Cord Blood Banking by Tiankai Zhang
Introduction
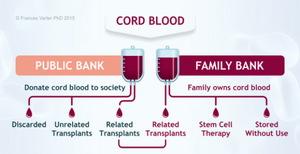
Cord blood banking is a service which basically offers science technology to help people preserve their cord blood. The cord blood today people are concerning about are mostly referring to the umbilical cord blood (UCB) which is a major source of primitive hematopoietic stem cells (HSC) and progenitor cells (HPC) that could be used for the treatment of certain diseases such as hematological malignancies, marrow transplantation, immunodeficiencies, hemoglobinopathies and inherited metabolic.[1][2] Currently, there are two major types of cord blood bank which are public and private. For the public bank, UCB of newborn infants could be donated to the bank by the consent of parents without any payment since the ownership of the UCB will belong to the bank after donation. And the inventory is available to the public and healthcare institution for them to look for partnership. However, for the private bank, the collection of UCB under parents' specific will and requires certain cost. The UCB in private bank is not accessible to public and will only for an autologous use such as the transplantation therapy of biological sibling.[3][4] Until 2016, there are 207 private cord blood banks over 54 countries and 158 international public cord blood banks in 36 countries with over 731000 units of umbilical cord blood.[4]
Processing
Collection Procedure
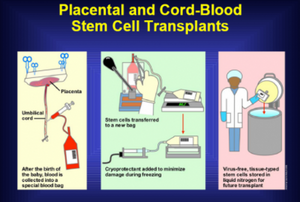
Before the collection is conducted, certain examinations should be performed on the donor infants' mother to exclude possible blood infectious diseases such as HIV and hepatitis. Also, if disability or hereditary diseases history are discovered for donors fetus, the collection could no be performed. In principle, the process of UCB collection must not hinder the delivery of baby. Between the time period of infant birth and placenta delivery, umbilical cord need to be clamped and sterilized to avoid the contamination by maternal blood or colon-derived fluids during during the vaginal birth due to the close contact of external side of umbilical cord and vaginal. Then the blood inside the umbilical cord will be led into a container with sterile and anti-coagulated condition by the method of venipuncture sterilely. This 5 minutes collection process seems simple to conduct, however, it requires more attention than people imagine. First, it is really easy to fail to lead blood out of umbilical cord according to Burton's theory.[6] The theory states that even more than 1 minute constriction such as clamping applied to the umbilical cord arteries after infants birth may results in vascular occlusion and finally lead to the failure of UBC extraction. Moreover, UBC contaminations are mostly occurred during this period so all the procedure must to be performed under sterile conditions.[1][3]
Storage
After collection, the collected UCB need to be measured for its physical characteristic including mass and volume. Additionally, mono-nuclear cells (MNC), total nucleated cells (TNC), and CD34+ cells are also measured since they are the significant parameters which are indicating the number of the stem cells.[1] After measuring all the significant parameters and before cryopreservation, red blood cell (RBC) depletion and volume reduction procedure are conducted in order to deplete most RBCs and increase the viability of stem cells because RBCs take more than half volume of the collection.
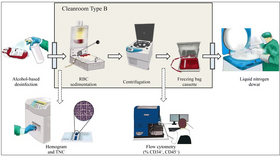
To implement this RBC separation, multiple methods could be applied including density gradient separation, sedimentation, rouleaux formation and centrifugation.[8] After separations, then UBC will be frozen by two optional methods which are red cell reduction (RCR) and plasma depletion (PD). For RCR, centrifugation will be used to isolate cord blood with most white blood cells.[3] Then 50% dimethyl sulfoxide (DMSO) will be added into the isolated cord blood to form the final cell suspension to be frozen. In the PD method, only plasma will be removed and rest part of the cord blood is mixed with 10% DMSO to be frozen. For both of the two methods, DMSO is used to prevent ice crystal formation inside the cells. However, since DMSO is toxic to the blood cells with more than 1% concentration at 37 degree celsius and more than more than 30 minutes time period, it need to be removed once the UBC is thawed.[1][3][9]
Clinical Therapy
Before the transplantation of the UBC, the recipient should be human leukocyte antigen (HLA) matched, even for the biological siblings donation. For the six haplo-identical HLA match tests, satisfy three of the six is the lowest limit for a transplantation whereas match all the six haplo-identical HLA is the ideal expectation. For the siblings, there are 1/4 chances to match six of six and 3/4 chances to match at least three of six.[1]
Pediatric patients
With the first UCB transplantation successfully applied on the five years old patient with Fanconi anemia in France, UCB transplantation was initially applied on pediatric patients. According to the reports, the pediatric patients with malignancies such as leukemia and multiple myeloma who accept the related transplantation have a recovery rate of 90% and graft-versus-host disease (GVHD) of10%. The 5-year disease-free survival (DFS) is 44%.[8] As for the unrelated transplantation, a research of 500 children with acute leukemia transplanted with cord blood compared with 300 children transplanted with unrelated donors shows that there is no significant difference of leukemia-free survival with one or two mismatch HLA in unrelated transplantation. However, for the nonmalignant disease such as Hurler's syndrome, a research of 300 children with Hurler's syndrome shows that the disease-free survival rate is 81% for 6 of 6 HLA match with unrelated transplantation but it decreases to 68% when 5 of 6 HLA match.[1][8][9]
Adult patients
After the promising results of UBC transplantation emerged in pediatric patient, it is then led to adult patients therapy. But the initial research shows that the adult patients with hematologic malignancies have only a 26% disease-free after cord blood transplantation. The reason for low disease-free survival rate for adult is because the relatively larger size of adult requires more hematopoietic stem cells than children. To overcome this problem, double cord blood transplantation (DCBT) is used and the DFS rate of hematologic malignancies increases to 76%. However, due to the increment of the cord blood transplantation, the non-relapse rate increase to 34% compared to 20% for single cord blood transplantation. To solve the high non-relapse rate, reduced intensity conditioning (RIC) has been used such as cyclophosphamide. As the result, a significant decreasing of the non-relapse rate has been observed.[1][8][9][13]
Future
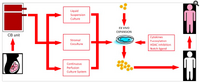
Due to the wide application of double cord blood transplantation, a greater incidence of GVHD has been observed. To overcome this problem, ex vivo expanded T regulatory cells which is a subset of CD4+ T cells may be used. As a novel method, besides been used to decrease the risk of GVHD, the expansion techniques is also promising for future UBC transplantation by combining the mesenchymal stem cell co-culture with notch ligand to achieve a relatively faster engraftment of cells during a short time period.[9]
Reference
[1] Butler, M. G., & Menitove, J. E. (2011). Umbilical cord blood banking: an update. Journal of Assisted Reproduction and Genetics, 28(8), 669–676. http://doi.org/10.1007/s10815-011-9577-x
[2] Kurtzberg, J. (2009). UPDATE ON UMBILICAL CORD BLOOD TRANSPLANTATION. Current Opinion in Pediatrics, 21(1), 22–29.https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3682484/
[3] Roura, S., Pujal, J.-M., Gálvez-Montón, C., & Bayes-Genis, A. (2015). The role and potential of umbilical cord blood in an era of new therapies: a review. Stem Cell Research & Therapy, 6(1), 123. http://doi.org/10.1186/s13287-015-0113-2
[4] Peberdy, L., Young, J., & Kearney, L. (2016). Health care professionals’ knowledge, attitudes and practices relating to umbilical cord blood banking and donation: an integrative review. BMC Pregnancy and Childbirth, 16, 81. http://doi.org/10.1186/s12884-016-0863-6
[5] Parent's Guide. How Banks Work. https://parentsguidecordblood.org/en/how (accessed May 5, 2017)
[6] Yao, A. C., Lind, J., & Lu, T. (1977). Closure of the human umbilical artery: a physiological demonstration of Burton's theory. European Journal of Obstetrics & Gynecology and Reproductive Biology, 7(6), 365-368. https://doi.org/10.1016/0028-2243(77)90064-8
[7] Byeong Keun Ha. Stem cells Transplant. http://pathology.medicine.iu.edu/research/research-projects/current-research/byeong-keun-ha-md-phd/ (accessed May 5, 2017)
[8] Ballen, K. K., Gluckman, E., & Broxmeyer, H. E. (2013). Umbilical cord blood transplantation: the first 25 years and beyond. Blood, 122(4), 491–498. http://doi.org/10.1182/blood-2013-02-453175
[9] Munoz, J., Shah, N., Rezvani, K., Hosing, C., Bollard, C. M., Oran, B., … Shpall, E. J. (2014). Concise Review: Umbilical Cord Blood Transplantation: Past, Present, and Future. Stem Cells Translational Medicine, 3(12), 1435–1443. http://doi.org/10.5966/sctm.2014-0151
[10] ResearchGate. The role and potential of umbilical cord blood in an era of new therapies: A review. https://www.researchgate.net/figure/279535172_fig2_Figure-2-Umbilical-cord-blood-collecting-processing-and-banking-basic-steps-In (accessed May 5, 2017)
[11] Page, K. M., Mendizabal, A., Betz-Stablein, B., Wease, S., Shoulars, K., Gentry, T., … Kurtzberg, J. (2014). Optimizing Donor Selection for Public Cord Blood Banking: Influence of Maternal, Infant and Collection Characteristics on Cord Blood Unit Quality. Transfusion, 54(2), 340–352. http://doi.org/10.1111/trf.12257
[12] BartsMS Blog. CrowdSpeak: the Russian experience of HSCT in MS. http://multiple-sclerosis-research.blogspot.com/2015/03/crowdspeak-russian-experience-of-hsct.html (accessed May 5, 2017)
[13] Seo, S. H., Shin, S., Roh, E. Y., Song, E. Y., Oh, S., Kim, B. J., & Yoon, J. H. (2017). Long-Term Quality Control Program Plan for Cord Blood Banks in Korea: A Pilot Study for Cryopreservation Stability. Annals of Laboratory Medicine, 37(2), 124–128. http://doi.org/10.3343/alm.2017.37.2.124
[14] Munoz, J., Shah, N., Rezvani, K., Hosing, C., Bollard, C. M., Oran, B., … Shpall, E. J. (2014). Concise Review: Umbilical Cord Blood Transplantation: Past, Present, and Future. Stem Cells Translational Medicine, 3(12), 1435–1443. http://doi.org/10.5966/sctm.2014-0151 (accessed May 5, 2017)
