Rios J, Zeller R, Barbero A, Martin I. "Recapitulation of endochondral bone formation using human adult mesenchymal stem cells as a paradigm for developmental engineering." Proc Natl Acad Sci U S A. 2010 Apr 20;107(16):7251-6.
[15] Thompson E, Matsiko A, Farrell E, Kelly D, O'Brien F. "Recapitulating endochondral ossification: a promising route to in vivo bone regeneration." 11 June 2014 Journal of Tissue Engineering and Regenerative Medicine. DOI: 10.1002/term.1918.
[16] Sheehy EJ. Mesallati T, Kelly L, Vinardell T, Buckley C, Kelly DJ. "Tissue Engineering Whole Bones Through Endochondral Ossification: Regenerating the Distal Phalanx." Biores Open Access. 2015; 4(1): 229–241. doi: 10.1089/biores.2015.0014.
[17] Huang AH, Farrell MJ, Mauck RL. "Mechanics and Mechanobiology of Mesenchymal Stem Cell-Based Engineered Cartilage." J Biomech. 2011 Jan 5. 43(1): 128.
