Edwards Lifesciences: The Artificial Heart Valve, by Nicole Raia
Edwards Lifesciences is currently a leader in heart valve tissue replacement products. With their headquarters located in Irvine, California, they manufacture products that help treat over 300,000 patients per year worldwide who suffer from heart valve problems [6].
Heart Valve Background
Definition and Function
A mammalian heart contains four valves: pulmonary, aortic, tricuspid, and mitral valves. A heart valves ensures that blood flows through the heart in the correct manner, acting similar to a door. To perform this function, each valve is made up of flaps that open and close along with the rhythm of the heart. [1]
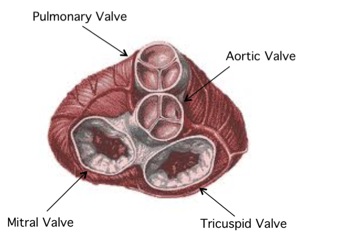
Heart Valve Problems
There are a few different problems that can affect heart valves and blood flow through the heart. Stenosis is a common heart valve condition in which the valve opening is abnormally narrow. This causes the heart to work harder due to a decrease blood flow through the valve [1]. Another condition is known as regurgitation, incompetence, or insufficiency. This condition is when a valve does not close properly possibly creating a backflow or leakage of blood [1]. Prolapse is a condition common in women in which the mitral valve does not close correctly allowing pressure to increase in the left ventricle [1]. This can eventually lead to blood leakage over time.
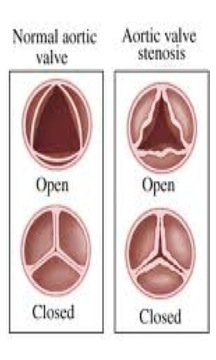
The causes to these conditions include aging, birth defects, diseases, and bacterial infections. The aging population has an increased chance of developing a heart valve conditions due to valve deterioration and the build up of calcium [1,3]. Calcification, an accumulation of calcium on the valve, mostly occurs on the aortic valve. Over time, this accumulation can cause the aortic valve to thicken, leading to stenosis. Degenerative valve disease, which is most common in the mitral valve, can lead to prolapse and cause blood leakage over time. Congenital defects that affect blood flow can also cause the above conditions. These defects may include irregular blood flow patterns, reverse blood flow direction, deformed valves, and/or partially or completely blocked valves [3]. Another common cause of heart valve conditions is rheumatic fever, an inflammatory disease. Rheumatic fever, common in children, can be caused by Streptococcus bacteria and can affect the heart and its valves [1,3]. The latest outbreak in the United States was in the 1980s but outbreaks in developing countries are still occurring [4]. Also bacterial endocarditis is known to damage heart valves leading to conditions affecting blood flow [3].
Motivation and Statistics
The need for artificial heart valves is continuously rising. Diagnostic techniques and tools are constantly improving allowing for earlier detection of abnormal valve conditions. Due to advances in society and healthcare, the average life span of the human population is increasing, which creates higher risk of age-related heart valve conditions. At this time, there are also numerous outbreaks of rheumatic fever in developing nations [6].
Cardiovascular disease is currently the leading cause of death in the United States. In 2008, it was estimated that 82,600,000 adults in the U.S. have been diagnosed with at least one cardiovascular disease. About 811,940 died in 2008 due to cardiovascular diseases [7]. It is projected that there are around 100,000 heart valve replacements per year in the US alone [8].
History of Artificial Heart Valves
The first idea of an artificial heart valve occurred in the 1950s by Campbel and Hufnagel separately. Both based their model on the ball-and-cage valve made for bottle stoppers in the 1800s. In 1952, Hufnagel implanted his valve into a human’s thoracic aorta, basically proving that artificial valve replacements were possible even though he was unsuccessful. Judson T. Chesterman was unsuccessful at implanting the first ball valve into a human’s mitral valve in 1955 when the patient died two weeks after the implantation. In 1958, Ellis and Bulbulian designed a caged-ball valve, but it was highly prone to thrombosis [9].

In 1958, Miles “Lowell” Edwards started to synthesis ideas about an artificial heart. Edwards was a retired engineer that specialized in hydraulics and pumps. Edwards, who suffered from rheumatic fever as a child, was interested in creating novel treatments for heart diseases. Edwards presented his hydraulic approach to create an artificial heart to Dr. Albert Starr at the University of Oregon Medical School. Their collaboration led to the design of the Starr-Edwards valve in 1960, which was the first caged ball valve system to ever be approved by the FDA [9].
After this, various artificial heart valves were created [9]. In the 1960s, research using tissue-based valves (either homografts or xenografts) began and evolved. Continuing with mechanical valves, the tilting disc and the hinged bi-leaflet valves were created in 1969 and 1977 respectively [11]. Currently, less invasive techniques for implantation as well as tissue-engineered valves are being designed and researched [6,8].
Types of Heart Valves and Their Associated Characteristics
In 1968, Edwards Laboratories (now Edwards Lifesciences) created the 9 commandments of prosthetics heart valves. These commandments include: embolism prevention, durability, ease of attachment, preservation of surrounding tissues, reduced turbulence, reduced blood trauma, reduced noise, use of tissue and blood compatible materials, and ease of storage and sterilization. Even with fulfilling these commandments, all artificial valves propose complications. Some common complications include hemolysis and disease associated with the implantation [11].
Mechanical
Mechanical heart valves include models such as the caged ball, the tilting disc, and the bi-leaflet [11]. Various companies, such as Metronic Inc., St. Jude Medical, and Baxter Edwards (now Edwards Lifesciences), have manufactured models of mechanical heart valves that are approved by the FDA [11]. The Starr-Edwards valve, the first successful artificial heart valve, was a caged ball valve. Today, St. Jude Medical is the global leader in mechanical heart valves. Currently, the main mitral heart valve of choice by physicians is St. Jude Medical’s leaflet valve [9].
The ideal property that makes mechanical valves so popular today is the fact that they are extremely durable. It has been proven that mechanical valves can last over numerous decades. So, mechanical valves are best for young patients who do not want to go through another operation to replace the valve in the far future [11,12].
Some unappealing complications are associated specifically with mechanical valves. The main, most detrimental complication is a high risk for thrombosis and bleeding. Due to this, a patient with a mechanical valve must undergo constant anticoagulant therapy. Another problem related to mechanical valves is their noise. They have been shown to be louder than that of tissue valves [11].
Tissue
Tissue valves can be separated into two different categories: homografts and xenografts [11]. Homograft valves are developed from human tissue while xenografts are from animal tissues. Most products and research have been localized around xenografts to avoid the lack of human donors. Currently, xenografts are either porcine (pig tissue) or bovine (cow tissue). Numerous techniques are utilized to make the tissue from these animals non-immunogenic. Treatments such as saline and alcohol solution baths, cell disruption treatments, and proteoglycan digestion allow the tissue to only contain dead cells, which makes it safer to implant into human patients [13].
Tissue valves, unlike mechanical valves, have a decreased risk of causing thrombosis and bleeding. This lifts the need for the constant anticoagulant therapy [11]. Tissue valves are also easier to implant than mechanical. Although both types of valves are functional, tissue valves look more natural due to their flexible flaps. New technology, such as techniques that involve minimal invasive surgery to implant tissue valves, is currently being developed and tested [6].
The major setback of having a tissue valve rather than a mechanical valve is durability. In contrast to the highly durable mechanical valves, tissue valves are only durable up to around 20 years [6,11]. So the probability of device malfunction increases as time goes on.
The First Successful Valve: the Starr-Edwards Heart Valve [9]
The first and only caged ball mechanical valve that was approved by the FDA is the Starr-Edwards Heart valve. Starr and Edwards were inspired to develop this successful valve by Ellis and Bulbulian’s caged ball design in 1958. In 1960, the Starr-Edwards valve, made from a methyl methacrylate (eg. Teflon) cage, a rubber ball (eg. Silicone), and a ring that was welded to the cage via acetone, was the first known successful mechanical heart valve implanted into a human patient.
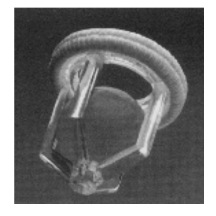
The Starr-Edwards valve was first tested in the mitral valve position of dogs. Many of these dogs died due to thrombosis. Starr and Edwards decided to cover the valve with a Silastic material to avoid this thrombosis. After the implantation of the caged ball valve with a Silastic shield, 80% of the dogs survived.
In human trials, Starr and Edwards decided to remove this Silastic shield to ease the transplantation procedure. The reasoning behind removing the shield is the fact that humans have a lower thrombosis rate and higher tolerance for surgery than do dogs. Eight patients, who all had severe progressive valvular disease caused by rheumatic fever, received the Starr-Edwards valve. Edwards mentioned that the “patients were ready to die or we weren’t allowed to work on them.” The first patient died 10 days after surgery due to an air embolism. To avoid this, the operative field was filled with carbon dioxide and also antifoaming agents were utilized. The second patient was the first success.
Between 1960 and 1998 over 175,000 Starr-Edwards valves were successfully implanted into patients.
Current Valves Manufactured by Edwards Lifesciences
Edwards Lifesciences abandoned the 1960 caged ball design and moved on to developing tissue-based valves. Currently, this corporation has over 15 different tissue valves that promote the survival of patients suffering from heart valve conditions. The following are a few selected models that I chose to focus on [6].
Carpentier-Edwards PERIMOUNT Aortic Heart Valve
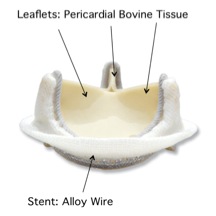
The Carpentier-Edwards PERIMOUNT valve, created in 1981, was the first biomechanically engineered aortic valve. It is made up of two components: tissue leaflets or flaps and a wire stent. The flaps are made from pericardial bovine tissue, which is the tough tissue that surrounds a cow’s heart [6]. The stent is made from an alloy wire that is flexible, resistant to corrosion, and nonmagnetic [14]. Using various techniques (ex. Neutralogic stress-free fixation, tissue thickness measurements, and tissue deflection tests), the valve can be adequately matched to the specific patient.
This valve is generally transplanted via open-heart surgery. It is durable up to 20 years and has a 17-year hemodynamic stability [6]. Like all tissue valves, this valve does not require constant anticoagulant therapy, but it is prone to short term durability and risk of stenosis and thrombosis. Today there have been over 500,000 Carpentier-Edwards PERIMOUNT aortic heart valves implanted [14].
Edwards Prima Plus
The Edwards Prima Plus is a porcine tissue valve. The unique aspect about this specific product is the fact that it is stentless. Stentless tissue valves have been proved to be more durable and have a longer hemodynamic stability than that of stented tissue valves. The valve consists of porcine aortic root preserved with a native aortic root cylinder. Edwards Lifesciences classifies this valve as a biograft, a combination of a xenograft and homograft. The root is reinforced with Darcon fabric. Along with its longer hemodynamic stability and higher durability, the stentless tissue is easier to implant due to its flexibility. This valve has risks of stenosis and thrombosis, similar to that of the Carpentier-Edwards PERIMOUNT aortic heart valve [6,15].
Edwards SAPEIN Transcatheter Heart Valve
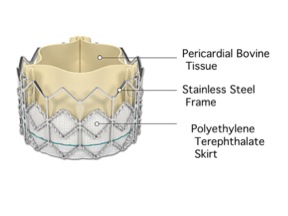
The Edwards SAPIEN is the one of the newest piece of technology developed by Edwards Lifesciences. The implantation of this valve involves minimal invasive surgery in contrast to the open-heart surgery needed in order to implant other tissue valves. This valve is ideal for patients in critical condition that would not survive open-heart surgery. It is also best for patients that suffer from severe aortic stenosis[6,17].
The valve is made up of three parts: tissue leaflets, metal frame, and a fabric skirt. The tissue leaflets are made of bovine pericardial tissue, similar to that of the Carpentier-Edwards valve model. A balloon-expandable stainless steel frame encases the leaflets. Also a polyethylene terephthalate skirt is attached for ease of implantation. Currently, Edwards Lifesciences manufactures this valve at two different sizes [6].
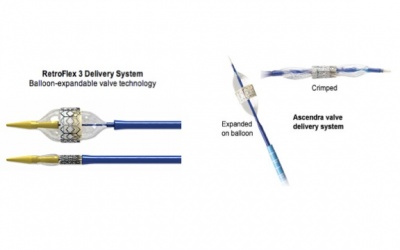
This unique valve can be inserted transfemorally (Retroflex delivery system) or transapically (Ascendra delivery system). Either way, the valve is crimped onto a catheter/balloon device. Under local anesthesia, surgeons use both echocardiographic and fluoroscopic techniques to insert the catheter correctly, which is inserted into the leg (transfemoral) or between the ribs (transpical). Once the catheter reaches the valve of choice, balloon is deployed, expanding and locking the valve in place [16].
This product became commercially available in Europe in 2007. But in the US the valve is undergoing a trial entitled PARTNER (Placement AoRTic TraNscathetER Valve Trial) [6]. This world’s first randomized clinical trial of a transcatheter heart valve started in 2007 and won’t be completed until 2014 [18].
The Edwards SAPIEN transcatheter heart valve comes with implantation-specific risks along with the general risks associated with tissue valves. There are complications with general catheterization through the cardiac system, balloon malfunction, and improperly expanded valves [6].
What About Tissue Engineering?
Tissue-engineered heart valves have been considered with the ever-growing innovative field of tissue engineering. Currently, Edwards Lifesciences devotes its resources to xenogaft/biograft tissue valves rather than tissue-engineered valves [6]. Many argue that tissue-engineered valves are not necessary due to the already successful artificial valves used today. But there is continuous research being done to create a tissue-engineered valve that can be integrated with the patients own system, leading indefinite durability [8].
References
[1] “What Is Heart Valve Surgery.” American Heart Association. 2007
[2] Image: http://www.womensheart.org/content/heartsurgery/heart_valve_replacment.asp
[3] Heart Basics: Diagnosis of heart valve disease. Webiste: http://www.yourheartvalve.com/heartbasics/pages/diseasediagnosis.aspx
[4] Rheumatic Fever. A.D.A.M. Medical Encyclopedia. PubMed. 2010. <http://www.ncbi.nlm.nih.gov/pubmedhealth/PMH0004388/>
[5] Image: http://www.health.com/health/library/mdp/0,,zm2794,00.html
[6] Edwards Lifesciences Website. <http://www.edwards.com/Pages/Default.aspx>
[7] Roger, V.L, et al. American Heart Association 2012 Update: A Report From the Heart Disease and Stroke Statistics. Circulation. 2012.
[8] Vesely, I. Heart Valve Tissue Engineering. Circulation Research (97):743-755. 2005
[9] Matthews, A.M. The Development of the Starr-Edwards Heart Valve. Texas Heart Institute Journal (25):282-293. 1998
[10] image: http://ht.edwards.com/sci/edwards/sitecollectionimages/edwards/newsroom/lowell_starr
[11] Grunkemeier, G.L, and S.H Rahimtoola. Artificial Heart Valves. Annu. Rev. Med. (41)251-263. 1990.
[12]Piazza, N. and J. Gregorie. Starr-Edwards Heart Valves. The New England Journal of Medicine (24). 2008 < http://www.nejm.org/doi/full/10.1056/NEJMicm071210>
[13] Stone, K.R. Xenograft Heart Valves. U.S. Patent 20050064591. 2005 <http://www.freepatentsonline.com/y2005/0064591.html>
[14] Frater, R.W.M., et al. The Carpentier-Edwards pericardial aortic valve: Intermediate Results. The Annals of Thoracic Surgery (53):764-771. 1992.
[15] Auriemma, S., et al. Long-Term Results of Aortic Valve Replacement with Edwards Prima Plus Stentless Bioprosthesis: Eleven Years’ Follow Up. Journal of Heart Valve Disease (15): 691-695. 2006
[16] Edwards Lifesciences: Your Heart Valve Website. <http://www.yourheartvalve.com/Pages/Default.aspx>
[17] Leon, M.B., et al. Transcatheter Aortic-Valve Implantation for Aortic Stenosis in Patients Who Cannot Undergo Surgery. The New England Journal of Medicine (363):1597-1607. 2010
[18] The PARTNER Trial: Placement of AoRTic TraNscathetER Valve Trial. ClinicalTrials.gov. 2007. <http://www.clinicaltrials.gov/ct/show/NCT00530894?order=4>
