Degradable Biomaterials for Cell Delivery by Timothy Sullivan
Introduction to Biomaterials
Biomaterials are materials intended to interact with biological systems for the evaluation, treatment, augmentation or replacement of tissues, organs or function of the body [1]. Biomaterials can be used in a wide-range of applications including wound healing as well as drug and cell delivery. The main type of biomaterials are polymeric, meaning that they are composed of either natural or synthetic polymers [2]. Polymers are macromolecules consisting of repeating subunits, the “mers.” They can be used for a wide variety of applications, from consumer goods to precise medical instruments.
Synthetic vs Natural Biomaterials
Popular natural polymers used in tissue engineering include alginate, fibrin, and collagen. These natural polymers have been in use for thousands of years; it was not until the last few decades that the use of synthetic polymers in tissue engineering has become feasible [3]. A few synthetic polymers commonly used in tissue engineering include polylysine (PLA), polyglycolide (PGA), their co-copolymer poly(lactic-co-glycolic acid) (PLGA), and poly(ethylene glycol) (PEG).
Natural and synthetic polymers each have strength and weakness. Natural polymers are by nature bioactive and area able to be remodeled easily in the body. However, they often cause immunogenic responses in the body and the purification involved in processing natural polymers to avoid disease transmission can be complex. Synthetic polymers are biologically inert and can be tailored to have more predictable properties. Since synthetic polymers are chemically synthesized, properties such as crystallinity, molecular weight, and mechanical properties can be controlled. The downsides include lacking cell adhesion sites without modification and potentially harmful products as a result of degradation.
Degradable Biomaterials
An important property of biomaterials is their biodegradability. Biodegradation is the process of a material’s breakdown meditated by a biological system [1]. In certain applications, a non-biodegradable material is preferable. This includes structural implants (such as a titanium hip replacement) or fake tooth. Other applications, including cell delivery, require that a material breakdown over time. This biodegradation can follow multiple routes and is dependent on the structure and chemical make-up of the specific material chosen. Degradation of the cell-seeded carrier allows for the controlled release of cells to a specific target. In some applications, a degrading scaffold allows for ingrowth of the surrounding tissue and shaping of tissues.
Example Structure of Biomaterials
Biomaterial Scaffolds
Each of the two specific cell carriers discussed in this article are scaffolds. The scaffolds are temporary 3-D structures that are meant to degrade over time and serve as support. Several properties are especially important for cell delivery applications. Biocompatibility describes how well a scaffold performs its application without releasing by-products that cause an immune response upon degradation. Biodegradability, as mentioned above, is an important quality. A requisite porosity is required, which allow for interaction with cells. Inadequate porosity can lead to poor in-growth and poor transport qualities [4].
Electrospun Fiber Mats
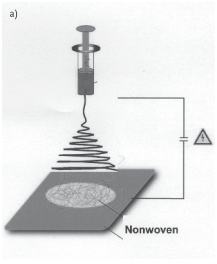
Electrospinning is a production technique used to produce small diameter fibers of a dissolved substance. Many electrospinning applications, including the two discussed in this article, use polymers as their material. Electrospinning fiber production is based on electrical forces rather than mechanical forces. A charged fluid jet is attracted to a substrate (sometimes oppositely charged), and the polymer fibers are pulled from the solution as the solvent evaporates [5].
An amount of polymer is dissolved in a solvent then put into a metal-tipped syringe. A voltage is applied to the syringe tip to create the charge difference. A weight presses down the shaft of the syringe at a specified rate, ejecting the polymer from the syringe tip. This ejected solution is attracted to a substrate (such as aluminum foil) and the polymer fibers begin to accumulate as the solvent evaporates out. An example set-up is shown in Figure 1 [5].
Electrospinning can produce fibers with different diameters depending on polymer used, concentration, voltage, and other parameters. The mats produced can have varying fiber density and porosity.
PLGA Biomaterials in Cell Delivery
PLGA Properties
PLGA Structure
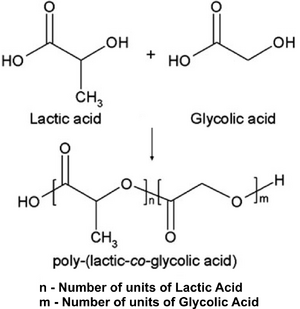
PLGA is the co-polymer made from lactic acid and glycolic acid monomers. Figure 2 shows the structure of PLGA and its component monomers [6]. The number of units with-in the polymer can be varied. PLGA is biocompatible and biodegradable; both naturally occurring monomers are minimally toxic. Physical properties such as degradation time and mechanical strength can be controlled by varying the ratio of the monomers in the polymer structure; increasing the lactic acid portion increases degradation time due to hydrophobicity and increasing the glycolic acid portion increases mechanical strength. Percent crystallinity is a function of PGA content [7].
PLGA Degradation
PLGA degrades through hydrolysis of its ester bonds. Water molecules attack the bond that links the lactic acid and glycolic acid portions of the polymer chains. The degradation is two step: the first is a random cleavage of ester bonds and the second step is significant mass loss. The first step begins to break up the chain into smaller portions; the chain loses mechanical stability and molecular weight drops. The second step occurs when the broken chain fragments are smaller in size. These individual portions that made up the polymer begin to dissolve and diffuse out of the matrix [8].
The degradation of the polymer occurs readily in the human body due to the abundance of water. Degradation time is controlled by varying the ratio of the monomers; lactic acid is hydrophobic which reduces the amount of water molecules interacting with the chain. The final forms of the degraded polymer are eventually reduced to CO2 and H20 by the Krebs cycle [9].
Injectable and Porous PLGA Microspheres to Form Highly Porous Scaffolds
Research Motivation
Injectable scaffolds are promising options in treating a variety of problems, due to the homogeneous mixing of scaffold and cells as well as the ability to inject the scaffold into cavities of any shape and size. The benefits of using porous microspheres include easy diffusion of nutrients and oxygen. Prior to research conducted by O. Qutachi et al. [10], porous microspheres were unable to fuse into a more organized structure at body temperature. Such a fusion would allow for the desired porosity characteristics from the microspheres while also providing the mechanical integrity and bulk properties of the scaffolds.
Methods

PLGA microspheres were produced by a mixture of PLGA 50:50 (equal mass of each monomer) dissolved in dichloromethane by the double emulsion method; first in phosphate buffered saline and then in 0.3% polyvinyl alcohol then washed with distilled water. The microspheres were mixed manually with RFP-NIH-3T3 fibroblasts (red fluorescent protein labelled 3T3 mouse fibroblasts) in solution to create a “paste”; this was transferred to a mould and kept at 37°C in a cell culture medium 24 hours. Upon removal from the oven and mould after 24 hours, the microspheres were fused together to create a scaffold structure as seen in Figure 3. The average percent porosity of the structure was 81.54% [10].
Results
An important component of this study was the amount of cells observed within and on the scaffold structure. This was quantified by fluorescence microscopy images of the RFP-NIH-3T3. Over time, the number of viable cells attached to and contained within the scaffold increased;
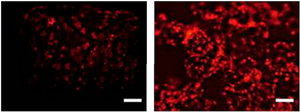
this indicated that the cells successfully proliferated. This can be seen in Figure 4 and Figure 5. Figure 4 shows the scaffold 14 days after seeding. The left image displays the scaffold as a whole and the right image displays a close-up image of the surfaces. It can be seen that cells have successfully infiltrated the scaffold and appear on the surface of the microspheres. Figure 5 shows two micrographs of the scaffolds 21 days post-seeding. A monolayer of cells covers the scaffold [10].

The researchers noted that cell adhesion has been problematic with PLGA scaffolds in the past; they were able to successfully adhere cells because of the surface treatment they applied during production of the microspheres. While specific injection solvents were not discussed, the researchers mentioned that tetraglycol has been used in a past application. The degradation of the spheres was not directly quantified in this study. However, since it is composed of PLGA it will degrade over time when injected into the body due to hydrolysis of its ester bonds. Future work will focus on studying the specific release properties of the scaffold [10].
PEG Biomaterials in Cell Delivery
Spinal Cord Injury Facts
According to the National Spinal Cord Injury Statistical Center, there are over 17,000 spinal cord injuries (SCIs) each year and up 347,000 people living in the United States with SCIs. These numbers are potentially deflated because they do not consider injuries that resulted in an individual who died at the scene of their accidents. Dying from a severe SCI is common as it often results in paraplegia of some form. Since 2010, 55% of patients treated for SCIs experienced some form of paraplegia (paralysis of lower body) or tetraplegia (paralysis in each of their limbs) [11].
These injuries often occur as a result of falls and car crashes; these types of injuries are not caused just by violent collisions in sports or reckless lifestyles. The average age of spinal cord injuries keeps reducing; from the 1970’s to today the average age has increaed from 29 years old to 42 years old. As people live longer, more-active lifestyles, they are more susceptible to SCIs. Longer lifespans mean that people will be living with their injuries for significantly longer periods of time and may require long-term care. Hospital stays average around 11 days for SCIs, with rehabilitation lasting more than a month. Only around 1% of patients discharged from the hospital completely recover by the time that they leave meaning that intensive rehabilitation and personalized care is often necessary [11].
The cost for treating SCIs can be enormous. The lifetime treatment costs of a severe tetraplegic injured at 25 years old is $4,729,788, at over $185,111 per year. Paraplegia patients injured at 25 years old often have lifetime costs north of $2,300,000 [11].
Life expectancy drops significantly as a result of severe SCIs. For example, a paraplegic injured at 40 years old has life expectancy drop by 32% and a severe tetraplegic injury at 40 years old results in a 50% drop in expected years. SCIs leave patients most susceptible to death by pneumonia and septicemia, but reduced physical exertion also leads to musculoskeletal diseases and increased rates of heart disease [11].
PEG Properties
PEG is a crystalline polymer with many applications in tissue engineering. PEG is a highly hydrophilic polymer, and when attached to more hydrophobic polymers it can increase their solubility. PEG is biocompatible, non-biodegradable, non-immunogenic, and resistant to protein absorption [12].
PEG Structure and Synthesis
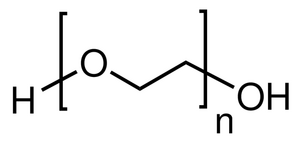
PEG is synthesized by the polymerization of either ethylene glycol or ethylene oxide. The length and molecular weight of the synthesized chains can be controlled by terminating the polymerization reaction. The general structure of a polyethylene molecule is shown in Figure 6 [13]. PEG comes in multiple configurations, including a branched, linear, and comb structure [12].
PEG Degradation
PEG is not biodegradable. For many tissue engineering applications where biodegradability is required, PEG must be co-polymerized or combined with a biodegradable polymer.
Spinal Cord Regeneration using Electrospun PLGA-PEG Scaffolds
Research Motivation
Current treatment techniques for spinal cord injuries are lacking. There is no current treatment to restore a paraplegic’s or tetraplegic’s mobility to conditions prior to the injuries. The longer an individual survives after an injury, they are more likely to experience some of the associate degenerative diseases (such as infect bed sores). As evidence of the ineffectiveness of current treatment techniques, it is estimated that 30% of individuals with SCIs are re-hospitalized one or more times in each year after their injuries. SCIs oftentimes lead to traumatic injuries that impair an individual for the many years, requiring constant and expensive care [11].
SCIs are not always severe as the spinal cord transection conducted in this research. However, when a severe SCI occurs and nerves are irreparably damaged, there are no comprehensive treatments that can restore their function. The technique developed in this study, if it is able to eventually be used to treat human SCIs, could revolution how severe SCIs are treated. If the results can be translated to human injuries the number of individuals experiencing limited and complete paralysis should drop. Patient life expectancies should rise and injury costs should decrease, especially when factoring in how much the current treatments cost each year [11].
Methods
One promising route in repairing severe SCI wounds is the regeneration of neurons by neural stem cells (NSCs). Two processes were used by Liu et. al [l] to procure NSCs: 1) a certain number of NSCs were isolated from the embryo directly (wt-NSC, wild-type neural stem cells) and, 2) mouse embryonic fibroblasts (MEFs) were extracted to create induced neural stem cells (iNSCs) [14].
MEFs were isolated from 14-day old CD-1 mouse embryos after being washed in PBS three times. The MEFs were cultured in induction medium, formed into neurospheres, and transduced with GFP retrovirus for 24 hours. The iNSCs were seeded onto glass coverslips with a medium containing growth factors and supplements initially, then changed to NSC differentiation medium and the cells were allowed to differentiate. The cells were incubated with several antibodies overnight (including Nestin, MAP2, GFAP, MBP, and Alexa-labeled antibodies) for immunofluorescence analysis. Hoechst 33342 was used to detect nuclei, and micrographs were taken with a Leica fluorescence microscope [14].
The researchers prepared scaffolds by electrospinning. Two configurations of PLGA (75:25) and PEG (at 15 wt% and 1.5 wt% PLGA:PEG), as well as 100% PLGA, were dissolved in hexafluoroisopranol and placed in syringes. A 15 kV voltage was applied, and the collector plate was 12 cm from the syringe to collect the mats [14].
Cell adhesion and proliferation within the scaffolds were measured after iNSCs were seeded onto the scaffolds. PLGA-PEG film and gelatin sponge were used to create the 3-D scaffolds. The scaffolds and gels were rolled into a cylinder, soaked in medium, and had culture medium containing cells injected inside. These were incubated and placed in culture medium [14].
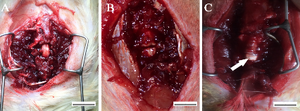
To diagnose the repair ability of the PLGA-PEG scaffolds, adult female mice were split into three groups of ten (ten each for PLGA scaffolds, PLGA-PEG scaffolds, and controls) and had their spinal cords transected. Scaffolds 2.0 mm in length were transplanted into the gap (shown in Figure 7). To determine SCI recovery, a scale from 0-21 was used (with 21 being normal limb movement) [14].
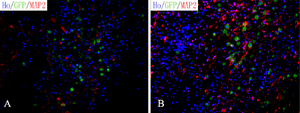
As part of this study, researchers wanted to see how iNSCs differentiated in vitro. By studying the expressed NSC markers, differentiated products could be determined. Three separate differentiation pathways were observed: neurons, astrocytes (non-neuronal cells the serve several purposes), and oligodendrocytes (non-neuronal cells that create myelin sheath). The researchers found that the iNSCs differentiated in the following percentages: 42.36% neurons, 37.92% astrocytes, and 8.88% oligodendrocytes. The WT-NSCs differentiated in the following percentages: 39% neurons, 36% astrocytes, and 9.90% oligodendrocytes. Cell adhesion on the PLGA-PEG scaffolds was significantly higher than on the PLGA scaffolds, and iNSC content was higher within [14].
Results
Cavity Filling
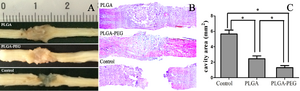
After two weeks, the implanted scaffolds had almost filled the cavity and had integrated with the host tissue. By eight weeks post-operation, the PLGA-PEG scaffolds had significantly better cavity repair than PLGA scaffolds only; this comparison along with control is shown in Figure 8. Cavity area was significantly reduced from the control group with both scaffolds, but the PLGA-PEG scaffold was superior [14].
Movement Recovery
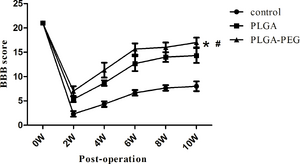
Analysis of the SCI recovery scale indicated that at all instances PLGA-PEG was superior to the PLGA scaffolds and control specimens. The “BBB score” comparison versus time is shown in Figure 9. Rats are able to do some healing reflexively of their spine [14].
Cell Proliferation and Imaging
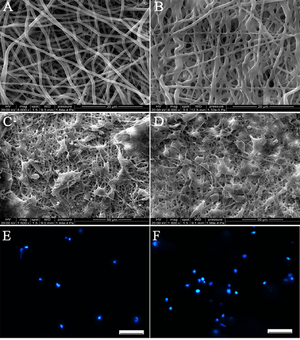
Figure 10 shows SEM images (labeled A-D) and nuclei Hoechst staining images (E,F). The different morphologies of the initial electrospun mat are shown in Image A (PLGA) and Image B (PLGA-PEG); the neural cell growth can also be seen in Image C (PLGA) and Image D (PLGA-PEG). Image E (PLGA) and Image F (PLGA-PEG) show a region Hoechst stained for nuclei detection; the PLGA-PEG mat has almost twice as much nuclei as the PLGA mat. The researchers observed the survival and differentiation of iNSCs in the PLGA-PEG graft and surrounding areas (shown in Figure 11) two weeks after implantation. Image A focuses on iNSCs (stained green), Image B focuses on Tuj1-positive neurons, and Image C focuses on GFAP-positive glial cells. In all images, implanted cells migrated from the graft site into the surrounding tissues. Figure 12 shows the same areas after 8 weeks and includes a histogram displaying the amount of present in the area. PLGA-PEG scaffolds performed significantly better than PLGA-only scaffolds at delivering higher quantities of neural cells when SCIs occur [14].
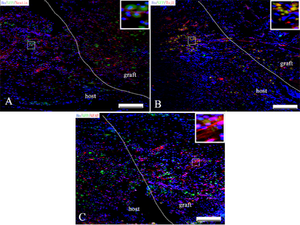
Conclusions
The PLGA-PEG scaffold studied by the researchers successfully delivered cells to the site of a SCI and helped expedite the healing process, ultimately leading to a more successful repair than a control group and specimens that were treated using PLGA-only scaffolds. The use of iNSCs proved to successfully heal severe SCIs within eight weeks. They also have the added benefits of causing less teratomas and are free from the ethical controversies that other stem cell treatments have. Inclusion of PEG into the scaffold led to drastic improvements in all metrics. Addition of the PEG made the scaffolds more hydrophilic, with increased cell adhesion and proliferation.
This research did have some flaws that the researchers hope to improve in future studies. Induction of the iNSCs needs to be improved to higher percentages – more induction should lead to more stem cells that can heal the wound and more efficient treatments. The researchers rely on a retrovirus to trigger reprogramming of the cells, which could lead to an infection of other health related issue. Surgery can always have complications, especially when working on areas close to nerves. There is the danger of a potential surgery going wrong and potentially making the problem worse. If a patient's immune system is in some way weakened (whether it be from natural causes or a side-effect of medication), there may be an increased chance of infection. And there is always the small possibility that a patient may be alergic to a specific polymer or its degradation by-products which may cause further health complications.
Moving forward, the researchers hope to scale-up this treatment to larger and more complex animals. The ultimate goal is to provide a technique to completely repair severe SCIs in humans.
History
Much of the research done on degradable biomaterials for cell delivery applications has been conducted in the last 30 years. According to Sakiyama-Elbert et al., the definition for "biomaterial" did not encompass tissue engineering scaffolds until 1987.
Since then, there has been research related to this application [15].
Relevant projects:
- 1993: Porous ceraminc vehicles for rat-marrow-derived osteogenic cell delivery created.
-1996: IKVAV peptide sequence containing scaffolds promoted neurite growth.
- 2001: Cartilage engineered by mesenchymal stem cell seeding inside of fibrin clots.
- 2002: PLGA scaffolds designed to mimic the spinal cord and seeded with murine neural stem cells increased recovery after spinal cord injury.
- 2003: Embryonic stem cells could differentiated inside of collage scaffolds.
- 2003: PLGA scaffolds can direct stem cell differentiation to neurons.
- 2004: Collagen scaffolds seeded with neural stem cells.
- 2005: Cartilage formation observed in rabbit knee defects from PLGA scaffolds.
- 2005: Effect of incorporating fibronectin and laminin into collagen scaffold on mouse embryonic stem cells tested.
- 2005: Injectable system of PLGA microspheres developed.
- 2005: Retinal progenitor cells seeded in PLGA scaffolds were effectively delivered in vivo.
- 2006: Hepatocytes generate from human embryonic stem cells in collagen scaffolds.
- 2006: Mechanical properties of synthetic polymer can influence stem cell differentiation.
References
[1]- Williams, D. The Williams Dictionary of Biomaterials. Liverpool, England, United Kingdom: Liverpool University Press, 1999.
[2]- Jiag, X. et al. Biodegradable synthetic polymers: Preparation, functionalization and biomedical application. Progress in Polymer Science 2012, 37, 237-280.
[3] – Nair, L.; Laurencin, C. Biodegradable polymers as biomaterials. Progress in Polymer Science 2007, 32, 762-798.
[4] – Lyons, F.; Partap, S.; O’Brien, FJ. Part 1: Scaffolds and Surfaces. Technol Health Care 2008, 16, 305-317.
[5] – Wendorff, J.; Agarwal, S.; Greiner, A. Electrospinning: Materials, Processing, and Applications. Wiley, 2012.
[6] Hatton, P. et al.; An Overview of Poly(lactic-co-glycolic) Acid (PLGA)-Based Biomaterials for Bone Tissue Engineering. International Journal of Molecular Sciences 2014, 15(3), 3640-3659.
[7] – PolySciTech. PLGA. http://polyscitech.com/PLGA/PLGA.php (accessed 02/28, 2017).
[8] – Avgoustakis, K. Polylactic-Co-Glycolic Acid (PLGA), Enclyclopedia of Biomaterials and Biomedical Engineering 2005, 1-11.
[9] - Tsuji, H. et al. Poly(Lactic Acid): Synthesis, Structures, Properties, Processing, and Applications. Wiley, 2010.
[10] – Rahman, C. Injectable and porous PLGA microspheres that form highly porous scaffolds at body temperature, Acta Biomaterial 2014, 10, 5090-5098.
[11] – National Spinal Cord Injury Statistical Center, Spinal Cord Injury (SCI) Facts and Figures at a Glance, 2016.
[12] – Schubert, U.S.PEG Review Poly(ethylene glycol) in Drug Delivery: Pros and Cons as Well as Potential Alternatives, Angewandte Chemie International Edition 2010, 49 (36), 6288-6308.
[13] – MP Biomedicals, Polyethylene Glycol (MW 1450).http://www.mpbio.com/product.php?pid =02199045 (accessed 03/01, 2017).
[14] – Liu, B. et al., Tissue-Engineered Regeneration of Completely Transected Spinal Cord Using Induced Neural Stem Cells and Gelatin-Electrospun Poly (Lactide-Co-Glycolide)/Polyethylene Glycol Scaffolds, PLOS One, 2015.
[15] - Willerth, S.M.; Sakiyama-Elbert, S.E. Combining stem cells and biomaterial scaffolds for constructing tissues and cell delivery, StemBook, ed., 2008.