BME100 s2014:T Group7 L5
OUR TEAM
 Role(s) |
 Role(s) |
 Role(s) |
 Role(s) |
 Role(s) |
GETTING STARTED
DNA lab mission statement: In this lab students are to apply and improve their understanding of how we read DNA biomarkers. These biomarkers are crutial in diagnoising and treating various genetic defects and thus is increasingly relevent in biomedical engineering. During this lab students are to learn about various pieces of lab equiptment to collect both accurate and valid data sets. After the lab strudents will have an accurate idea of the functions of experimental equiptment such as the PCR machine and how these devices are chaning to improve the field.
How DNA is Analyzed
SYBR Green Dye Through the usage of a reagent named SYBR Green Dye our DNA sample will be stained green. By staining the sample DNA green we can then pass a beam of blue light through the droplet and the amount of green light emitted will allow a measurement of how much DNA is present. This occurs because the double stranded DNA, which provides the best results, will no longer be able to absorb the green light and will then reflect it.
Single-Drop Fluorimeter
Please insert picture of Fluorimeter
The purpose of the single-drop fluorimeter is to emit a blue light into a sample droplet. The device holds a slide in place upon which we place a droplet of our SYBR Green dye and DNA solution is placed. The light is then passed through the droplet which results in the emmition of green light.
How the Fluorescence is Applied to Measure DNA
Using a smartphone we are to take pictures of the flourimeter as it passes light through our sample droplet. The green light waves will be reflected by the SYBR Green dyed DNA and thus we will have an image to analyze on ImageJ which will show us how much green light was reflected. This data directly correlates with the amount of DNA present to reflect the light.
Procedures
Smart Phone Camera Settings
- Type of Smartphone: Apple IPhone 4s
- Flash: off
- ISO setting: 800
- White Balance: auto
- Exposure: auto
- Saturation: auto
- Contrast: auto
Calibration
1. Place the IPhone into the cradle.
2. Adjust the height of the Single-Drop Fluorimeter using plastic plates so that the droplet is reasonably centered on the camera.
3. Place the IPhone at a reasonable distance from the Fluorimeter, make sure to denoted this distance so that other readings are all at the same distance. We used 7cm for our first trial and 9cm for the second.
Solutions Used for Calibration
| Initial Concentration Calf Thymus DNA Solution (μg/mL) | Volume Calf DNA solution (μL) | Dilution volume SYBR Green solution (μL) | Final concentration, DNA in SYBR Green solution (μg/mL) |
| 5 | 80 | 80 | 2.5 |
| 2 | 80 | 80 | 1 |
| 1 | 80 | 80 | 0.5 |
| 0.5 | 80 | 80 | 0.25 |
| 0.25 | 80 | 80 | 0.125 |
| 0 | 80 | 80 | 0 |
Placing Samples onto the Fluorimeter
1. Place a new pipet tip on the pipet.
2. Pipet 80 μL of the SYBR Green solution in line with the blue light.
3. Discard the used pipet tip into a plastic cup and then attach a new pipet tip.
4. Pipet 80 μL of the calf thymus solution, the DNA solution being tested, and mix it with the SYBR Green droplet.
5. Follow the calibration procedures and prepare to take three pictures of the droplet.
6. Close the lid on the box to prevent extraneous light from entering the system then take the pictures.
7. When the pictures have been taken remove the droplet via the pipet and then discard the tip.
8. Adjust the fluorimeter slide to prevent cross contamination.
9. Repeat steps 1-8 until you have a total of three images for each of your concentrations.
Data Analysis
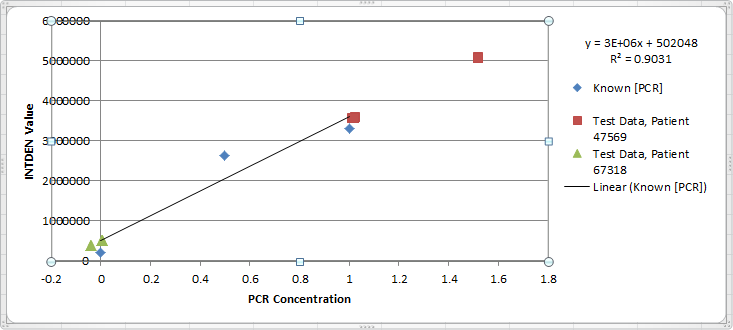
Representative Images


| Positive Control | Negative Control | 47569 - 1 | 47569 - 2 | 47569 - 3 | 67318 - 1 | 67318 - 2 | 67318 - 3 | |
|---|---|---|---|---|---|---|---|---|
| Raw INTDEN | 3241807 | 279609 | 5067434 | 3565695 | 3558270 | 516334 | 517861 | 380470 |
| 3250234 | 280103 | 5071287 | 3548796 | 3551278 | 516846 | 519123 | 374896 | |
| 3249871 | 279015 | 5067401 | 3575543 | 3561433 | 514675 | 516014 | 386431 | |
| Average PCR Concentration | 0.913253 | -0.07415 | 1.521795 | 1.021216 | 1.018741 | 0.004761 | 0.005271 | -0.04053 |
| Average Corrected PCR Concentration | 0.076104 | -0.00618 | 0.126816 | 0.085101 | 0.084895 | 0.000397 | 0.000439 | -0.00338 |