Biomod/2011/Aarhus/DanishNanoArtists:Results
RNA/DNA hybrid self-assembly
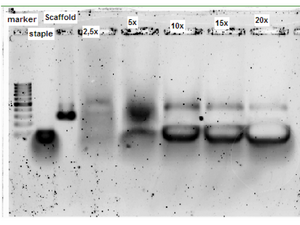
Initially an experiment was made to determine the optimal molar ratio between scaffold and staple strands. Self-assembly was attempted with the following 5 different scaffold to staple molar ratios: 1:2,5, 1:5, 1:10, 1:15, 1:20. In [Figure] we see a band shift, indicating assembly, in the last four lanes. The self-assembled structure has a lower mobility than the pure scaffold strand, suggesting that it folds into a more rigid structure. The scaffold strand has a mobility corresponding to approximately 300 nt, which is somewhat lower than the actual length of 370 nt. The higher mobility is likely due to the secondary structure of the scaffold strand caused by internal base pairing [Figure]. The length of the scaffold has, however, been verified on a denaturing gel (data not shown). In [Figure] a sharply defined band is observed at a ratio of 1:10. Further increasing the staple strand concentration does not seem to further improve self-assembly. For this reason, the molar ratio 1:10, was used in subsequent analysis of the structure.
The design of RNA/DNA hybrid was made in such a way, that the self-assembly will take place in a particular order. Initially the center square is assembled and then the rest, so that the structure close together. This stepwise self-assembly was investigated by making the self assembly with an increasing number of staple strands present (Figure). It is seen that only by the addition of the second last staple strand(I1), a clearly defined band emerge. Here, a very clear band shift compared to the original scaffold is also seen, so it is clear that there has been a change in structure from the initial unfolded scaffold strand. By adding the first staple strand we see no clearly defined bands, which means that the structure does not assume any particular solid structure in these steps. It appears, however, that the vague smear moved up a bit for each added staple strand, which indicates that the structure is assembled in succession, but it is first by the addition of I1 that the structure assumes a fixed structure. The unclear smear seen in the first 6 tracks can also be caused by contamination with ribonuclease, which will break down the structure. In this case, the clearly defined bands in the track 10 and 11 suggest that the overall structure is resistant to degradation by ribonuclease. The conclusion cannot be drawn from this analysis alone.