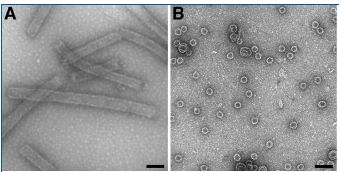
Biomod/2011/MIT/Origami/Capsid

Introduction
Viruses have a protein shell called capsid which is essential for survival of the virus. It encloses the viral genome and is where the process of replication occurs. Without the capsid, the virus would not be able to infect other cells, which is why it could be a good target for treatment of HIV.
Structure
HIV has a conical capsid made up of viral 25.6kDa CA protein (p24) arranged into a mostly hexagonal lattice, similar to a honeycomb. It protects and carries the viral RNA genome inside. The capsid is also surrounded by a layer of matrix protein (p17), a lipid bilayer, that helps anchor gp120 and gp41. The CA proteins arrange into hexamers and pentamers. This assembly is controlled by an electrostatic switch. There are seven pentamers closing off the wider end of the cone, which in turn is made up of around 250 hexamers, and five pentamers closing off the other end. Pentamers are distributed asymmetrically among said regions. Close interactions between two adjacent CA domains stabilize this shell. At first, the immature capsid is spherical and then it undergoes a rearrangement to form the mature conical capsid. Little is known about this process, but studies have shown that the CA domains are usually cylindrical, and a single-point mutation allows them to fold into other shapes.
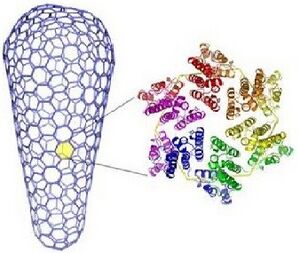
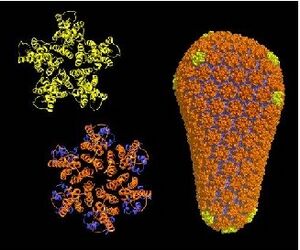 Left: Honeycomb-like structure of the HIV capsid. (Bardi, Target Health Global) Right: Conical capsid made up of hexamers (orange) and pentamers (green) (Pornillos, et al., Nature)
Left: Honeycomb-like structure of the HIV capsid. (Bardi, Target Health Global) Right: Conical capsid made up of hexamers (orange) and pentamers (green) (Pornillos, et al., Nature)
The CA proteins act like a scaffold for virus assembly. These proteins are composed of two independently folded rigid domains, amino-terminal domain (NTD) and carboxy-terminal domain (CTD), held together by a flexible linker, so overall, the protein has some range of mobility. The domains have an α-helical secondary structure. The NTD is responsible for folding into the hexamers and pentamers, while the CTD connects all the rings to form the lattice. The CTD wraps around the pentamers and hexamers and then links to another CTD to connect adjacent subunits. The CTD subunits are free to rotate relative NTDs, allowing hexamers and pentamers to bend into different angles. Pentamers are smaller, since only 5 CA proteins link at the NTD compared to the 6 proteins required for hexamers. There are also less of them because the positively-charged amino acid arginine is found in the center of both hexamer and pentamer rings, and because pentamers are smaller, there is a stronger repulsive force, causing them to be less stable and thus favoring the production of hexamers.
Assembly
Capsid assembly is an intracellular process. The formation of immature viruses is driven by the interactions between CA domains of the Gag polyprotein in the plasma membrane of a host cell. It is also an energy dependent process. HP68, a host protein that contains two ATP/GTP binding sites, associates with the partially assembled Gag complexes and helps to complete their assembly into immature capsids. Gag protein is the major structural component of the immature HIV virus. It forms into hollow spheres that are surrounded by an electron-dense doughnut-shaped structure. Once these particles are released, the Gag polyprotein is digested by protease, leading to a change of shape and assembly of the capsid and the mature virus. This digestion occurs sequentially:
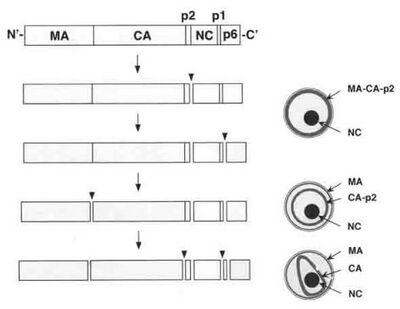
- One The nucleocapsid domain and spacer peptide p2 complex is cleaved.
- Two The electron-dense core condenses while the matrix domain-capsid domain-p2 complex remains attached to the viral envelope.
- Three The matrix domain-capsid domain complex is cleaved.
- Four The capsid domain-p2 complex is released and the capsid condensates into spheres.
- Five Capsid domain/p2 is cleaved to form the conical capsids.
A single point mutation on R18A causes the proteins to assemble into spheres and cones, rather than their wild type-cylindrical shape. Gag is cleaved into three proteins: the matrix protein, which associates with the membrane, the nucleocapsid protein, which contains the viral genome, and the CA proteins, which make up the mature capsid. Studies show that the capsid forms within 8-9 minutes after nucleation of an assembly site. The capsid encloses an electron-rich core.
(A) Wild-type cylindrical shape. (B) R18A mutated proteins. (Pornillos et al., Structure of Full-Length HIV-1 CA: A Model for the Mature Capsid Lattice)
There are three regions of Gag required for capsid assembly: Membrane-binding domain: It signals the Gag precursor to bind to the membrane for budding. After infection, it dissociates from the membrane and moves to cytoplasm. Interacting domain: NTD and CTD interactions to form the mature capsid Late Domain: Regulates processes after budding occurs but before release. Studies also suggest that actin is involved in the assembly and mechanisms of the capsid, but more research still needs to be done as its function is still uncertain.
The basic assembly unit is made up of two CA subunits linked by the CTD. Refolding of the first 13 residues into a β-hairpin destabilizes the immature NTD hexamers. Cleavage at the C-terminus appears to be the "maturation switch." Upon capsid maturation, the diameter of the hexamer increases, making holes smaller, and the shell wall thins out.
Capsid assembly is a rapid, dynamic, self-assembly process, but the specific mechanisms are still unknown. The CA dimers condensate into disordered clusters and the lattice is nucleated by the appearance of one hexamer unit. The lattice then spreads rapidly. It is a cooperative process, with best fit cooperativity parameter larger than 1. There is a termination step that might involve the capping of growing CA tubes. It has been simulated by coarse-grained models. It seems that viral RNA plays a scaffolding role during assembly. The cone spans the entire width of the virion with its narrow end in close contact with the viral MA, suggesting that the lattice is nucleated at the narrow end.
In vitro, recombinant CA self-assembles at high ionic strength or upon addition of crowding agents. Crowding agents are mimicked in vitro by adding a high concentration of inert natural or synthetic polymers. Changes in overall charge don't prevent assembly. The critical concentration for self-assembly in vitro is 78µM at pH 6 or 39µM at pH 8 and 1M NaCl. The rate at 2.25M NaCl is roughly proportional to the third power of the protein concentration. The assembly rate increases with increasing salt and protein concentrations. CA induced with salt polymerizes in vitro into long cylindrical structures. When incubated with RNA at pH 8 and low NaCl concentration (0.1M), it polymerizes into hollow tubes, and at high salt concentration (0.5M) into a mixture of both cones and cylinders. Without the presence of RNA, the ionic strength needed is high in order to polymerize. CA proteins are also pH sensitive. Epitopes on CA become inaccesible when the pH is below 6.8. There is a feature in common for in vitro assembly of CA: a slow dialysis step in which a change in solvent conditions, usually the NaCl concentration, leads to polymerization.
Mechanisms
After infection of the cell, the virus starts to replicate using the same mechanisms of the infected host cell and begins to create new capsid subunits via the cell's protein synthesis mechanism. While the capsid is being assembled, a 'door' is created at one end of the capsid that will let viral RNA into the capsid. An interaction between the 5' untranslated region of the RNA genome and the nucleocapsid domain within Gag drives packaging of viral RNA. Cyclophilin A (CypA) needs to bind to the CA protein soon after the virus enters the target cell. It binds to an exposed proline-rich loop in the NTD to be incorporated into the HIV virion. Cells lacking CypA are defective in the reverse transciption. Once it infects the cell, cellular enzymes strip away the capsid and proteins p24, which coats the genome, and p17, which lines the capsid on the inside, and the HIV genome is released. The average lifetime of each oligomer decreases as temperature increases.
Treatment
here are currently no FDA approved drugs that target the capsid. However, some inhibitors appear to target the NTD and CTD lattice in infected cells: CAP-1, a small molecule that binds to the bottom of the NTD, and two versions of a peptide inhibitor, CAI and NYAD-1. CAP-1 acts in the late stage of replication, but does not stop further infection by already infected particles. CAI is a twelve-residue peptide that inhibits capsid assembly of both mature and immature viruses by binding peptides between helices. NYAD-1 is a derivative of CAI.
Amino acid substitution in the NTD can also disrupt assembly of the capsid.
The antiviral compound PF-1385801 was found to inhibit replication, so analogs were synthesized. PF-3450074 worked against all strains of HIV-1 and PF-3759857 was active against HIV-2. Those particles that were treated with PF-3450074 were missing the representative central density of mature capsids and showed irregular budding, meaning it was affecting CA-CA interactions in capsid assembly and therefore the core lacks the stability it needs.
PF-3450074 makes hydrophobic and stacking interactions with the CA domain and forms hydrogen bonds via water molecules. A model of the PF-3450074/CA complex was built where specific interactions can be studied (PDB code: 2XDE).
Why is the capsid a good target?
- Overall, the structure of the capsid is held together by protein-protein contacts. If even one of these are disrupted, assembly fails.
- The NTDs and CTDs are conserverd among the different strains of HIV.
- It is where interactions between viral proteins that are essential for virus survival take place, so by targeting the capsid these would be inhibited.
- Resistance to drugs that target other sites has already developed.
Goal of origami
Because there aren't any drugs available that target capsid assembly, a DNA origami that helps with drug delivery to the capsid is not possible. Furthermore, little is known about the capsid opening mechanism so it would be hard to target this stage of HIV infection. Based on the research done so far, the best origami would prevent the capsid from assembling in the first place. It would be even better if we found a way to also disrupt the lattice of already assembled capsids, destroying them.
Idea: Synthetic CA proteins assembled into a hexamer or a pentamer that will interact with the rest of the CA domains to make up the capsid and once it is fully assembled signal the origami to fold or disassemble shortly after, thus breaking the entire capsid and preventing the RNA genome from transcribing.
Interactions
- NTD-NTD interactions: A NTD interacts with another NTD intermolecularly to form rings. These interactions are mediated by the first three α-helices of each subunit, forming a fifteen-helix bundle in the pentamers and an 18-helix bundle in the hexamer. The sidechains in these bundles form a hydrophobic core while the polar residues at the periphery are hydrophilic.The majority of this interface is created by hydrophilic contacts. Water molecules bridge polar side chain and backbone atoms. Helices 1 and 2 make NTD-NTD contacts.
- NTD-CTD interactions: CTDs interact intermolecularly with inner NTD rings to keep the lattice together. Helix 4 of the NTD is inserted into a groove on the CTD from an adjacent CA molecule to futher stabilize intrahexameric contacts. Extended side chains from helix 8 of CTD pack against C-terminal of helix 3 and N-teminal end of helix 4 of NTD.
- CTD/NTD dimerization: side chain of Tyr145 is at junction of NTD and CTD. It dimerizes through parallel packing of helix 2 across a dyad.
- Subunit-subunit interactions are weak. Stabilized by hydrophobic interactions. Helix 2 of one subunit packs itself lengthwise against helices 1 and 3 of the adjacent subunit.
- The Major Homology Region (MHR, 20 conseved amino acidson the CTD) forms a hydrogen bonding network that interconnects with helices 1 and 2 (Gln155, Gly156, Glu159, Arg167). Glu159 and Arg167 form hydrogen bonds to main-chain amide and carbonyl groups across the MHR loop and a salt bridge forms.
- S149 on helix 7 participates in the CTD-NTD contacts strengthening the hexamer.
- The hydroxyl group of S109 forms a hydrogen bond with Q114 that stabilizes helices 5 and 6.
- CTD/CTD dimerization: hydrophobic interaction between residues at N-terminues of CTD (Tyr145, Thr148, Leu157) and residues in helix 9 (Val181, Trp184, Met185, Val191)
- The most prominent interface for capsid dimerization is helix 9 and hte preceeding group (174-189) in CTD.
- salt bridge between residues 1 and 51
- Interface formed by CTD dimers of 3 neighboring hexamers might be involved in organizing intermolecular contacts.
NTD
- Residues 1-145, 7 α-helices
- Refolds into a β-hairpin structure stabilized by a salt bridge
- Asymmetric arrowhead shape
CTD
- Residues 151-231, 4 α-helices
- Negatively charged side chains
- Contains a conserved section of 20 amino acids called the Major Homology Region (MHR) that makes up the interior surface of the hexamer
- Exists predominantly as dimer in solution at low ionic strength
- Affinity of dimer: Kd~10^-5 M
Liker
- Residues 146-149, helix 9
Holes
- There is a roughly cylindrical hole at the center of the hexamer 26.3Å in diameter and spaced at 74.2Å intervals, and a trilobed hole formed by the spokes of the hexamers.
- Small macromolecules diffuse in and out
- Residues 11-32 and 87-100 are oriented toward the holes.
- Gp120/gp41 protein complex tails are postulated to reside in these holes.
Dimensions and Orientations
- Internal diameter: 25Å
- External diameter: 100Å
- Inter-ring spacing: 107Å / hexamer-hexamer spacing 90-100Å
- Protein shell: ~72Å thick
- Conical shell: ~120nm x 60nm
- NTD: 45 x 35 x 18 Å
- CTD: 28 x 28 x 20 Å or 27 x 29 x 38 Å?
- cones need to adopt one of five allowed cone angles: 19.2, 38.9, 60, 83.6, 112.9
- CTD may be positioned either in the same vertical axis than that of the NTD or slightly tilted
- Hexamers are twisted with respect to the cone axis
- Shortest intermolecular NTD-NTD distances within a hexamer are between helices 2 and 3
- Shortest intermolecular CTD-NTD distances are between helices 4 and 8
- Helices 1-3 form a wedge with an angle ~60.
- α-carbon - α-carbon distances across opposite Arg18 in pentamers: 10.9Å, in hexamers 16.5Å