Durotaxis and its role in directing engineering tissue development by Tyler Kimball
Durotaxis
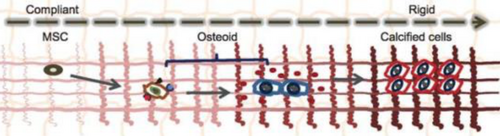
Durotaxis is the migration of cells based on the structural gradients of the rigidity of the substrate (referring to ECM, bone, or any scaffold construct)(figure1).The elastic modulus is used to measure the stiffness of the substrate which changes with each cell type.[1] The cells typically migrate in the direction of higher stiffness (figure 2).[2] The rigidity of the substrate is known to affect migration, immunology, morphology, motility, and various other processes due to the interactions between the extracellular matrix and the cytoskeleton.[1](figure 3)
The stiffness of the substrate allows cells to generate forces using focal actin-myosin complexes onto the the substrate. These forces produce focal adhesion kinases which controls the overall structure of the cytoskeleton. The cytoskeleton is highly dependent on the stiffness of the substrate which is important because the cytoskeleton determines the morphology of the cell as well as the chemical responses in the signaling pathways. Controlling the stiffness is therefore key in generating the type of cell that is needed.[3]
Figure 2 shows the elongation that is experienced by the cells when they sense stiffness
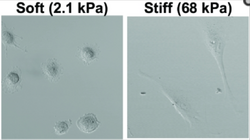
Figure 3 shows the affects of different cellular cross linkages on migration. ![Figure 3.Cellular migration using different scaffolds [3]](https://oww-files-thumb.sfo3.cdn.digitaloceanspaces.com/d/d6/Cellmigration.jpg/250px-Cellmigration.jpg.png) ]
]
History
▪1892: Ramón y Cajal predicted axons navigate in response to gradients of cues
▪1914: Migration occurs due to the substrate in the microenvironment (Harrison)
▪1934: Contact guidance – migration directed by substrate topology during embryonic development (Weiss)
▪1990's: Cytoskeletal stiffness, morphology, motility, and fibronectin fibril assembly was proven to influence migration
▪ 20th century: It was discovered that cells could detect and respond to substrate stiffness
Motivation
Understanding how to stimulate or inhibit the migration of cells is crucial for cellular regeneration and treatment. The medical field is driving the need for this research to potentially solve problems like host rejection makes durotaxis in engineering a hot topic.
Bone Scaffolds
For years, doctors have been using allograft bone transplants and other replacement methods that are cost inefficient, cause inflammation, extensive pain, and are prone to rejection or replacement after so many years of use. Recent studies in the mechanical properties of bone have produced a bone scaffold that has similar rigidity and porosity to real bone and would allow for the integration of cells completely in less than four weeks. This is possible because the scaffold mimics the stiffness of the mesenchymal stem cells. As said before, the mimicry of the rigidity of the ECM allows for the bound stem cells to create focal adhesion forces which give the the cytoskeletal organization. The organization of the of the cytoskeleton is what gives the cells their shape and thus function.[4][5][6]
Durotaxis in Cancer
Cancer cells use durotaxis extensively affecting the lives of more than 200,000 people every year. One of the key properties that enable cancer to succeed is its stiffness. Tumors can be found to be as much as 10 times stiffer than the normal breast tissue this dramatic gradient causes the pull of endothelial cells to its mass allowing for tumor growth and for metastasis. [4] By finding ways to counteract these properties doctors could slow down the progression of cancer and make treatment more successful.
Conclusions
Tissue engineering and regenerative medicine have recently been found to be greatly affected by the durotaxic properties of the ECM. Engineers have been able to use the information that they have recently discovered to make huge advances in cancer research and bone replacement with more improvements guaranteed to come. Hopefully scientists will be able to answer more questions using the research they find with gradient hydrogels and experimentation.
References
[1] Ulrich, Theresa A., Elena M. de Juan Pardo, and Sanjay Kumar. “The Mechanical Rigidity of the Extracellular Matrix Regulates the Structure, Motility, and Proliferation of Glioma Cells.” Cancer research 69.10 (2009): 4167–4174.
[2]Friedl, Peter, and Bettina Weigelin. "Interstitial leukocyte migration and immune function." Nature immunology 9.9 (2008): 960-969.
[3]Sant, Shilpa et al. “BIOMIMETIC GRADIENT HYDROGELS FOR TISSUE ENGINEERING.” The Canadian journal of chemical engineering 88.6 (2010): 899–911.
[4]Breuls, Roel G.M, Timothy U Jiya, and Theo H Smit. “Scaffold Stiffness Influences Cell Behavior: Opportunities for Skeletal Tissue Engineering.” The Open Orthopaedics Journal 2 (2008): 103–109.
[5]Quintero, Andres J, et al. “Stem Cells for the Treatment of Skeletal Muscle Injury.” Clinics in sports medicine 28.1 (2009): 1–11. PMC. Web. 21 Mar. 2017.
[6]Isenberg, Brett C. et al. “Vascular Smooth Muscle Cell Durotaxis Depends on Substrate Stiffness Gradient Strength.” Biophysical Journal 97.5 (2009): 1313–1322. PMC.
[7]Wang, N; Butler, JP; Ingber, DE (21 May 1993). "Mechanotransduction across the cell surface and through the cytoskeleton.". Science. 260 (5111): 1124–7.
[8] Halliday, NL; Tomasek, JJ (March 1995). "Mechanical properties of the extracellular matrix influence fibronectin fibril assembly in vitro.". Experimental Cell Research. 217 (1): 109–17.
[9]DiMilla, Paul A., et al. "Maximal migration of human smooth muscle cells on fibronectin and type IV collagen occurs at an intermediate attachment strength." The Journal of cell biology 122.3 (1993): 729-737.
[10] Plotnikov, SV; Pasapera, AM; Sabass, B; Waterman, CM (21 December 2012). "Force fluctuations within focal adhesions mediate ECM-rigidity sensing to guide directed cell migration.". Cell. 151 (7): 1513–27.