Bone Marrow on a Chip, by Emily Brackett
Background
Bone Marrow
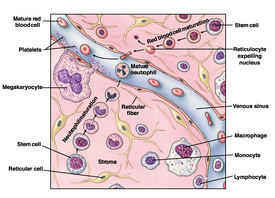
Bone marrow is important for the creation of components of the blood from stem cells. Figure 1 shows stem cell differentiation into red blood cells and neutrophils. Bone marrow also contains megakaryocytes responsible for the creation of platelets, lymphocytes, adipocytes, and many other important cell types [1]. These many cell types, their many functions, and their necessary chemical and physical environments cause bone marrow to be difficult to maintain in culture.
Organ on a Chip
There are lots of successful models of organs on a chip that incorporate mechanical, physical, and chemical cues from the environment to culture tissue. For example, the lung on a chip model incorporates vacuum chambers into the device such that suction or pressure can be applied to mimic the physical and mechanical atmosphere of the lung [2]. Current models for bone marrow on a chip include functionalized surfaces for attachment, porous materials to induce growth, and the inclusion of growth factors and cytokines to maintain stem cells.
History
1974 - First proliferating culture of hematopoietic cells in vitro [5]
1980s - Research in microfluidics developed lab on a chip technology [3]
Late 1990s - Cornell University researchers begin tissue on a chip models [6]
Early 2000s - Research in bone marrow on a chip changes from cell sorting to cell culture [5]
2014 - Between January and April of 2014, 350 papers regarding bone marrow and microfluidics were published
Current Models
Current bone marrow on a chip models incorporate physical, mechanical, and chemical properties that contribute to the “bone” environment. For example, some devices use platforms sufficiently stiff, responsive, or shaped to induce growth [6]. Some of these platforms are used to mimic the porous nature of bones, some utilize filters to prevent cells from being flushed from the device, etc [6][7]. Mechanical cues, such as low flow rates to prevent detachment, or pressure to mimic the body, are necessary [8].
Chemical cues such as growth factors and cytokines are often used to prevent differentiation [9]. Now, there are several pre-made medias, additives, and other chemical signals available, such as mouse stem cell factor (MSCF) kits. These kits utilize recombinant proteins to mimic the environment of the body [10]. Some chemical signaling is focused more towards the bone and marrow interface by functionalizing the surface of the microfluidic with ECM proteins such as fibronectin.
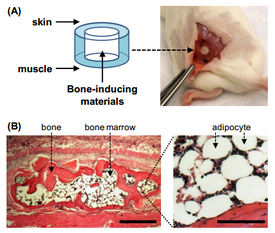
Models have also utilized in vivo capturing of bone marrow cells and combined microfluidic culture. For example, Figure 2 shows a hollow, barrel shaped PDMS subcutaneous insert. This insert induces growth of bone marrow into the chamber, which can then be surgically removed and cultured in a microfluidic device [6]. There are other models similar to this which have porous, bone-like, growth-inducing materials which are used in vivo and then transported to a microfluidic culture chamber. In vivo models have also shown formation of vasculature.
Market
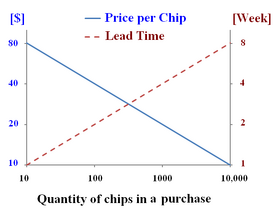
Mouse studies cost approximately $3500 per mouse [11]. While monolayer culture methods can be less expensive, they are also a much less accurate mimic of the human body. Microfluidics cost approximately $10-80 dollars per chip [12] (See Figure 3). Further research is needed in the field of bone marrow microfluidics to be able to asses whether the cost of performing a microfluidic experiment yields more accurate results in comparison to mouse studies, necessitating a shift from mouse studies to microfluidics.
References
[1] "Histology Pictorial Guide - Blood and Haemopoiesis Pg.4." Histology Pictorial Guide - Blood and Haemopoiesis Pg.4. N.p., n.d. Web. 15 Apr. 2014.
[2] Diviya D. Nalayanda, Christopher Puleo, William B. Fulton, Leilani M. Sharpe, Tza-Huei Wang, Fizan Abdullah (2009), “An open-access microfluidic model for lung-specific functional studies at an air-liquid interface”
[3]Gao, Peter. "Landmark Studies in the History of Bone Marrow-derived Hematopoietic Stem Cell Research - Peter Gao - Discovery Medicine." Discovery Medicine. Johns Hopkins University, n.d. Web. 15 Apr. 2014.
[4] S.C.Terry,J.H.Jerman and J.B.Angell:A Gas Chromatographic Air Analyzer Fabricated on a Silicon Wafer,IEEE Trans.Electron Devices,ED-26,12(1979)1880-1886.
[5]Baker, Monya. "A living system on a chip." Nature 471.7340 (2011): 661-665.
[6] Torisawa, Y. S., et al. "Bone marrow-on-a-chip." 16th International Conference on Miniaturized Systems for Chemistry and Life Sciences, Okinawa, Japan. 2012.
[7] Schirhagl, Romana, et al. "Microfluidic purification and analysis of hematopoietic stem cells from bone marrow." Lab on a chip 11.18 (2011): 3130-3135.
[8] Park, Sang-Hyug, et al. "Chip-based comparison of the osteogenesis of human bone marrow-and adipose tissue-derived mesenchymal stem cells under mechanical stimulation." PloS one 7.9 (2012): e46689.
[9] Zhang, Wenting, et al. "Patient-Specific 3D Microfluidic Tissue Model for Multiple Myeloma." Tissue Engineering Part C: Methods (2014).
[10] Recombinant Mouse Stem Cell Factors (SCF);MAN0004193;Life Technologies: Frederick, MD, April 15, 2014.
[11] Anft, Michael, and Bill Cigliano. "Of Mice and Medicine." Johns Hopkins Magazine. N.p., n.d. Web. 15 Apr. 2014.
[12] "Pricing." UFluidix Pricing Comments. N.p., n.d. Web. 15 Apr. 2014.