BME100 f2015:Group14 8amL6
| Home People Lab Write-Up 1 | Lab Write-Up 2 | Lab Write-Up 3 Lab Write-Up 4 | Lab Write-Up 5 | Lab Write-Up 6 Course Logistics For Instructors Photos Wiki Editing Help | |||||||
|
OUR COMPANY
LAB 6 WRITE-UPBayesian StatisticsOverview of the Original Diagnosis System DNA samples were collected from patients and distributed amongst 17 teams of 6 students each to diagnose a total of 34 patients with disease-associated SNP. The DNA samples were diluted with a buffer solution and SYBR Green, then a drop of this solution was placed inside the fluorimeter, and three images were taken of each sample. These three images acted as our repeated trials. To further reduce error, we compared the DNA samples to a positive and negative control, made sure to keep light out of the fluorimeter, and tested our patient DNA after calibrating the machine. Our data could have been affected by the quality of the camera (many images were slightly blurry), light leaking into the system, and there was a lot of room for human error with the pipetting system, and everything had to be completed one at a time. The material the fluorimeter was made of was a flimsy cardboard, and this could have had an effect on the validity of our results. There was also a lot of waste involved with the process, including numerous slides and even more pipette tips. However, image J worked well as a software to distinguish between the positive and negative samples. The class data displayed 6 blank sets, 16 negative results, 8 positive, and 4 inconclusive.
In Calculations 1 and 2 we calculated Bayes values very close to 1.00. This means that there is a close to 100% probability that a patient will get a positive final test conclusion given a positive PCR reaction, or a negative final test conclusion given a negative PCR reaction. This indicates good reliability of the individual PCR replicates for concluding if a person has the SNP disease or not. Our Bayes values for calculations 3 and 4 were quite low, close to 0.5, which indicates a 50% probability of a final test result correctly diagnosing a patient with the disease. A 50% chance of a correct diagnosis means that PCR was not reliable at all for predicting the development of the disease. Three possible sources of error that our data could have been affected by are the quality of the camera which in turn effects the quality of the image produced, the possibility of light leaking into the system as we where taking our image due to the lack of a reliable flap, and the possibility of human error while pipetting the reagents and DNA. Intro to Computer-Aided DesignTinkerCAD
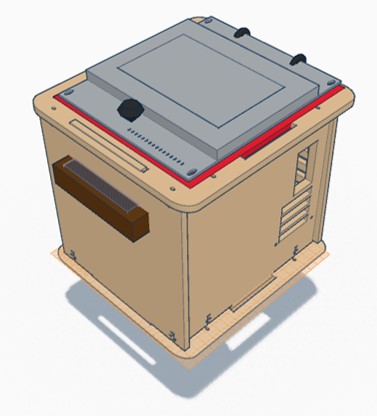
In our design PCR machine, we chose to focus on altering the lid and increase the ease with which it can be used and sealed, and the size of the machine itself. By adding additional sealant around the entire lid, we where able to improve upon the seal created without requiring additional operator effort. In addition to this, we included latches to ensure that the seal remains intact and the device insulated as the measurements are taken, and included a convenient handle to allow for easier manipulation of the lid. In addition to altering the lid, we nearly doubled the size of the device itself to allow for the machine to accommodate more samples.
Feature 1: Consumables
Example of the Multichannel Micropipette
Feature 2: Hardware - PCR Machine & Fluorimeter
The major weakness of the fluorimeter used in the previous lab was the camera system. Due to varying sizes of cell phones and the placement of their cameras it was difficult to properly align the lens with the edge of the test slides. Also, since the phone was loosely placed inside the fluorimeter box, maintaining the position of the camera was difficult. To remedy this, the new product's fluorimeter system will be equipped with a way to align and hold a cell phone camera in place outside of the fluorimeter box so that it is aimed properly at the edge of the test slides through a hole in the side of the box. A seal will made between the edge of the camera lens and the edge of the box to prevent light from entering the system.
| |||||||