BME103 s2013:T900 Group1
| Home People Lab Write-Up 1 Lab Write-Up 2 Lab Write-Up 3 Course Logistics For Instructors Photos Wiki Editing Help | |||||||||
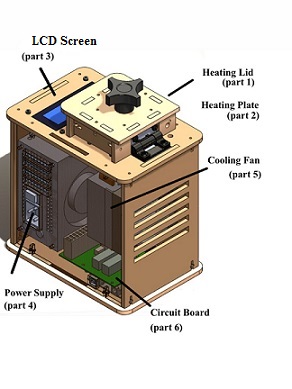
OUR TEAMLAB 1 WRITE-UPInitial Machine Testing The device displayed above, commonly referred to as an "Open PCR machine," is a mechanism that performs polymerase chain reactions that amplify DNA by creating a dynamic environment with extreme temperature changes. Upon undergoing mutilpe cycles of continual heating and cooling, the DNA present within the samples are separated then rapidly replicated resulting in multiple copies from a single strand of DNA. The PCR machine is hooked up to a computer using a usb cable allowing the user to control the desired number of cycles and temperatures for an experiment. Once the information is submitted into the system, the DNA experiences environmental changes controlled by the heating plate, heating lib, and cooling fan. The circuit board towards he bottom of the device then computes the information and transfers it to the LED screen presenting immediate results.
Because the Open PCR machine relies on a complex network of connections and parts, the function of the system will be disrupted if any connections were to be rearranged or unplugged. As proof, after unplugging the LCD screen(part 3) from circuit board (part 6), the machine did not display any information on the LCD screen. Likewise, the white wire that connects Circuit board (part 6) to heat block (part 2) allows the PCR machine to collect information about the temperature, but when unplugged, the machine does not read the temperatures accurately. In order to have a effective PCR system, it is imperative that all connections are correct and that each component of the machine is working properly. Test Run The initial sample test run took place on Tuesday, march 5 in the PCR introduction Lab with machine 9. After loading the required software onto a computer, a sample test was run with the user friendly design of the software. The first run was very efficient with minimal issues and the information provided by the device corresponded well with the computer. However, my team and I did eventually observed some disadvantages with the open PCR machine. Each cycle that took place while the device was operating took an extremely long time to complete, and eventually, we experienced the inconvenience of waiting over two hours for multiple cycles to run when performing our experiment. Another noticeable flaw in the device was that the information displayed on the LCD screen did not always correspond with the information being transmitted into the computer software. It was constantly behind with readings and fluctuated between information; therefore, the machine has the potential to throw off data collection if it is not monitored or maintained correctly.
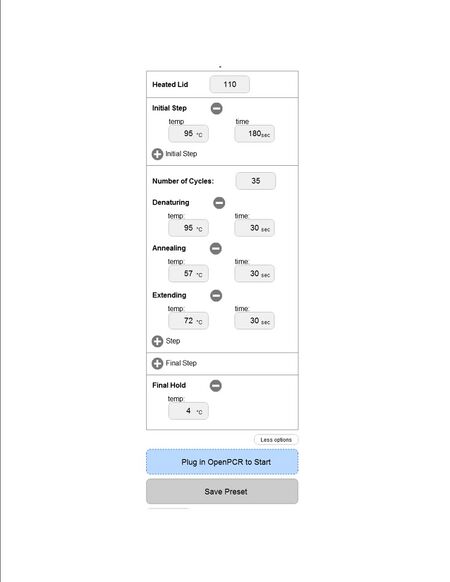
ProtocolsThermal Cycler Program Stage 1
Stage 3 DNA Sample Set-up
DNA Sample Set-up Procedure
PCR Reaction Mix
DNA Sample/Primer Mix
Research and DevelopmentSpecific Cancer Marker Detection - The Underlying Technology Polymerase chain reaction (abbreviated PCR) is the process by which a selected strand or segment of DNA is replicated various times. Each step in the process is vital for the process to occur efficiently. Purposes of Each Component: Template DNA: This serves as the template that allows for the entire reaction to occur. Without this, the designed primers won't be able to bind to anything and as a result TAQ polymerase wouldn't be able to do combine the dNTPs to generate a large growth in DNA strands. Primers: These are required to bind to the template DNA. These in a sense serve as a signal for the TAQ polymerase for where to start constructing a complimentary strand of DNA. These are pre-created to bind only to a specific sequence of DNA, the cancerous one in this case. A forward and a reverse primer are both needed because DNA polymerase can only work in one direction, so the same sequence must be made in opposite directions in order to satisfy the fact that DNA is composed of two strands: a leading and lagging strand. TAQ Polymerase: This is the enzyme that generates the new sequence of DNA using an original strand of DNA as a template. It binds to the primers, and collects the dNTPs that are floating around in the mixture to bind these complimentary to the given strand until the desired sequence is generated. Magnesium Chloride: This acts as a catalyse for the TAQ Polymerase, helping it do its job. It helps by making the process go by faster than it would without it. dNTPs: These are unpaired nucleotides that are floating around in the entire mixture. These are collected by the TAQ polymerase and form the nucleotide sequence that is complimentary to the template DNA sequence.
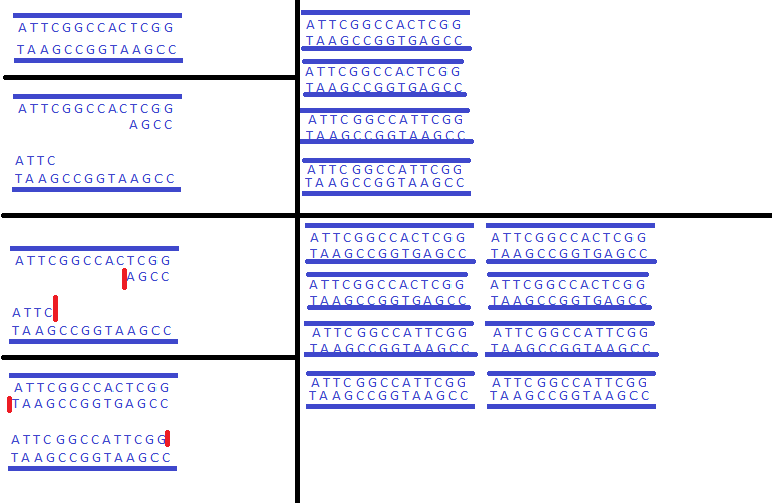
This helps us because only the cancerous DNA is replicated, whereas the original template DNA is not replicated at all. This is because the primers are designed to bind only to the cancerous sequences of DNA. This is because that specific type of cancer's DNA sequence was analyzed and a primer generated that binds to that analyzed, cancerous sequence. As such, when the primers detect this, they bind to those, and the DNA polymerase begins generated a sequence of DNA for this strand. This does not occur in the normal strand of DNA, simply because it doesn't have that specific sequence, and as such the primers won't bind to it. The mutation in the cancer allows for those specific primers to bind to that sequence, but the opposing, normal strand will not bind to it, even when the strand is continuously copied because the mutation isn't present there. As such, the cancerous sequence will be replicated, while the normal strand remains uncopied. This image shows exactly how the process works. In the beginning, at the top left, is the template DNA once it starts up. The second image down shows what occurs when the temperature is increased, then decreased. The template opens up allowing for each component of the PCR to go between the two strands, and once it's cooled down, the primers bind to each corresponding end of the DNA sequence that matches it. Keep in mind, that these primers were designed to bind only to the segments that have the corresponding cancer gene, otherwise it won't bind to them. What occurs next is when the temperature goes up, which is the next two images below. TAQ polymerase is activated, goes to the primer, and begins creating a segment of new DNA using the dNTPs that are floating around to create this DNA strand, as well as the Magnesium Chloride to act as a catalyst. This then creates the two new strands of DNA, and this continues, causing the DNA strand to grow exponentially over time. The image only shows what occurs with the cancerous DNA, however. The normal DNA strand is also replicated, but nowhere near as rapidly as the cancerous one due to the primers that are in the solution not matching up with that of the non-cancerous strand of DNA. It replicates at a non-exponential rate, leaving a far larger abundance of the cancerous DNA throughout the process.
| |||||||||