Distillation - Brandon Johnston
Background and Motivation

Distillation columns use heat as an energy separating agent to purify components of a mixture based on differences in boiling points. Distillation is the most widely used separation process used industrially, where the energy from such separations accounts for 8% of total energy consumption in the U.S. [2] At the macro-scale, the process allows for the more volatile component, or components, to rise to the top of the column as a vapor as the less volatile component, or components, falls to the bottom of the column as a liquid. Mixture containing components with a larger relative volatility will result in a better separation. Additionally, the components travel through trays in the column. Columns contain multiple trays, which contain both liquid and vapor, and assist in obtaining a better separation if compared to a single stage distillation; the greater number of trays, the more pure the components will be post-separation. Each tray has a unique temperature and composition. Reboilers and condensers are also used to reinsert liquid and vapor, respectively, back into the column. The more volatile component is removed from the column as the distillate while the less volatile component is removed as bottoms. Distillation is used in both industrial and academic settings. [3]
Theoretical Analysis
Governing Equations
There are a number of important equations necessary to adequately model a distillation separation. First, the equilibrium between components and phases must be established. The equilibrium model can come from an equation of state, such as Van der Waals, Redlich-Kwong, or Peng-Robinson. For the simplest type of distillation, a binary distillation, the feed is composed of a more volatile component, the light key (LK), and a less volatile component, the heavy key (HK). Ultimately, the LK will be the distillate product and the HK will the bottoms product. Using Raoult's Law along with the assumptions that the binary mixture forms an ideal solution and follows the Ideal Gas Law, the following equation can be applied to model the equilibrium: [3]
In the above equation, α is the relative volatility between the components while x and y are the liquid and vapor phase, respectively, of component one. From here, mass balances can be applied to the column with a number of boundaries, including the entire column, the top half of the column, and the bottom half of the column. A mass balance of the entire column results in what is called the q-line, which describes the feed entering the column: [3]
In the q-line, q is equal to the the ratio of change in molar reflux rate across the feed to the feed and [math]\displaystyle{ z_F }[/math] is the mole fraction in the feed. A mass balance around the top half of the column results in the rectifying line: [3]
In the rectifying line, R is the reflux ratio, or the ratio of material being sent back into the column to the material being collected from the condenser. [math]\displaystyle{ x_D }[/math] here is the mole fraction of the light key in the distillate product. A mass balance around the bottom half of the column results in the stripping line: [3]
Here, [math]\displaystyle{ V_B }[/math] is the boil-up ratio, or the ratio of material being sent back into the column from the reboiler to the material being collected as bottoms product. [math]\displaystyle{ x_B }[/math] is the mole fraction of the light key in the bottoms product. If all of the equations are plotted on the same axes, where y is plotted as a function of x, along with a 45 degree angle line from (0,0) to (1,1), a McCabe-Thiele Diagram can be made.
McCabe-Thiele Diagram
A McCabe-Thiele diagram depicts all of the governing equations on the same plot in order to model the distillation column at hand performing a specific separation with specific parameters. [4] A researcher can easily perform the analysis necessary to build a McCabe-Thiele diagram, with the desired product molar ratios, and determine how many trays or stages are necessary in the column in order to achieve the desired separation. Once constructed, a McCabe-Thiele Diagram looks like the following:
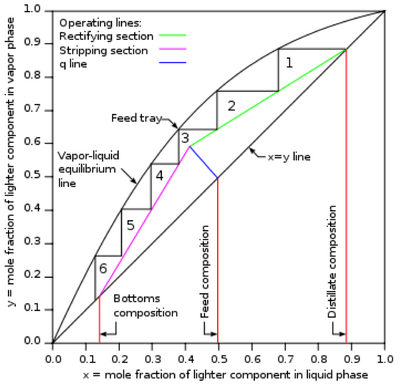
A number of things can be derived from a McCabe-Thiele diagram. As stated prior, the number of theoretical stages needed to achieve the desired separation can be derived from the number of "steps" in the diagram. Each step corresponds to a theoretical stage. The optimal feed tray or location can also be determined from where the q-line intersects the 45 degree line. As depicted in the example diagram, some separations require very few stages. Others, however, require hundreds. An extremely rough estimate of capital costs can be inferred from the determined number of theoretical stages.
Other types of distillation
Issues with distillation arise when the feed mixture becomes more complicated, such as non-ideal equilibria, multi-component, or azeotropes, which is when the mixture has a minimum or maximum boiling point. Other problems occur when the boiling point of the components is too high for feasible energy inputs. In order to overcome such limitations, a number of non-simple distillation techniques have been developed: [3]
- Pressure swing distillation - when simple distillation columns are used in series at different pressures. The altered pressures change the azeotropic temperature which allows for adequate separation.
- Extractive distillation - when another material is added to the solution that has a higher affinity to mix with one component over the other, resulting in the elimination of the azeotropic limitation.
- Reactive distillation - when a certain reaction is occurring in the mixture as it is being fed into the column with the product being the light key. This results in the distillation of the product occurring as it is being formed.
- Vacuum distillation - when distillation is occurring at very low temperatures, allowing for the boiling point of the components to be decreased. This allows for the energy put into heating the column to be less.
Uses
The uses of distillation can be boiled down into two categories: industrial, or large-scale settings, and lab-scale, or small-scale settings. Industrially, distillation is often used for the purification of chemicals used in everyday life. The petroleum industry uses distillation to separate crude oil into its components, such as diesel gasoline, kerosene, and other hydrocarbons. Distillation is also used in water purification processes, such as the desalinization of water or the demineralization of water. Another high-profit industry that centers around distillation is the alcohol industry. Many alcoholic beverages are distilled from the grans and fruits in which they are derived from. Additionally, argon is distilled from air in order to use it as an inert gas or to be used to protect the filament in light bulbs. Distillation is used in many other industrial applications where the difference in boiling points between two components in a mixture can be exploited.
On a smaller scale, in a laboratory setting, distillation has a number of useful applications, often for purification purposes. For example, distillation is used to remove water from solvents in order to obtain anhydrous solvents for reactions that are extremely sensitive to minute amounts of water. Distillation is also used to "flame dry" glassware, which results in ultra-dry glassware. The technique is also used to purify reagents and products that may be in a mixture with other compounds. Additionally, distillation is used to remove solvents from compounds in order to obtain the compounds in pure form.
Application in Microfluidics
Distillation at the macro-scale is well studied, optimized, and extensively used in both industry and lab-scale settings. In general, separation at the micro-scale introduces a number of benefits: decreased separation times, increased performance, decreased costs, decreased use of reagents, and increased control over fluidic parameters. [7] However, in contrast to other separation techniques at the micro-scale, distillation poses a number of logistical issues when scaled down to the micro-scale. [8][9] For example, the vapor/liquid equilibrium nature of distillation is difficult to control and manipulate at the micro-scale, where capillary forces dominate over gravitational forces, a crucial component of macro-scale distillation. [10] Researchers have overcome these limitations by using inert carrier gases to guide the flow of vapor or using microfluidic channels lined with micropillars to guide the flow vapor and liquid. Using these techniques, researchers have been able to develop microfluidic distillation techniques that reap the benefits of scaling down.
Multistage Distillation
In order to create a microstage distillation apparatus on a microfluidic chip, researchers at University College London used a single, curved channel in order to facilitate separation between liquid and vapor. [10] The group heated one end and cooled the other in order to achieve the temperature gradient necessary for separation. Initially, issues occurred where the mass balance of the entire system was off: the output flow rates of the chip were not high enough to account for the solution entering the chip. This imbalance resulted in flooding of the system and was attributed to a lack of flow of both liquid and vapor within the system. The lack of flow was a direct result of the capillary forces dominating over the gravitational forces: the liquid was more prone to attaching to the walls of the device than it was to fall due to gravity. The difference in dominate forces between the macro-scale (gravitational forces) and the micro-scale (capillary forces) ultimately resulted in clogging of the microfluidic channels, which the group alleviated by incorporating rows of micropillars into the main channel. The group offered an explanation for the micropillars that insisted the pressure drop due to the pillars is what allowed the liquid to flow adequately. This pressure drop overcame the increased pressure due to the capillary forces. The flowing liquid is what ultimately eliminated the flooding. Of all the configurations investigated, serpentine channels with a diameter of 600 microns and a length of 40 centimeters. When this configuration was heating from 42 degrees Celsius to 95 degrees Celsius and a 50 mole percent solution of acetone in water was introduced in the middle of the channel, the distillate product had a purity of 95 mole percent acetone and the bottoms had a purity of 3 mole percent acetone, corresponding to a distillation column with 4 stages. In conclusion, the group was able to demonstrate a reproducible, multistage distillation process on a microfluidic chip that resulted in a separation comparable to a macro-scale distillation.
Microfluidic Distillation for Methanol Detection
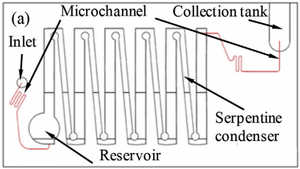

Coupling separation techniques with the micro-scale of microfluidics allows for separation and detection of components with extremely low amounts. A group from the National Pingtung University of Science and Technology created a microfluidic device that used distillation to detect the concentration in methanol in a methanol-ethanol-water mixture, such as spirits. [11] The group injected a mixture into a reservoir and then used a nitrogen carrier gas to continuously move the mixture through boiling and heating chambers until the mixture reached a collection tank containing DI water, where the mixture would cool down. The inert carrier gas is crucial to the microfluidic distillation, as the pressure increase from the capillary forces would be too great for flow otherwise. Without the carrier gas moving the mixture, it would clog the microfluidic channel and separation would not occur. The boiling chambers were at the bottom of a serpentine condenser at 95 degrees Celsius and the heating chambers were at the top of the same serpentine condenser at 80 degrees Celsius. The solution would be boiling up by the boiling chambers and then condensed by the heating chambers. Water and ethanol, with boiling points at higher temperatures than methanol, would condense and fall back into the boiling chamber while the methanol would continue through to the collection tank. From the collection tank, the mixture would be reacted with a mixed indicator and the concentration of methanol would be determined using a spectrophotometer. The system was used to analyze spirits and, if coupled with a microfluidic spectrophotometer, could easily be translated to point-of-care applications. This system demonstrated an average distillation efficiency of 97.9% with methanol concentrations in the range of 300-800 ppm.
Other Applications in Microfluidics
Other groups have created microfluidic distillation devices for the detection of a specific compound. Researchers at National Cheng Kung University created a device to separate sulfuric acid into sulfur dioxide and water in order to detect sulfur dioxide. [12] Researchers at the Massachusetts Institute of Technology used microfluidic distillation to complete a multi-step triflate synthesis. [13] The distillation step was used to switch the dichloromethane solvent to either dimethylformamide or toluene. Additionally, researchers at Kawamura Institute of Chemical Research devised a microfluidic device that could carry out vacuum membrane distillation. [14]
References
- Naik, S.; Schornak, B.; Seadeek, C.; Wesorick, S.; Robertson, M.; Nalbandian, A.; Cotton, S.; and Hoffman, N. Distillation Columns. Visual Encyclopedia of Chemical Engineering. http://encyclopedia.che.engin.umich.edu/Pages/SeparationsChemical/DistillationColumns/DistillationColumns.html (accessed Feb 23, 2018).
- Sholl, D.S.; and Lively, R.P. Seven chemical separations to change the world. Nature, Comment. 2016, 532(7600), 435-437.
- Henley, E. J.; Seader, J. D.; Roper, D. K. Separation Process Principles, 3rd ed.; Wiley: Hoboken, NJ, 2011.
- McCabe, W. L.; Thiele, E. W. Graphical Design of Fractionating Columns. Industrial and Engineering Chemistry. 1925, 17, 605-611. (http://dx.doi.org/10.1021/ie50186a023).
- Eden, M. R. CHEN 4470 – Process Design Practice. Presented at Auburn University, Auburn, Alabama, February 26, 2013.
- Sylvan, R. What Are the Uses of Distillation in Industry? Bizfluent. https://bizfluent.com/about-5941994-uses-distillation-industry-.html (accessed Feb 23, 2018)
- Streets, A.M.; Huang, Y. Chip in a lab: Microfluidics for next generation life science research. Biomicrofluidics. 2013, 7(1), 011302. (http://dx.doi.org/10.1063/1.4789751)
- Krizek, T.; Kubickova, A. Microscale separation methods for enzyme kinetics assays. Anal. Bioanal. Chem. 2012, 403, 2185-2195. (http://dx.doi.org/10.1007/s00216-012-5744-x)
- Khandurina, J.; Guttman, A. Microscale separation and analysis. Curr. Op. Chem. Bio. 2003, 7, 595-602. (http://dx.doi.org/10.1016/j.cbpa.2003.07.001)
- Lam, K.F.; Cao, E.; Sorensen, E.; and Gavriilidis, A. Development of multistage distillation in a microfluidic chip. Lab on a Chip. 2011, 11, 1311-1317. (http://dx.doi.org/10.1039/C0LC00428F).
- Wang, Y.-N.; Liu, C.-C.; Yang, R.-J.; Ju. W.-J.; and Fu, L.-M. Microfluidic distillation chip for methanol concentration detection. Analytica Chimica Acta. 2016, 912, 97-104. (http://dx.doi.org/10.1016/j.aca.2016.01.047).
- Ju, W.J.; Fu, L.M.; Yang, R.J.; and Lee, C.L. Distillation and detection of SO2 using a microfluidic chip. Lab Chip. 2012, 12(3), 622-626. (http://dx.doi.org/10.1039/c1lc20954j).
- Hartman, R.L.; Naber, J.R.; Buchwald, S.L.; and Jensen, K.F. Multistep Microchemical Synthesis Enabled by Microfluidic Distillation. Microreactor Networks. 2010, 49, 899-903. (http://dx.doi.org/10.1002/anie.200904634).
- Zhang, Y.; Kato, S.; and Anazawa, T. Vacuum membrane distillation on a microfluidic chip. Chemical Communications. 2009, 19, 2750-2752. (http://dx.doi.org/10.1039/B823499J).
