BME100 f2015:Group7 8amL6
| Home People Lab Write-Up 1 | Lab Write-Up 2 | Lab Write-Up 3 Lab Write-Up 4 | Lab Write-Up 5 | Lab Write-Up 6 Course Logistics For Instructors Photos Wiki Editing Help | |||||||
|
OUR COMPANY
LAB 6 WRITE-UPBayesian StatisticsOverview of the Original Diagnosis System In the final florescence test for the disease SNP, each team used PCR samples prepared in the previous lab of two patients that they were assigned to. There were a total of 34 different patients. In our group, a couple people worked to set up the box, phone stand, and fluorimeter, while others obtained our PCR samples and other materials. During the actual experiment, one person was responsible for pipetting, another for taking pictures, and another for keeping track of which samples we were testing. In order to prevent error, we took note of the order that we tested samples in and paid attention to which samples were being pipetted from. We also made sure that the slide appeared to have the appropriate amount of liquid on it. We used different locations of the actual slides in an attempt to prevent contamination, and we also made sure to eject used pipette tips for the same purpose. We took three images of each sample, as per the procedure, for redundancy. Because each subject also had three PCR samples, this made the chance for an outlier to alter our results much lower. In ImageJ we used the same area to calculate the number of fluorescent pixels in each image. Regarding the class data, final conclusions were correct for 13 of the 34 patients. It should also be mentioned that for 6 of the patients there was no test conducted, and the results were inconclusive for another 4 patients. When a final conclusion was made, the class was therefore correct in 13 of 24 cases. Our group had some ambiguity in our results, as we had one positive and two negative PCR tests for both patients. After deciding not to count on of the positive tests because it appeared to be an outlier, we came to the conclusion that both patients were negative. We assume that some mistakes, either human or device, were made to give us results that were contradictory. What Bayes Statistics Imply about This Diagnostic Approach According to the statistics, calculation 1 showed that the probability that the patient would get a positive final test conclusion given a positive PCR reaction was close to 1.00. This demonstrated that when one of a patients three different PCR tests came back positive, the patient was very often given a positive final conclusion. The rates of positive PCR reactions and positive final conclusions were also very similar. Therefore, the PCR results and final conclusions portray very similar pictures of the patients. Calculation 2 demonstrated that the probability that a patient would get a negative final test conclusion, given a negative diagnostic signal was also very high. Moreover, the rate of negative PCR tests and the rate of negative final conclusions were very similar. Across both calculations, the PCR results were highly correlated with the interpretation of those results in the form of final conclusions. According to the results from calculation 3, there was an almost identical rate of patients that developed the disease and patients who were given positive final conclusions. However, the probability that a patient would develop the disease given a positive final test conclusion was very low. While our test accurately predicted the rate of patients with the disease, it did not accurately identify patients that possessed the SNP. Calculation 4 shows a 10% disparity between the probability of a patient not developing the disease and the probability of a negative final test conclusion, meaning that there was a slight difference between the rate of healthy patients predicted and the actual number of healthy patients. However, the probability that a patient would not develop the disease given a negative final test conclusion was fairly high at between 0.50 and 1.00. Our test, while not completely accurate, was able to identify the majority of patients who did not possess the SNP. One source of human error that may have affected the Bayes values in a negative way is contamination, which could have occurred in either step. Many of the patients who did not develop the disease had positive PCR results. If DNA was accidently introduced to these samples, it could explain why these patients, who did not actually have the disease SNP, came up positive in these tests. Another source of human error could have been mistakes in pipetting. If different quantities of the sample were used in the fluorescence tests than intended, it would have an impact on the number of pixels that the software detected, and therefore would influence the Bayes results. One source of both device and human error in our group was using the phone stand, which was much too small for the cell phone that we used. Attempting to get the phone to fit in the right way meant that the position of our stand changed slightly during the course of the experiment. The device not functioning properly along with our attempts to reposition it may have resulted in error. Intro to Computer-Aided DesignTinkerCAD Our Design
Feature 1: ConsumablesList of Consumables: Reagents: primers, SYBR Green, buffer, and PCR mix, all readied in tubes. Pipetting tools: Specialized micro-pipette, discardable micro-pipette tips
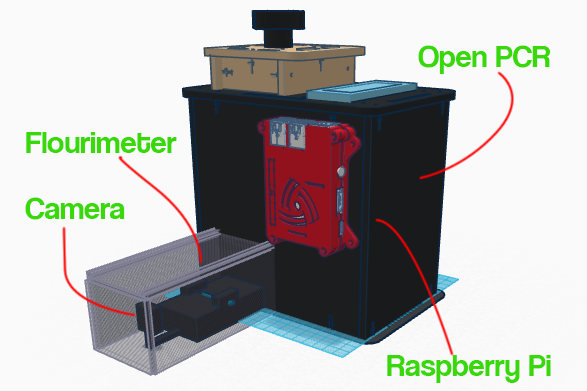
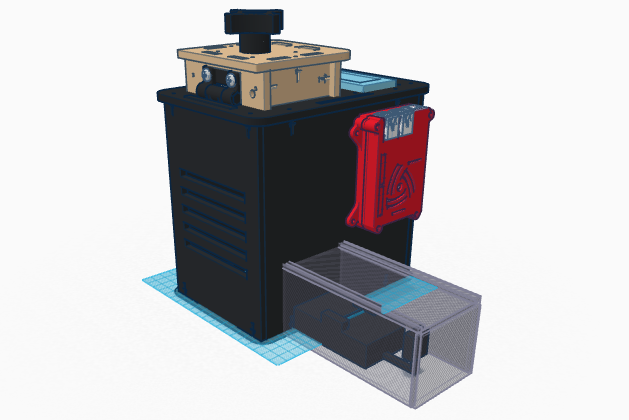
Feature 2: Hardware - PCR Machine & FluorimeterOur design follows the design of the OpenPCR but with a few additions such as the Flourimeter and the Raspberry Pi. One of the difficulties we had when analyzing the droplets to determine whether people were positive an negative was that although we kept the shape of the circle around the droplet in Image J constant, the droplets were not always fully enclosed in the circle. To prevent this we set up a camera connected to a Raspberry Pi, this way, the distance from the drop is constant and along with the same camera angle will allow for more accurate measurement. The Raspberry Pi is a cheap computer which runs several operating systems. With the addition of the Raspberry Pi into our product, the data processing of the droplet has been automated to facilitate the process. The Raspberry Pi comes set up specifically for the flourimeter and features a HDMI port so that it can be connected to most monitors for the display of all of the information
| |||||||