BISC220/S11: Mod 3 Lab 10
Investigation of PARP-1
The apoptotic pathway in cells is regulated by a group of enzymes called caspases. Caspases are proteolytic enzymes that are activated by autoproteolysis (self cleavage ie capases 8 and 9) or cross proteolysis (ie. caspase 9 cleaves caspase 3). Autoproteolytic caspases are initiators - they are responsible for responding to the "death signals" by undergoing self-cleavage and as a result become active enzymes. The initiators then can cleave downstream caspases called effector caspases like caspase 3. Caspase 3 and other effector caspases are responsible for the degradation of many proteins that control important cellular pathways like DNA damage repair, integrity of the nuclear lamina and the cytoskeleton.
One such protein cleaved by caspase 3 is PARP-1 poly(ADP-ribose)polymerase-1. This protein is found in all human cells and it is involved in the DNA repair pathway. This enzyme adds branched ADP-ribose polymers to proteins like histones, DNA repair proteins and transcription factors so they function properly. The ADP-ribose polymers change the binding properties of the proteins. Without these proteins functioning properly regulation of transcription and DNA repair does not function properly.
The functional PARP-1 protein is 116 kD in size. When cleaved by caspase 3 there are two non-functional pieces - 85 kD and 24 kD. Today we will start the process of identifying when, after treatment with etoposide (VP-16), does the caspase cascade start effecting its downstream targets like PARP-1. To do this we will run control and treated whole cell lysates on an 8% polyacrylamide gel and blot this gel to a nitrocellulose membrane for Western blot analysis. Next week we will finish the Western by probing with a mouse anti-PARP-1 monoclonal antibody.
Protocol
Cells were treated with either 500 uM etoposide (VP-16) or as a control DMSO alone over a time course of 8 hours. Cells were incubated at 37°C during the experiment. Samples were taken at 2 hour intervals (0, 2, 4, 6 and 8). At each interval 1x106 cells were pelleted in a microcentrifuge.
- Resuspend the cell pellet in 33.3 μl of PMSF/glucose buffer (50mM glucose, 25 mM Tris, pH, 8.0, 10 mM Na2EDTA, and 1 mM PMSF)
- Add 16.7 μl of urea/SDS buffer (50 mM Tris, pH 6.8, 6M urea, 6% β-mercaptoethanol, 3% SDS, and 0.003% bromophenol blue)
- Place in ice bucket.
- For each sample obtain a clean insulin syringe.
- Slowly suck each sample up into the syringe and out again to shear the cells and break them open. Do this 5 times per sample.
- Heat the samples to 65°C for 15 minutes.
- Spin down the lysates for 2 minutes at top speed in a microfuge.
- Load 20 ul of each sample onto an 7.5% SDS-PAGE gel.
- Run the gels for 35-45 minutes at 200 Volts
Gel Loading:
Lane 1 — 20 µl of pre-stained MW standards (do not need to be boiled)
Lane 2 — 20 µl of Control 0 hours lysate
Lane 3 — 20 µl of Experimental 0 hours lysate
Lane 4 — 20 µl of Control 2 hours lysate
Lane 5 — 20 µl of Experimental 2 hours lysate
Lane 6 — 20 µl of Control 4 hours lysate
Lane 7 — 20 µl of Experimental 4 hours lysate
Lane 8 — 20 µl of Control 0 hours lysate
Lane 9 — 20 µl of Experimental 6 hours lysate
Lane 10 — 20 µl of Control 6 hours lysate
Lane 11 — 20 µl of Experimental 8 hours lysate
Lane 12 — 20 µl of Control 8 hours lysate
Finishing the SDS-PAGE & Starting the Anti-PARP-1 Western blot
Turn the power source controlling your gel electrophoresis to OFF. Remove your gel from the apparatus and separate the plates. Carefully cut off the wells at the top of the gel with a razor blade. Be careful not to tear the gel. Take it over to the blotting apparatus.
The holder for the blotting apparatus is color coded so the black side should end up facing the cathode (black electrode) and the clear side facing the anode (red). The sandwich of choice, therefore, will be made in the following manner, keeping all parts wet in the blotting buffer in the plastic container provided at your bench:
- Wear gloves when handling the nitrocellulose membranes. The membrane is white, and will be given to you sandwiched between two pieces of blue protective paper. Remove the top piece of blue paper. In pencil, label the top left corner of your piece of nitrocellulose (NC) with your initials. Be careful not to tear the membrane with the pencil tip. Pour some blotting buffer into your small plastic container. Immerse the NC in blotting buffer so that it is wet evenly.
- Place the blotting holder black side down in another plastic container after filling the container half full with blotting buffer.
- Open the blotting holder, wet one of the sponges with blotting buffer and place it on the black side of the holder.
- Wet 2 pieces of 3 mm paper and place them on top of the sponge.
- Wet your gel with blotting buffer in the small plastic container and place the GEL on top of the 3 mm paper. To make the left-most lane (Lane 1) of the gel come out on the left side of the blot, orient your gel so that Lane 1 (containing the stained MW ladder) is on the right side of the blot sandwich.
- Place your wet NC on top of the gel.
- Add 2 more pieces of moistened 3 mm paper on top of the NC. Use a broken plastic pipette like a rolling pin to gently “roll” out any air bubbles out from between the gel and the NC.
- Place a second moistened sponge on top of the 3 mm paper.
- Close the clear side of the holder, pushing the clasp down and along the top of the holder.
- Place the holder into the blotting tank so that the clear side faces the red pole and the black side faces the black pole.
- Two blots can be run in each tank. Place a frozen ice compartment into the tank. Fill the tanks so the buffer is up to the top of the gel. Connect the top of the tank tightly. Connect the power supply and run the blots at 100 volts for one hour. Be sure the stir bar is free to stir to keep the buffer cold. Watch the current. It should read 0.15-0.20 amps during the run.
- After an hour, disconnect the power and remove the holders. Remove the NC, rinse it with distilled water, and stain it with Ponceau S protein stain to see if the proteins were transferred properly to your membrane. Pour the 25ml provided of PonceauS onto your NM, placed in the small plastic container and rock back and forth for 15-30 sec. Pour stain back into 50ml conical tube (DO NOT DISCARD!). Rinse off the protein stain by pouring distilled water onto stained blot, rock, and discard water into sink. Look for approximately equal protein bands in all lanes loaded with cell lysates and look for presence of MW marker bands. Once you have determined that your transfer worked and that all lanes have approximately the same amount of protein, allow the membrane to dry a bit, then wrap it in plastic. Label it with your name and lab section. Store the NM in the refrigerator in a container provided by your instructor until the next lab.
Fluorescence Microscopy
Background on Fluorescence Microscopy
A substance is said to "fluoresce" when it emits light following excitation by light or another energy source. The fluorescence microscope is specially designed to filter light, since fluorescent molecules absorb and emit light at different wavelengths (Figure 1). The light source passes through one filter that allows only the wavelength that is absorbed by the fluorescent molecule of interest to pass through to the sample. This excites the fluorescent dye. A second filter allows only the emitted wavelength to pass through to the eyepiece and thus the eye of the observer.
Today you will view cells from the VP-16 timecourse that have been stained with the DNA binding dye Hoescht-33342. What will you expect to see in the VP-16-treated cells as compared to the control cells? We will observe the Hoescht fluorescence using a microscope filter set that is optimized for another DNA binding dye, DAPI, since Hoechst and DAPI have similar excitation and emission wavelengths. Your instructor will demonstrate how to use the fluorescence microscope during lab and how to take digital photographs of representative fields of the HL-60 cells. The files for these digital photos will be made available for you to use in your lab reports.
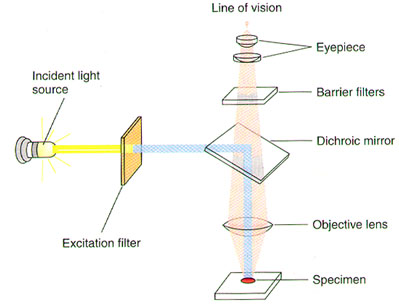
Figure 1. Optical System of a Fluorescence Microscope (Lodish H, et al. Molecular Cell Biology, p.140.)
Instructions for using the Fluorescent Microscope: Media:Using the Nikon 80i Fluorescence Microscope and Camera.doc
References:
Beltz, B.S. and G.D. Burd. (1989) Immunocytochemical Techniques: Principles and Practice. Blackwell Scientific Publications, Cambridge, MA.
Lodish H., Berk A., Zipursky S., Matsudaira P., Baltimore D. and Darnell J. (2000) Molecular Cell Biology, 4th ed. W.H. Freeman and Co., New York.
Watson, S.J. and H. Akil. (1981) Immunocytochemistry: Techniques, trials, and tribulations. Neurosci. Comm., 1: 10-15
Protocol
- 0.5x106 cells were pelleted and the supernatant discarded at each time point for control and etoposide (VP-16) treated cells.
- The cell pellets were mixed with 50 ul of 100 ug/ml Hoescht-33342 and incubated on ice for 5 minutes.
- The cells were then fixed with 10% formalin to preserve them for our use.
- The cells have been stored at 4°C
We will bring slides, coverslips, a P-20 pipette and tips to the microscope room. We will dispense 8 ul of fixed cells onto our side and cover it with a coverslip. We will then look at the cells to determine shape and size as well as degree of chromatin condensation. Hoescht dye emits at 492 nm and excites at 356 nm - similar to DAPI DNA staining.
Note: Not all cells will have condensed chromatin. It will be beneficial once we have taken a lot of pictures to go through them and determine the percentage of cells under each condition that have condensed chromosomes. I am expecting less than 50% for any time period.
Lab 8: Cell Culture
Lab 9: Apoptosis - DNA
Lab 11: Apoptosis - Protein 2
Lab 12: Imaging Presentations
Media Recipes