BISC219/F12: Gene Mapping Info
An Investigation in Classical (Forward) Genetics: Linkage Analysis, Mapping the Mutation, Complementation Testing, DNA Sequencing Analysis, Bio-Informatics
In Project 2, you will progress through the normal sequence of events in forward (classical) genetics. Forward genetics starts with finding a worm with an aberrant phenotype that is likely to be caused by a defect in a protein encoded by a mutated gene. Your overall goal is to locate and characterize the mutation that causes a dysfunction, seen as an aberrant phenotype so that we can better understand the functional significance of a gene, gene region, or a gene product in C. elegans and in other species. When we are able to make these structure/function connections by studying mutant worms and identifying the gene responsible for the defect, the main goal is not so much to understand defective gene function in worms, but rather, to be able to extrapolate the function of normal genes by seeing what their gene products are unable to do when altered. We are interested in worm genes because the genome of most eukaryotes astonishing similar. Many worm genes have homologues in other eukaryotic species, including Homo sapiens.
To do this involved forward genetics study we start with an interesting phenotypically aberrant worm. One of our main goals is to pinpoint the location of the mutation on a specific gene. In order to do this we first must find out through linkage analysis the relative location of the gene associated with the mutation: which chromosome is it on? We will be able to find out if the aberrant phenotype is caused by a mutated sex-linked or autosomal gene and, if autosomal, on which autosome the defective gene is located. Our eventual goal is to pinpoint the exact location of this mutation in a gene on a chromosome (to map the mutation). We will use complementation analysis to find out if the mutation we are studying is, possibly, a newly discovered one or a previously characterized defect. To conclude our function/structure analysis, we hope to identify the exact change in the bases of the gene that causes its product to function abnormally. We will find the exact mutation through DNA sequencing of the mutated gene and by comparing it to the wild type sequence. From this sequencing and bioinformatics work, we may be able to extract new information about functionally significant regions of the gene and its product. This structure/function relationship may be significant in a broader context than in just our nematode species. We will look for homologs/orthologs in other species.
To begin, you will first examine a plate of mutant worms and determine how they are different from wild type. You will then pick your mutants to a separate plate, confirm that they are true breeding (meaning that the worm has two copies of the defective gene), and begin our forward genetic analysis. The first step in identifying a new gene associated with an interesting mutant phenotype, is usually a long, tedious process that requires applying a mutagen (UV or some mutagenic chemical like EMS) to wild type worms and then looking through thousands of normal worms to find a good candidate mutant. To make this study easier for you, mutants have been found and isolated for you and you just have to move some to a new plate to ensure they are true breeding.
Once you have recovered your mutant and confirmed its phenotype as true breeding (by examining its progeny), you will next perform linkage testing: determining on which chromosome (linkage group) your mutation is located. This task is a prerequisite to mapping. You must find out the relative location of the gene responsible for the mutation (find the chromosome it is on) before you can determine the exact location of the mutation on a gene. Linkage Analysis is accomplished by determining the segregation behavior of your unmapped mutation relative to standard reference markers (e.g., mutations whose location is already known). Recall that unlinked mutations will segregate independently (your basic dihybrid inheritance as first observed by Gregor Mendel) whereas linked mutations will not.
In practice, linkage tests are performed using the following steps (where "d" (dpy) represents your recessive mutant tested with a set of reference markers "u" (unc)). The markers d and u must be visibly distinguishable. Since homozygous mutant males usually will not mate, the desired double heterozygote is constructed by mating males heterozygous for your dpy mutation but wild type for all other genes including the reference mutation (d/+;+/+) with hermaphrodites homozygous for the reference mutation unc (+/+; u/u). The genotypes of the F1 hybrids will be (+/d;u/+) and (+/+;u/+). We are only interested in the double heterozygote (+/d;u/+). The F1 hybrids containing only u are not useful. To select the (+/d;u/+) heterozygotes, we let 4 to 5 individual F1's self fertilize on their own individual plates (one on each plate). We score the progeny of the F1 individuals (the F2) for linkage. Only F1 worms which produce d/d homozygotes are scored, since those are the (+/d;u/+) parents. The d/d homozygotes should be found on 50% of the plates.
F2 progeny of each class are counted in the (+/d;u/+) plates: wild-type (+/+;+/+); d (d/d;+/+); u (+/+;u/u) and du double (d/d;u/u). If assortment is independent, progeny will be:
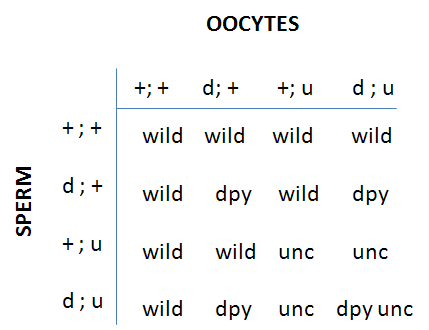
Or 9/16 wild; 3/16 d, 3/16 u; 1/16 du (that is our good old friend the 9:3:3:1 ratio)
On the other hand, if the markers are closely linked, double homozygotes (d u/d u) would occur only through two recombination events. Such an event might occur in both the sperm or the occyte and those recombinant gametes must come together. If the probability of a recombination event is p, and if the event produces wild-type and double mutant recombinant chromosomes, then the probability of getting the double mutant chromosome in an individual gamete is p/2. The chance of an individual gamete which is a (d u) recombinant combining with another (d u) recombinant is (p/2) x (p/2). If the map distance between 2 mutations is 10%, then the probability (P) of recombination occurring is p = 0.1. p/2 is 0.05 and (p/2) x (p/2) is 0.0025. Consequently only about 2 - 3 worms in a thousand will be double mutants if the genes were 10MU apart. That is significantly lower than the 63/1000 one would expect if the genes were not linked (1/16 of the progeny). Therefore, the test for linkage is usually the virtual if not complete absence of the double mutant class (d u/d u). You will use this first assesment to determine which chromosome the dpy mutation is on (the chromosomal location of each of the unc mutations is known).
Once you determined on which chromosome your dpy and unc are (for successful mapping they must be on the same chromosome), we will perform a two factor cross, which in C. elegans requires that we first construct a double mutant (du/du). As discussed above, the appearance of a double mutant in the F2 requires the union of 2 recombinant gametes -- a very rare event. It is unlikely therefore that we will find a double mutant among the F2 progeny if the mutants are linked. The probability of a recombination event having occurred on one of the two homologues is much better; that is, there will be many more progeny that are genotypically du/d+ or du/+u than du/du. It would exhibit the dpy mutation's phenotype and would segregate double mutants (du/du) as one quarter of its progeny. To find a double mutant for mapping, we choose 5 individuals of the Unc phenotype from the F2 linkage testing plate to self fertilize and look for the segregation of the du/du double mutant among their progeny.
To map, one crosses a homozygous double mutant hermaphrodite (du/du) with wild type males. The heterozygous F1 males will be mated to double homozygous recessive Dpy Unc hermaphrodites in a test cross and the number of F2 individuals of each different phenotype are then counted. As discussed above, four phenotypes will be observed: wild type, Dpy, Unc, and Dpy Unc. The map distance can be calculated any number of ways. You will determine map distances using the formula: RF (recombinant frequency) = the number of single mutants (dpy and unc single mutants totals) divided by the total number of worms counted * 100 (to obtain it in % recombinants and thus in map units).
Complementation, DNA sequencing of the mutated gene associated with the abberrant phenotype, and bioinformatics analysis will be described later.