CH391L/S2013 Alesha Stewart Mar 27 2013
Structure of a DNA glycosylase searching for lesions- Banerjee A, Santos WL, Verdine GL
Background & Introduction
DNA glycosylases are proteins responsible for DNA damage recognition and repair [4]. These enzymes search entire genomes for single nucleobase lesions to initiate base excision repair via Brownian motion. The ability of DNA glycosylase to seek and find damaged bases is a wonder of science, especially since there are only minor differences between impaired and unimpaired bases. Thermo energy fuels this efficient process, as no biochemical forms of energy are expended. Details concerning the function and execution of DNA glycosylase are still under investigation, but the article “The Structure of a DNA Glycosylase Searching for Lesions” intends to shed light on the subject [1].
In this study, the authors utilized a bacterial DNA glycosylase (Bacillus stearothermophilus MutM) that specifically associates with 8-oxoguanine (oxoG). OxoG is a form of DNA damage from reactive oxygen species, where guanine is oxidized. As a result, the lesion can cause mismatched base pairing or mutations leading to genome instability and cancer if the damage goes unrepaired [2]. There is an oxoG resistance pathway in bacteria to protect against such damage (the “GO” system). MutM is the component of the system responsible for excising oxoG:C base pairs [3].
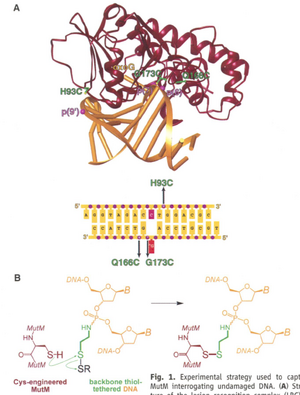
When MutM binds to an oxoG DNA base it represents the lesion recognition complex (LRC), which was used to investigate the recognition and repair method Fig 1A. Images of the LRC indicated that MutM physically flips the oxidized nucleobase out from the DNA helix to position it into its active site Fig 1A. Extrahelical base excision is now believed to be a universal technique used for all DNA glycosylases [1].
The mechanism of how extrahelical base excision is executed, however, is still undetermined. There are three proposals previously considered to explain how DNA glycosylases search, find, and repair damaged nucleobases- 1) It actively extrudes every base from the DNA; 2) It detects damaged bases that were spontaneously extruded from the DNA; or 3) It identifies lesions and activates the extrusion from the DNA. The researchers focused on the latter option, intrahelical lesion recognition, as it is the most kinetically favorable [1].
Methods
DNA and MutM Synthesis
Mutant MutM proteins were synthesized from B. stearothermophilus MutM gene with point mutations and megaprimer mutagenesis. The DNA oligomers with and without lesions were synthesized using a DNA synthesizer. Phosphoramidate- cystamine backbone modifications were also made for DXL with the cysteine-engineered MutM.
Crosslink formation
Different length carbon linkers and phosphate positions on the DNA backbone were investigated for DXL of MutM. Two, three, and four carbon chain thiols were tethered to the phosphates to serve as the linker to attach the cys-engineered protein during incubation (2:1 protein to DNA concentration). Non-reducing SDS-PAGE confirmed the crosslinking quality fig S1, and the product was purified on a monoQ column and concentrated to 250 μM.
Crystallization
X-ray crystallography was used to determine the crystal structures of the protein-DNA complexes. X-rays were generated using a synchrotron.
OxoG containing complexes were crystallized using the 1:1 hanging drop vapor diffusion method (13−18% (w/v) PEG 8K, 100 mM sodium cacodylate, pH 7.5, 50 mM Mg(CH3COO)2 and 0.1 % βME reservoir solution), cryoprotected with glycerol, and preserved in liquid nitrogen for data collection. There was initial model for refinement from the isomorphous structure of the LRC of MutM. Then the model was built with continuous rounds of rigid body refinement, energy minimization, simulated annealing and grouped B-factor refinement.
Crosslinked, undamaged complexes were formed using the 1:1 hanging drop vapor diffusion method at 4°C (12-18% PEG 8K, 100mM sodium cacodylate pH 7.0 and 0-5% glycerol reservoir solution). These crystals were also cryoprotected with glycerol and frozen in liquid nitrogen for data collection. Crystal structures were solved with molecular replacement, which requires a model of the structure in advance to serve as the initial search model to conduct the translation and cross rotation search. The models were built as previously stated.
Results
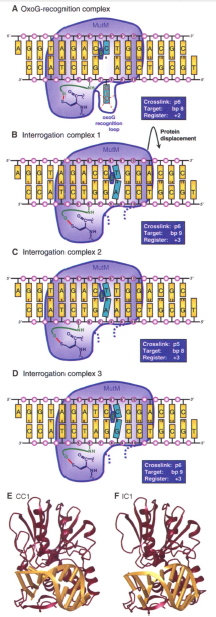
Intermolecular disulfide cross-linking (DXL) of the LRC was used to develop structures of MutM in the process of examining undamaged DNA (G:C base pair). The crosslinks were used to hold the complexes in position for crystallization and detection. The best positions and linkers for the DNA-MutM crosslinks were determined. The Q166C mutation with a two-carbon tether on phosphate 6 (Q166C/p6’) produced the fastest DXL at the purest and highest yield to form Interrogation complex 1 (IC1) fig S1. The IC1 crystal structure was solved with 2.0 Å resolution.
IC1 was intended to interrogate a G:C base pair at the same position of the oxoG:C base pair in the LRC. When the LRC crosslink position on the DNA backbone was phosphate 6 (p6) it targeted base pair 8 (the oxoG:C bp) Fig 2A. However, the crystal structures of IC1 determined that with the crosslink position at p6, it targeted bp 9 (an A:T bp) Fig 2B. Here the DXL between the protein and DNA allowed MutM to shift itself further down the DNA than in the LRC.
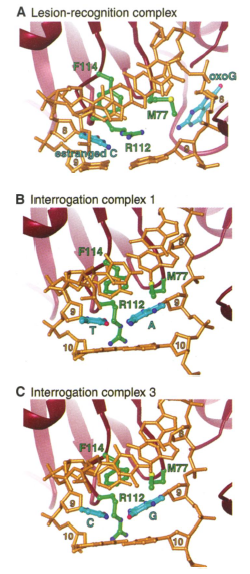
Structures of the LRC revealed the damaged oxoG base inverted from the DNA helix and positioned in the extrahelical MutM active site, causing the DNA to bend 80 degrees. Three main protein residues then fill the void created from this inversion. The Phe114 residue inserts itself into the helical stack, while the Arg112 and Met77 residues enter the vacant space and form hydrogen bonds and van der Waals interactions with the DNA Fig 3A. These protein-DNA interactions are different in the interrogation complexes, because here the target base pairs are still intact and intrahelical. Although the Phe114 residue still inserts the helix and buckles the target base pair, Arg112 and Met77 no longer enter the helix and interrelate with the DNA. This weakens the contact between the DNA-protein complex, and allows for the shifting we see in the interrogation complexes.
Since the IC1 was interrogating the wrong bp, the proper crosslink position and register to target the intended G:C bp was investigated. Adjusting the crosslink still resulted in the complex targeting an A:T bp, which lead the researchers to believe there was a MutM preference for A:T. The bps were switched in IC1 to create G:C bp at the 8, 9 and 10 positions (IC3) to force MutM to target the intended G:C bp Fig 2D. Each of the interrogation complexes had similar structures and properties (including the DNA bending) to the LRC. These findings suggested the interrogation complexes were an "early intermediate" formed to extrude base pairs and insert them into the MutM active site.
Another protein-DNA interaction that was different with the interrogation complexes, was their contact with ordered water molecules Fig 4. The interaction with the water molecules were thought to serve as a lubricant to aid the protein sliding up and down the DNA helix. Lastly, it was determined that DNA glycosylase MutM utilizes its Phe114 residue to serve as a probe to search for and detect lesions while they are still within the DNA helix, since it inserts itself into damaged and undamaged DNA.
Conclusion & Significance
MutM was used to learn more about how DNA glycosylases function. The course of recognition and repair was explored by using DXL to capture crystal structures of the enzyme during the interrogation process. The structures indicated that MutM utilizes a probe to pinpoint the oxoG lesions via intrahelical lesion recognition. Upon recognition, the damaged bp is extruded from the helix and the extrahelical base excision repair process begins. As MutM interrogates the DNA its selection process is key, however; it is more kinetically favorable for it to extrude oxoG bases than undamaged bases [1].
References
- Banerjee A, Santos WL, Verdine GL. Structure of a DNA Glycosylase Searching for Lesions. Science 311, 1153-7 (2006).
- Fromme, JC and Verdine, GL. Structural insights into lesion recognition and repair by the bacterial 8-oxoguanine DNA glycosylase MutM. Nature Structural Biology 9, 544-552 (2002).
- Fromme, JC and Verdine, GL. DNA Lesion Recognition by the Bacterial Repair Enzyme MutM*. J. Biol. Chem. 278, 5543-51548.
- Fromme JC, Banerjee A, Verdine GL. DNA glycosylase recognition and catalysis. Curr Opin Struct Biol. 14, 43-9 (2004).
- Sheila S. David, Valerie L. O'Shea & Sucharita Kundu. Base-excision repair of oxidative DNA damage. Nature 447, 941-950 (2007).
Note
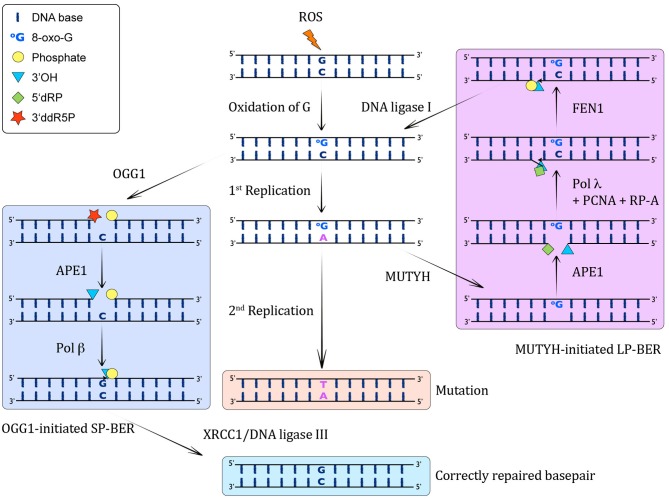
Information on how MutY repairs the mismatched oxoG:A base pair.
Right column: The A:8-oxo-G base pairs can be recognized by MUTYH, which catalyzes the excision of the wrong A from opposite 8-oxo-G, leading to the formation of an AP site. This AP site is further processed by APE1, which results in a 1 nt gap with 3′OH and 5′dRP moieties. The incorporation of the correct C opposite 8-oxo-G and one more nucleotide is performed by pol λ in collaboration with the cofactors PCNA and RP-A, thus performing strand displacement of the downstream DNA strand. FEN1 cleaves the 5′ flap, leading to a 5′P moiety, which can be ligated by DNA ligase I to yield an intact C:8-oxo-G containing double-stranded DNA. This C:8-oxo-G is then again substrate for OGG1-mediated removal of 8-oxo-G (left column).
Reference: Enni Markkanen, Julia Dorn, and Ulrich Hübscher. MUTYH DNA glycosylase: the rationale for removing undamaged bases from the DNA. Front Genet. 2013; 4: 18.