Buddy Ratner by Brandon Johnston
Introduction

Early Life
Buddy Ratner was born on January 19, 1947 in Brooklyn, New York. Born into a lower middle class family, Ratner immediately gained a passion for science, especially chemistry. He attended Wingate High School in Brooklyn, New York, where his love for science further blossomed under the leadership of his science teachers. It was in high school that his interest in polymers began to take flight. He enrolled in undergraduate studies at Brooklyn College, later becoming part of the City University of New York. As an undergraduate, he worked in a stockroom under the supervision Dr. Milton J. Rosen, who Ratner convinced to give a lecture about polymers to fellow students in an advanced course. This lecture further convinced him that polymers would be an area of interest for him. He graduated from Brooklyn College in 1967 with a bachelor of science in chemistry and immediately began graduate school at the Polytechnic Institute of Brooklyn, where his passion for polymer research thrived.[1]
Graduate Studies
At the time of Ratner’s enrollment, the Polytechnic Institute of Brooklyn had one of the only well structured polymer science programs in the country. Under the supervision of Dr. Dan Bradley, he began studying membrane transport via the use of hydrogels, as well the physical chemistry side of polymers. Two years into the project, Dr. Bradley succumbed to a stroke, and Ratner finished his projects with Dr. Irv Miller, the Chairman of Chemical Engineering at the institute. In 1970, Ratner attended a lecture from Dr. Allan Hoffman, a professor at the Massachusetts Institute of Technology. Hoffman was studying polymer hydrogels in technology and Ratner’s interest in Hoffman was immediately sparked. In 1972, Ratner received his PhD in Polymer Chemistry with thesis research on a hydrogel membrane for an artificial kidney. He was trying to understand how the hydrogel could be used as a blood filter. As soon as Ratner received his PhD, he moved to Seattle to work with Hoffman as a post-doc in his new home: the University of Washington.[1]
Post-Graduate Research
His early research at the University of Washington involved work funded by the Atomic Energy Commission. The project had two parts: to create new materials through radiation-grafted polymers, and to graft polymer hydrogels to elastomers to be used in a plutonium 238-powered heart replacement. Hoffman’s group used gamma radiation from a cobalt 60 reactor in order to graft one polymer to another, or, potentially, a hydrogel to a surface. The goal of the research was to create a material that could be used to implant a plutonium 238 nuclear power source, with a core temperature of 1400 degrees Celsius and a surface temperature of 40 degrees Celsius, into the stomach of a human. Unfortunately, the project failed due to the adverse effects of radiation. After completing his post-doc work in 1975, he became a Research Assistant Professor at the University of Washington, where he has spent all his time during his long and fruitful carrier.[1]
Honors and Affiliations
Affiliations[2]
Current
Professor of Chemical Engineering and BioEngineering (1975)
Michael L. & Myrna Darland Endowed Chair in Technology Commercialization
Director of University of Washington Engineered Biomaterials (UWEB) (1996)
Elected Member of the National Academy of Engineering, USA (2002)
Editor of the Journal of Undergraduate Research in BioEngineering
Advisory Board Member of Biointerphases
Past
Past President of Society for Biomaterials (1991-1992)
Past Director of the National Institutes of Health-funded National Electron Spectroscopy for Chemical Analysis and Surface Analysis Center for Biomedical Problems (1985-1996)
Past President of the American Institute for Medical and Biological Engineering (AIMBE) (2002-2003)
Elected President of the Tissue Engineering Society of North America (TESNA) (2003)
Served on National Advisory Council of the National Institute of Bioimaging and Bioengineering, NIH (2009-2013)
Past Vice President of the Tissue Engineering Society International (TESI) (2003-2005)
Fellowships[2]
American Institute of Medical and Biological Engineering (AIMBE)
American Vacuum Society (AVS)
American Association for the Advancement of Science
Biomedical Engineering Society (BMES)
American Chemical Society (ACS)
International College of Fellows Biomaterials Science and Engineering
Polymer Division of American Chemical Society
Tissue Engineering and Regenerative Medicine International Society (FTERM)
Society for Biomaterials
Awards[3]
2016 Kammermeyer Lecturer, University of Iowa
2015 Distinguished Service Award, ACS Division of Polymer Chemistry
2015 Langmuir Lecture, American Chemical Society (COLL Division)
2015 Most Cited Paper Award, Annals of Biomedical Engineering
2014 University of Washington School of Medicine 2014 Lifetime Innovator and Inventor Award
2011-2012 AVS Biointerphases Lectureship
2010-2012 UW Entrepreneurial Faculty Fellow
2012 George Winter Award, European Society for Biomaterials
2012 Honorary Professor of Sichuan University
2012 Journal of Materials Science-Materials in Medicine “Best Paper Published in 2011”
2011 Pierre Galletti Award of the American Institute of Medical & Biological Engineering
2010 McGowan Distinguished Lecturer, University of Pittsburgh
2010 Annual Faculty Lecturer, University of Washington
2009 Chandra P. Sharma Award, Society for Biomaterials & Artificial Organs (India)
2009 Acta Biomaterialia Gold Medal Award
2008 BMES Pritzker Distinguished Lecturer Award
2008 J. Edward Berk Lecture Medal
2008 Listed in AIChE’s “One Hundred Chemical Engineers of the Modern Era”
2008 Frontiers of Science Award, Society of Cosmetic Chemists
2008 Kewaunee Lecturer, Duke University
2007 Bayer Lectureship, University of Akron
2006 C. William Hall Award, Society for Biomaterials
2004 Founders Award, Society for Biomaterials
2004 Distinguished Lecturer, University of Utah
2002 Elected to the National Academy of Engineering of the United States of America
2002 Medard W. Welch Award, American Vacuum Society
2002 Chair, Roundtable on Biomedical Engineering Materials and Applications (BEMA)
2000 Science In Medicine Lecturer, University of Washington
2000 Joe Smith Distinguished Lecturer, University of California, Davis
2004-2005 Robert F. Rushmer Professor of Bioengineering, University of Washington
2001-2004 Washington Research Foundation Endowed Professor of Bioengineering
1999 American Vacuum Society Distinguished Lecturer
1998 C.M.A. Stine Award for Materials Science, AIChE
1996 Van Ness Lecturer, Rensselear Polytechnic Institute
1995 Chair, Gordon Research Conference on Biocompatibility & Biomaterials, July 23-28
1991 Perkin Elmer Physical Electronics Award for Excellence in Surface Science
1990 Burlington Resources Foundation Faculty Achievement Award for Outstanding Research
1988 Clemson Award for Contributions to the Literature
Extras[4]
Buddy Ratner has authored over 400 peer-reviewed articles and over 20 patents. He is the lead editor for Biomaterials Science: An Introduction to Materials in Medicine. Ratner has helped launch eight companies based on his research and is a consultant for a number of other companies.
Current Research[5]
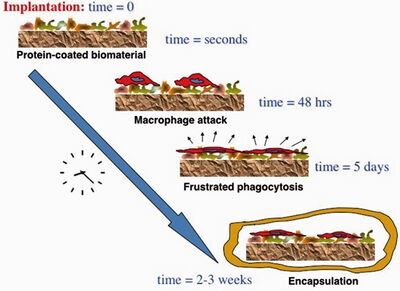
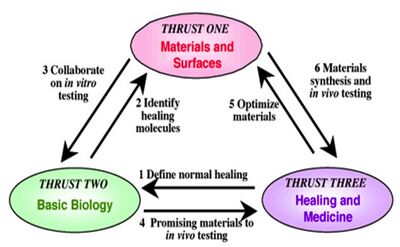
Dr. Ratner currently has two primary research and education programs: the University of Washington Engineered Biomaterials (UWEB), a National Science Foundation (NSF) Engineering Research Center, and BioEngineered Autologous Tissue (BEAT), an NIH Bioengineering Research Partnership grant. Dr. Ratner’s research is focused on creating and implementing an advances group of biomaterials. A major issue with the biomaterials of today is that they create what is known as a “foreign body response”. This occurs when macrophages, fibroblasts, and other cells and proteins attack an implant and form a collagenous pocket to cut off the device from the rest of the body. This “pocket” is called a foreign body capsule, and it successfully and dramatically lowers the devices ability to do its job. Whereas most implanted medical devices trigger this “foreign body response”, Ratner’s group aims to, instead, create “biomaterials that heal”. This phrase encompasses a variety of tasks, including the exploitation of macrophages and the triggering of specific reactions while simultaneously repressing other reactions. The research of UWEB is broken up into three “thrusts”:
- The first thrust, titled “Materials and Surfaces”, aims to provide the designs of biomaterial surfaces and coatings, as well as provide the strategies to control biology at the surface of the biomaterial. This thrust’s main objective is to be able to create materials that do not elicit the foreign body response, such as inflammation.
- The second thrust, titled “Basic Biology”, deals with creating biomaterials that are integrated into the body as if they are of the same organism, far unlike today’s biomaterials. This thrust works very closely with the discovery and implementation of biomolecules as well as the exploration of biomaterials on animal models. The main objective is to create biomaterials that not only allow for implantation into animals by bypassing the foreign body response, but also are accepted and integrated into the body.
- The third thrust, titled “Healing and Medicine”, moves beyond the synthesis of biomaterials and works to study the results when these advanced biomaterials are incorporated into animal models in vivo and human models in vitro via engineered organoids.
The third thrust uses biological principles that were identified by the second thrust to advance the biomaterials that were designed in the third thrust. The three thrusts form a tightly associated network that results in the design and implementation of an advanced class of biomaterials that heal.
Selected Publications
Material Structure
Design of infection-resistant antibiotic-releasing polymers: I. Fabrication and formulation and II. Controlled release of antibiotics through a plasma-deposited thin film barrier (1999)[6][7]
One of the many problems that arise from implanted biomaterials is the causing of infections. These infections occur when bacteria stick to the surface of the implant and form a biofilm, resulting in what is known as a device-associated infection. In 1999, Ratner and his group created an antibacterial device via a ciprofloxacin-loaded polyurethane matrix. This matrix effectively releases antibiotics at the device surface, lowering the level of bacteria and warding off possible infection. They studied a variety of combinations of pore-forming agents and drug loadings, resulting in poly(ethylene glycol) (PEG) as the optimal pore-forming agent and a one to one ratio of PEG to ciprofloxacin as the optimal loading cocktail. These conditions resulted in the device being able to resist Pseudomonas aeruginosa accumulation, a pneumonia causing bacteria, for up to five days. However, he also concluded that the first order rate of release and the initial “burst” of antibiotic when introduced into the bacteria medium was not optimal for sustained release. In a subsequent paper, Ratner optimized conditions by studying how a plasma-deposited thin film barrier would affect the controlled release of antibiotics. He was able to successfully control the initial release “burst” of antibiotics, which led to a longer release of antibiotics, by depositing a plasma thin film over the polyurethane matrix. He concluded that the optimal conditions included etching the plasma with argon, pretreating it with BMA for 1 minute at 80 watts, and then depositing the plasma onto the matrix at 40 watts for 20 minutes. Not only would this allow for the sustained infection-resistant characteristics of this type of device, but this information could be adapted and modified to fit a drug delivery platform to study the controlled release of drugs.
Photo-patterning of porous hydrogels for tissue engineering (2007)[8]

Pore size and geometry strongly influence how a cell will behave in vitro. A pore must be big enough so that a cell can move around, but not so big that the cell cannot attach to where it needs to. Having the ability to control pore size and geometry and tailor it to different situations is a must in tissue engineering. In 2007, Ratner and his group designed a porous, degradable scaffold with which they were able to make unique, well-defined architectures. These scaffolds were made out of poly(2-hydroxyethyl methacrylate) hydrogels that were made porous via the sphere-templated scaffold fabrication technique. The patterned, porous hydrogel was creating via a photomask with a crosslink-initiating light: the areas under the photomask did not crosslink whereas the exposed areas crosslinked. This resulting in the ability to create channels ranging in size from 360±25 to 730±70 μm in a 700 μm thick hydrogel with pore diameters ranging in size from 62±8 to 147±15 μm. To assess the potential of these scaffold as tissue engineering devices, the group first attached type I collagen to the scaffolds and then seeded on skeletal myoblast cells. As a result, cell elongation, cell spreading, and fibrillar formation were observed, all things pointing to these scaffolds as being valid biomaterials that can be tailored to different needs.
Proangiogenic scaffolds as functional templates for cardiac tissue engineering (2010)[9]
One of the many, many benefits of biomaterial scaffolds is that you are able to modify the scaffold to fit the need in vitro, and then implant that specialized scaffold into the area of interest. In 2010, Ratner and his group created a hydrogel and shaped it to fit their needs, integrated the scaffold into host myocardium, and then instructed the scaffold to reconstruct the architecture of functional cardiac tissue. The scaffold was made from a poly(2-hydroxyethyl methacrylate-co-methacrylic acid) hydrogel, which had been shaped into the original architecture of the myocardium via microtemplating. These scaffolds contained 60 μm-diameter, parallel channels with pore diameters ranging from 20 to 40 μm. The channels organized cardiomyocyte bundles while the pores helped to enhance angiogenesis. When human cell-derived cardiomyocytes were implanted onto the scaffold and cultured in vitro, they proliferated for two weeks and were able to reach adult heart density. Furthermore, when a mixture of cells including cardiomyocytes was introduced into the system, the nonmyocytes migrated into and through the pores, which were originally meant for mass transfer of myocytes, whereas the cardiomyocytes stayed in the channels. When the scaffold was treated with a serum-free medium, the nonmyocytes died off, which resulted in a predominantly cardiomyocyte-containing scaffold with cells that were able to live up to two weeks in vitro. With further studies, such a scaffold could be used to heal infarcted human hearts with minimal invasion.
Degradable, Thermo-Sensitive Poly(N-isopropyl acrylamide)-Based Scaffolds with Controlled Porosity for Tissue Engineering Applications (2010)[10]
One of the main components of created a biomaterials is designing a structure that mimics nature or is structurally applicable to the task at hand. For example, a non-porous, non-degradable object will not work well for the delivery of a drug. It is with these ideas in mind that Dr. Ratner’s group created a fully degradable, thermo-sensitive, pore controlled scaffold based on poly(N-isoproyl acrylamide). These biodegradable hydrogels were made via the photo-polymerization of N-Isopropylacrylamide with 2-methylene-1,3-dioxepane and polycaprolactone dimethacrylate. Not only were these hydrogels biodegradable with tunability, but, along with their degraded counterparts, proved to be non-cytotoxic, owing to their potential to be used in the body without harmful side affects. Pore sizes were controlled via the sphere-templated scaffold fabrication technique during synthesis and via temperature. An increase in temperature from 25 degrees Celsius, roughly room temperature, to 37 degrees Celsius, the temperature of the body, results in a decrease in the pore size from a 55 μm-diameter to a 39 μm-diameter. The group was able to efficiently load these hydrogels with fibroblast cells at 25 degrees Celsius and then lock the cells in proangiogenic-sized pores by increasing the temperature. These “trapped” fibroblast cells were able to grow on the scaffold as well as penetrate into it, resulting in a cell sheet within five days. In the future, the group looks to study how these hydrogels can be used as a bioreactor to culture smooth muscle cells via the cycling of expansion and contraction.
Biocompatibility, Bio-Acceptance
Fibrinogen adsorption, platelet adhesion and activation on mixed hydroxyl-/methyl-terminated self assembled monolayers (2006)[11]
The success of biomedical devices that come into contact with the host’s blood hinges on if the blood recognizes the implant as foreign, and how strongly the blood reacts to it. In order to gain a better understand of how device surface modifications affect this reaction, Ratner, along with a group from the University of Porto in Portugal, investigated how fibrinogen adsorption, platelet adhesion, and platelet activation was affected by altering the surface functional groups of self-assembled monolayers (SAMs). They made a variety of materials, including different ratios of methyl- and hydroxyl-terminated alkanethiols on gold, and studied the affects using radiolabeled human fibrinogen (HFG), scanning electron microscopy, and glutaraldehyde-induced fluorescence technique. The study resulted in the linear decrease of HFG adsorption to materials with an increase in the amount of hydroxyl-terminated groups, which also increases the hydrophilicity. More specifically, SAMs with 65% and 100% incorporation of hydroxyl-terminated alkanethiols resulted in a lack of platelet adhesion and activation, which means these materials could be used for implants that must come into contact with blood. Furthermore, the 65% hydroxyl-terminated SAMs pre-immersed in albumin showed a low affinity for binding platelets, which was in contrast to when they were pre-immersed in HFG. 100% hydroxyl-terminated SAMs have a higher affinity for HFG than for albumin whereas the 65% SAM does not, which can be attributed to the 100% SAM’s overall lower protein binding or by the potential hydrogen bonds between the hydroxyl groups and the amines of HFG’s native structure, which 100% hydroxyl-terminated SAMs have more of. The idea of an increase in hydrophilicity resulting in an increase in biocompatibility is fairly trivial, but the group was able to assess the exact mechanism, as well as tunable parameters of platelet adhesion, that could be used to create implants that result in a healing process.
Platelet and leukocyte adhesion to albumin binding self-assembled monolayers (2011)[12]
In order to elicit a foreign body response, biomaterials must invoke the attack of proteins, cells and bacteria, which cause things like implant infections, collagenous capsules, and implant breakdown. However, there exists a type of surface that is biologically resistant to these types of things: non-fouling surfaces. These surfaces would not be ideal for implants, as it is necessary for selective proteins and cells to interact with the implant in order to implement the “biomaterials that heal” idea. Therefore, being able to selectively choose which proteins and cells come into contact with an implant is key for success. In 2011, Ratner and his group used non-fouling, tatraethylene glycol-terminated SAMs as a background surface while studying an 18-carbon ligand’s ability to selectively and reversible adsorb albumin. Ethylene glycol polymers had already been implemented as non-fouling surfaces, so the groups objective was to study how different percentages of attached 18-carbon ligands would affect albumin binding. The group found that SAMs with 2.5% coverage by 18-carbon ligands caused the surface to selectively and reversibly bind albumin from plasma without binding platelets. Increasing amounts of ligand coverage resulted in an increase amount of albumin attachment, but also resulted in the increase in platelet adhesion and leukocyte adhesion. In conclusion, they were able to create a system that selectively and reversible absorbs a particular protein, but more importantly, they were able to show how surfaces can be tailored to various conditions and circumstances. This information could be highly useful in designing various types of biomaterials.
Biomaterial-Induced Angiogenesis To Address Peripheral Vascular Disease: The Application of Sphere Templated Hydrogels (2013)[13]
Peripheral arterial disease is an arterial blocking disease of the legs that can lead to muscle ischemia, disabilities, and the potential for limb loss. In 2013, Ratner and his group studied the potential of using sphere-templated hydrogel scaffolds to create an angiogenic response in vivo, ultimately restoring circulation to ischemic limbs. The group made three scaffolds to work with: poly(2-hydroxyethyl methacrylate) scaffolds with longitudinal channels and spherical pores, dubbed “channeled” scaffolds, poly(2-hydroxyethyl methacrylate) scaffolds with only pores, dubbed “porous” scaffolds, and polytetrafluoroethylene, with channel-like spaces between fibrils, dubbed “control” scaffold. They found that the porous scaffolds were the most efficient material in cell infiltration. The channeled scaffolds, however, were the most efficient material in vascular growth, although their lumen widths were small. The original channeled scaffolds contained 30 μm-diameter channels, so the group also created 60 μm-diameter channel scaffolds in a failed attempt to increase the lumen width. The data did not lead to the results the group was looking for, but it did shed light on how a defined, uniform channel system is more beneficial to cell infiltration than randomly dispersed channels, such as in the control scaffolds. Though more research and work must be put into this type of scaffold to induce circulation in ischemic limbs, the potential is there.
Porous Implants Modulate Healing and Induce Shifts in Local Macrophage Polarization in the Foreign Body Reaction (2014)[14]
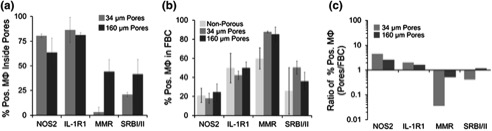
In order to create the optimal conditions for “biomaterials that heal”, we must understand why biomaterials have such a hard time being integrated into the body. This includes the foreign body reaction or foreign body response. The foreign body response is when macrophages, fibroblasts, myofribroblasts, and other cell types attack the implant and attempt to encapsulate it in a collagenous capsule. This strongly inhibits the function of the implant. In 2014, Ratner and his group studied how macrophage polarization creates the foreign body response of implants. They did this by studying sphere-templated hydrogels, with 34 μm, uniform pore diameters being the optimal size for reduced fibrosis and increase vascularization. The group hypothesized that improved healing is a result of shifting the ratio of M1, pro-inflammatory cells, to M2, pro-healing cells. After studying scaffolds of varying pore sizes, the results showed that macrophages in implanted pores shifted towards the M1 macrophage compared to the external cells, which shift towards M2 macrophages, with this shift being greatest for the 34 μm diameter pore sizes. Therefore, an increase in the ratio between M1 and M2 inside the scaffold and an decrease in the same ratio outside the pores results in a pro-healing environment, which leads to reduced fibrosis, reduced foreign body response, and increased vascularization. Though the knowledge that porous materials reduce the foreign body response, Ratner was able to assign a mechanism to such a response. These biological cues can then be taken into account when creating implants, or other mechanisms can be made to increase the ratio of M1 to M2 inside the pores.
Other Publications
Dr. Buddy Ratner has also released a number of review articles, which not only pose as a synopsis of current, state-of-the-art research, but are also a collection of his thoughts and ideas, some philosophical, some scientific. These papers include some of his most cited publications, including “Biomedical surface science: Foundations to frontiers” [15] and “Biomaterials: Where we have been and where we are going.” [16] These are some of his review articles, which include hypotheses of where we will be in the field in ten to twenty years. His more philosophical works include “A pore way to heal and regenerate: 21st century thinking on biocompatibility,” [17] which questions that idea of biocompatibility and how we measure it, and “Going out on a limb about regrowing an arm.” [18] Dr. Ratner is not only a founding father of biomedical engineering, tissue engineering, and various materials and methods, but he is also still paving the way and contributing to the greater good of the scientific process.
References
[1] Buddy Ratner, an oral history conducted in 2000 by Frederik Nebeker, IEEE History Center, Hoboken, NJ, USA. [1]
[2] Colorado State University Biography on Buddy Ratner [2]
[3] University of Washington, Department of Bioengineering, Biographies Website [3]
[4] Northwestern International Institute for Nanotechnology, News & Events Website [4]
[5]Ratner, B. D. “Research Areas.” Research. University of Washington Engineered Biomaterials. Web. 31 March, 2017. [5]
[6] Kwok, C. S., Wan, C., Hendricks, S., Bryers, J. D., Horbett, T. A., Ratner, B. D. (1999). Design of infection-resistant antibiotic-releasing polymers: I. Fabrication and formulation. Journal of Controlled Release, 62, 289-299. [6]
[7] Kwok, C. S., Horbett, T. A., Ratner, B. D. (1999). Design of infection-resistant antiobiotic-releasing polymers II. Controlled release of antibiotics through a plasma-deposited thin film barrier. Journal of Controlled Release, 62, 301-311. [7]
[8] Bryant, S. J., Cuy, J. L., Hauch, K. D., Ratner, B. D. (2007) Photo-patterning of porous hydrogels for tissue engineering. Biomaterials, 28, 2978-2986. [8]
[9] Madden, L. R., Mortisen, D. J., Sussman, E. M., Dupras, S. K., Fugate, J. A., Cuy, J. L., Hauch, K. D., Laflamme, M. A., Murry, C. E., Ratner, B. D. (2010). Proangiogenic scaffolds as functional templates for cardiac tissue engineering. Proceedings of the National Academy of Sciences, 107(34), 15211-15216. [9]
[10] Galperin, A., Long, T. J., Ratner, B. D. (2010). Degradable, Thermo-Sensitive Poly(N-isopropyl acrylamide)-Based Scaffolds with Controlled Porosity for Tissue Engineering Applications. Biomacromolecules, 11(10), 2583-2592. [10]
[11] Rodrigues, S. N., Goncalves, I. C., Martins, M. C. L., Barbosa, M. A., Ratner, B. D. (2006). Fibrinogen adsorption, platelet adhesion and activation on mixed hydroxyl-/methyl-terminated self assembled monolayers. Biomaterials, 27, 5357-5367. [11]
[12] Concalves, I. C., Martins, M. C. L., Barbosa, J. N., Oliveira, P., Barbosa, M. A., Ratner, B. D. (2011). Platelet and leukocyte adhesion to albumin binding self-assembled monolayers. J. Mater. Sci.- Materials in Medicine, 22(9), 2053-2063. [12]
[13] Terasaki, D., Sobel, M., Irvin, C., Wijelath, E., Ratner, B. D. (2013). Biomaterial-Induced Angiogenesis To Address Peripheral Vascular Disease: The Application of Sphere Templated Hydrogels. ACS Symposium Series, 1134, Ch. 15, 245-257. [13]
[14] Sussman, E. M., Halpin, M. C., Muster, J., Moon, R. T., Ratner, B. D. (2014). Porous Implants Modulate Healing and Induce Shifts in Local Macrophage Polarization in the Foreign Body Reaction. Annals of Biomedical Engineering, 42(7), 1508-1516. [14]
[15] Castner, D. G., Ratner, B. D. (2002). Biomedical surface science: Foundations to frontiers. Surface Science, 500(1-3), 28-60. [15]
[16] Ratner, B. D., Bryant, S. J. (2004). Biomaterials: Where we have been and where we are going. Annual Review of Biomedical Engineering, 6, 41-75. [16]
[17] Ratner, B. D. (2016). A pore way to heal and regenerate: 21st century thinking on biocompatibility. Regenerative Biomaterials, 3(2), 107-110. [17]
[18] Ratner, B. D. (2013). Going out on a limb about regrowing an arm. J. Mater. Sci.- Materials in Medicine, 24 (11), 2645-2649. [18]
