Biomod/2011/Columbia/MotorProTeam:ProjectDevelopment

Research Topic
Brainstorming for the project began in February 2011. The following ideas were taken into consideration for the competition:
- Genetic engineering of hemoglobin
- Microtubule sheets for force multiplication of kinesin
- Electric power from ATP
- Smart indicator supermolecule (multi-receptor recognition by multifunctional drug delivery vehicle)
- Slow protein unfolding by motor proteisn
- Self-Healing
- Nanofactory using molecular shuttles
- Circuit assembly by molecular shuttles
We ultimately decided on an adaptation of the second idea (microtubule sheets for force multiplication of kinesins). This choice was based on interest, feasibility, and the potential application of the prospective projects. We aim to create a universal transport system that can selectively carry nano- and micro-scale cargo over macroscopic distances.
Structure
Brainstorming
From this point, we met to design the structure of the device.
Initially, we aimed to create a raft-like surface, that is, an array of microtubules of uniform length, pointed in the same direction, and connected either by biotin-streptavidin bonds. This idea, although elegant, proved unfeasible. When multiple microtubules are connected, they form wrap around and join on their own ends, forming spool-like structures rather than a raft-like structure. Controlling the length of each microtubule also presented a challenge to the idea. We considered using dynein (another motor protein) as an alternative to kinesin because its short length would theoretically allow the microtubules to push against one another and align. However, the challenges of creating microtubules of uniform length that would bind to each other in a flat shape (rather than a bundle) seemed insurmountable, thus we deemed this structure implausible.
Another potential alternative we considered was a cylindrical system that operated on bead-geometry as opposed to the motility assay. A cylinder would be constructed of microtubules, and cargo would attach to the cylinder. Kinesin molecules on a fixed surface would pass the microtubules along thereby moving the cargo. The benefit of this structure is in the high surface area to volume ratio of the cylinder. This would allow a large amount of cargo to be carried along. Unfortunately, however, the bead geometry is much less effective and less studied than the gliding geometry. We therefore continued theorizing and discussing potential structures for the universal transporter.
While waiting for the patterned silicon wafers to arrive, we brainstormed more ideas for the structure. These included constructing a sheet out of biotinylated nano- or microspheres. When the solution containing the biotin spheres dries out, the spheres form an array. Biotin-streptavidin bonds could then link microtubules to the bottom of the array. Cargo could then load onto the top surface of the array. However, early tests showed that this array did not form as readily or uniformly as we had hoped, and the geometry presented for attaching the microtubules and cargo seemed non-ideal. Thus, we eventually rejected this idea as well.
The next, and probably most promising idea resembled a nano-truck constructed from microtubules, motor proteins, and a sheet of PDMS or other polymer as the loading bed. Microtubules would attach to the bottom surface of this sheet, allowing the entire structure to move as one unit when placed on kinesin coated surface.
Polymer sheets
Our ideas on what the shape the polymer bed would take went through several iterations. Initially we imagined high-surface area structures such as a finned (similar to the designs shown below) or open-topped box design, which would allow more cargo to bind per structure. Beds with designs of this type could be created by patterning the sacrificial layer and allowing the polymer from the PDMS mold to flow in and fill the necessary features before curing.
 |
 |
However, for the sake of simplicity we opted to work mainly with simple rectangular prisms. This saved us an extra step (adding a pattern to the sacrificial layer) that could have easily been added on once we had the rest of the design functional.
PDMS, however, could not be made on a small enough scale to fit in the flow cell, so we chose a polyurethane alternative (Norland Optical Adhesive 73) to create the structure.
Implementing the design
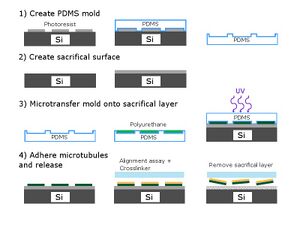
The structure would have to be small enough to fit inside a flow cell (roughly 100 microns by 1cm by 1cm). In order to achieve this type of size, silicon wafers were patterned with a photoresist mask. Handling such small particles manually would be virtually impossible, so a combination of microtransfer molding and a lift-off technique would be used to acquire small squares of PDMS.
SU-8 photoresist was used to print a pattern on a wafer to serve as a master. PDMS molds were created using this master. Next, a sacrificial surface was made. A plane of featureless AR5214 photoresist was added to the top of a second silicon wafer. The PDMS mold was micro-transferred onto the AR5214 sacrificial layer. The polyurethane prepolymer was used to fill in the features of the PDMS mold, and the structure was placed on top of the sacrificial layer. The prepolymer was cured using UV and the PDMS mold was peeled off. This resulted in a layer of photoresist with a pre-polymer pattern.
 |
 |
 |
A flow cell was created on the silicon wafer with the photoresist and pre-polymer patterning. The standard flow procedure was used, and microtubules would align and stick to the prepolymer. Next, a 25% by volume acetone solution was used to dissolve the sacrificial layer, thereby freeing the pre-polymer structures. This would result in a solution with polyurethane structures coated with aligned microtubules, which, when added to a new flow cell, would theoretically move upon a kinesin coated surface.
Alignment
After deciding on a structure, we investigated methods to align microtubules.
The simplest method initially appeared to be using magnetic fields to align the microtubules. The procedure was unfeasible to perform within the constraints of the lab equipment because magnetic fields had to be applied in the process of growing the microtubules.
Another method of alignment we investigated involved channels. Channels can be constructed out of PDMS and force the microtubules to align. However, there is no method to remove the microtubules from the channels without losing the alignment. We considered making the channels on the lower side of the structure and align the microtubules directly onto it. These microtubules, however, would not be functional because microtubules need to protrude from the channel on the bottom of the polyurethane structure in order to move along the kinesin motors below it. The height of the channel for this purpose would need to be around 12 nm (half of the diameter of a microtubule), and this level of detail proved impossible to achieve using the standard lithography techniques available to us.
One alternative was using shear force force from a fluid flow within the flow cell to align microtubules. Shear flow in a solution where the microtubules have been reversibly immobilized would produce alignment if applied for a long enough duration (the specific timings are still unclear). Whether this would produce alignment according to polarity as well remains unclear. Shear flow remains the simplest method we tried, and our laboratory is currently in the process of acquiring more advanced equipment that would allow for sustained, uniform flow over the current manual delivery system.
A final method to align microtubules entailed adhering the microtubules on one side through the use of a surface boundary. Applying a piece of tape during surface preparation creates a boundary between two regions, one with the requisite casein and kinesin necessary to adhere a microtubule and a blank region which microtubules do not adhere to. We attempted using this boundary in conjunction with shear flow in the hopes that microtubules that had partially adhered to the coated region would more readily align as the flow pushed their free tails into the blank region along the streamlines of the fluid flow. This method seemed promising, however it greatly reduced the yield of microtubules that had the potential to be aligned.
Cargo
The transport system we developed was intended to transport cargo on the nano and micro scale. As a proof of concept, we would use biotinylated nanospheres and microspheres of different sizes in order to test the efficacy of the transport structure. Later, we would try to design our structure so that it could bind to different kinds of biomolecules like proteins and DNA fragments etc by chemically modifying the top surface.
Three types of linkages were investigated for attaching microtubules and cargo to the structure. While microtubules need to bind permanently to the underside of our structure, the cargo should be able to load and unload at the respective loading and unloading stations.
- The biotin-streptavidin bond
This bond is well-known for its high strength and specificty. It is it is suited to permanently attach the microtubules to the structure because of its durability. Microtubule movement on kinesins remains unaffected by attaching the microtubules to biotin or streptavidin.
- Antibody-antigen bond
This bond is characterized by high specificity from a combination of non-covalent Hydrogen bonds and Van der Waal forces. These bonds can be easily dissociated by a change in pH or temperature. The antibody-antigen bonds are therefore potential candidates for attaching cargo to the structure.
- DNA Strands
DNA strands have high specificity and allow us to exert precise control on the strength of the bond by adjusting the length of the DNA strand. An increased advantage is that kinesin motors generate enough force to shear the bonds and break them. This property can be used to load and unload the cargo effectively. The bonds between DNA strands can be broken in two ways: the zipper geometry which requires less force and the simultaneous breaking which requires a greater force.
For the purpose of the project, three bond strengths were used on cargo. The weakest bonds bind cargo to the loading station, the intermediate ones bind cargo to the transporter, and the strongest ones bind cargo to the unloading station. The logic behind this is simple: At the loading station, the cargo breaks the weaker bond at the station and make a bond of intermediate strength with the transport structure. At the unloading station, the cargo should break from the intermediate strength bond with the transporter and attach to the unloading station via the strongest bond. DNA strands allow us to control the bond strength and are therefore good candidates for attaching cargo.