BISC110/S11: Series 4 Lab 11 Enzymology
Series 4- Experiment in Gene Regulation & Enzymology
Gene Regulation — Enzyme Assay
The final series of experiments in BISC 110/112 will concentrate on the relationship between genes and proteins. In lab today, you will be using a eukaryotic model organism, budding yeast, to demonstrate how protein synthesis can be regulated at the genetic level.
Genetic Regulation of Enzyme Production in Yeast by Glucose Repression
Yeast are eukaryotic organisms that have been used for centuries to ferment beverages and to leaven bread. However, the usefulness of these simple, eukaryotic, unicellular microbes extends outside of the kitchen. Yeast can be used as powerful tools for genetic research because they have a short generation time, well-defined metabolic pathways, and many interesting mutants.
The most widely studied species of yeast is Saccharomyces cerevisiae, the organism with which you will be working with today. This is the species that is commonly used for baking and brewing. The medium commonly used to grow yeast is called YEP+2% glucose. It contains 1% yeast extract, 2% peptone (which are the YE, and the P in “YEP”), and 2% glucose. Peptone refers to partially hydrolyzed protein. When grown in liquid medium, the presence of the yeast cells causes the solution to appear cloudy.
The production of enzymes needed to metabolize all the different sugars that can be used by yeast is subject to genetic control within the cell. Yeast cells multiply the fastest in a medium containing 2% glucose because they can most efficiently use this monosaccharide in several metabolic pathways. When yeast cells are grown in YEP+2% glucose, the transcription of genes for enzymes that are needed to break down sugars other than glucose is repressed, so those enzymes are not produced within the cell. However, if the yeast cells are grown in a medium in which glucose is absent or present in a very low concentration, the genes for the production of enzymes that metabolize alternative sugars are de-repressed, and those enzymes are produced.
This type of genetic regulation of enzyme production is called glucose repression, and it is common in both prokaryotic and unicellular eukaryotic organisms. The exact mechanism of glucose repression varies depending on the type of cell, but the basic reason for this repression is universal. By repressing the production of certain metabolic enzymes when sufficient glucose is present in the environment, the cell prioritizes the use of the most efficient nutrient (glucose) first, and does not waste energy producing enzymes required to breakdown other sugars that may not be present or that require more energy to catabolize.
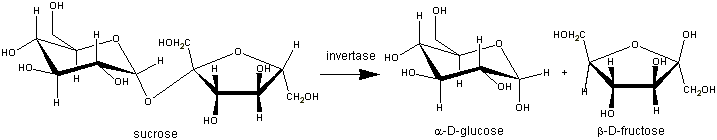
You should review the differences in the structures of glucose and other carbohydrates, looking in particular at the difference between glucose and sucrose. If sucrose is added to YEP medium and glucose is reduced, the yeast must derepress production of an enzyme called invertase (also called sucrase) that catalyzes the breakdown of sucrose into glucose and fructose in order to facilitate yeast growth.
Your goal in lab today is to demonstrate this concept that yeast make invertase only when there is an insufficient amount of glucose in the environment. In order to determine whether or not the genes for invertase production are being repressed or expressed, you will perform an invertase assay in lab today. Using this assay you will determine the amount of invertase produced by normal, "wild type" yeast grown in YEP+2% glucose (normal glucose medium) and in YEP+0.05% glucose (minimal glucose medium).
A strain of yeast that is unable to make invertase due to a genetic mutation is also available for your investigation. The inability to produce invertase should adversely affect the growth of the mutant cells in a medium that contains sucrose as the sole sugar. The mutants should also grow poorly in a medium containing only minimal glucose because, after the small amount of glucose available is exhausted, growth will cease. However, the mutants should grow almost as well as the wild type cells on YEP+2% glucose. Why?
Based on genetic regulation of invertase production by glucose repression, form a hypothesis about the relative amounts of invertase that the wild type and mutant strains of yeast will make when grown in media containing 2% glucose or in 0.05% glucose. Remember that the mutant yeast strain cannot make invertase due to a defective invertase gene.
Invertase Assay
Our invertase assay uses a yellow reagent called DNS (43.9 mM 3,5-dinitrosalicylic acid and 250mM NaOH) that changes to an amber color in the presence of reducing sugars, such as glucose and fructose. Reducing sugars can act as mild reducing agents due to an aldehyde group. Because sucrose is not a reducing sugar, the activity of the invertase enzyme can be determined by the intensity of the amber color in the reaction tubes after time is allowed for sucrose hydrolysis and subsequent addition of DNS reagent. Invertase catalyzes the conversion of sucrose into glucose and fructose, which results in the DNS reagent turning amber. The intensity of the amber color can be measured in a spectrophotometer as absorbance, which is proportional to the amount of glucose and fructose produced during hydrolysis.
You will use DNS reagent to assay the activity of both purified invertase and any invertase that has been produced by the two yeast strains grown in either 2% or 0.05% glucose. To test the purified invertase indirectly, you will add some to a solution of sucrose, allow time for the reaction and measure the amount of glucose and fructose that has formed. This amount will be compared to a reaction tube with sucrose but no invertase (the negative control).
To test invertase produced by yeast, you will use wild type and mutant strains that have been grown overnight in YEP+2% (normal) glucose and YEP+0.05% (minimal) glucose. You will add the yeast to a solution of sucrose, and the invertase that was made by the yeast will convert the sucrose in each reaction tube to glucose and fructose, which will then be detected using DNS.
Before you begin your experiment, you need to get organized. Enzyme assays require precise timing since enzymes are not consumed by the reactions they catalyze. Consequently, enzymes will continue to drive a reaction until the reaction has reached equilibrium. Instead of waiting for equilibrium, you will perform your assays so that each test tube will react for exactly 15min. This means you will add sucrose to the invertase that is in the test tube, staggering the time you start each reaction. Precisely 15min after you have started each invertase catalyzed reaction, you will end it by adding the DNS reagent. The DNS reagent raises the pH in the reaction tube and inactivates the invertase.
Procedure for Invertase Assays
- Obtain 8 x 13mm test tubes, and label them 1–8 with a Sharpie® permanent marker.
- Into tube 1 put 0.6mL of deionized water. This tube will be used to blank the spectrophotometer.
- Into tube 2 put 0.5mL of 6.0mM glucose and 0.1mL of deionized water. Glucose is a reducing sugar. Since the assay measures the amount of reducing sugars produced in each reaction, this will be your positive control. If after running your reactions, there is no color change in this tube, then you will know that there is a problem with the DNS reagent or the temperature of the 95°C water bath.
- Into tube 3 put 0.1mL deionized water. Eventually it will have sucrose in it also and will serve as your negative control since the DNS reagent is not expected to react with sucrose. If you see a color change for this tube after you perform your reactions, then you will know that something about the assay itself is not working.
- Into tube 4 put 0.1mL of purified invertase enzyme. The tube with the purified invertase (1mg/mL in pH6 potassium phosphate buffer) is on the instructor’s bench labeled as "enzyme". Be sure to mix the solution of enzyme just before you take any out. After the assay is completed, you will be able to compare the color change in tubes 3 and 4 to know how much sucrose was converted to glucose and fructose by the purified invertase enzyme.
- Tubes 5–8 will be used to assay the invertase that has been produced by the wild type and mutant yeast cells. The cells have been grown for you and can be found on your bench in four 13mm tubes. Each yeast strain has been grown in both YEP+2% glucose and YEP+0.05% glucose. The cells grow almost 10X better in normal glucose than in minimal glucose, as you'll be able to see from the turbidity of the tubes. In order to assay approximately the same number of cells, pipet 0.5mL of 2% glucose-grown wild type cells into tube 5, and 5mL of 0.05% glucose-grown wild type cells into tube 6. Next pipet 0.5mL of 2% glucose-grown mutant cells into tube 7 and 5mL of 0.05% glucose-grown mutant cells into tube 8.
- Harvest the cells by spinning tubes 5–8 in a clinical (benchtop) centrifuge for 4 min (the tubes do not have to be covered; they won't break). You must turn the timer knob all the way to the right before bringing it back to 4 min. to get the centrifuge to start and to go at full speed. Remember that the centrifuge must be balanced and that the volumes in the four tubes are not identical. Use your timer as the centrifuge timers are not always accurate.
- After the spin, the yeast cells will be pelleted at the bottom of the tubes. Do not use a pipette to remove supernatant. Invert each tube over the sink, in one quick turn of your wrist so the the tube is completely upside down. The pellet will NOT fall out! Without turning the tube back upright, touch the inverted tube lip to a paper towel to remove a few more drops of media. When the pellet is drained, turn the tube upright. If you are confused about how to do this, ask your instructor to demo the technique. Add 2.5mL of deionized water to each tube to wash the cells. Vortex each tube to resuspend the cells in the water, then spin tubes again for 5min in the centrifuge, and discard supernatant as before.
- Wash the cells a second time with another 2.5mL of water. Vortex and spin them in the centrifuge a third time for 5min. The supernatant above all four of the cell pellets is discarded as described previously. Note the color of the supernatant after the last wash. It should be colorless (not yellow). If it is still yellow, then wash all four cell pellets one more time. When cells are adequately washed and drained add 0.1mL of deionized water and resuspend the cells.
- You are now ready to begin your assays. Take your rack of 8 tubes, a 1mL pipette, your timer, your 3mM sucrose solution and your notebook to one of the 55°C water baths. Put your tubes into the water bath. Then, every 30sec, add 0.5mL of the 3.0mM sucrose solution to the next tube and return that tube to the water bath. Follow the timing listed in the table below.
| Tube# | Contents | Add 0.5ml of 3mM Sucrose |
Add 3ml of DNS reagent |
|---|---|---|---|
| 1 | water | do not add | 14:00 min. |
| 2 | glucose | do not add | 14:30 min. |
| 3 | sucrose | 00:00 min | 15:00 min |
| 4 | invertase | 00:30min. | 15:30min. |
| 5 | WT yst/2.0% glucose | 01:00 min | 16:00 min |
| 6 | WT yst/0.05% glucose | 01:30 min | 16:30 min |
| 7 | MUT yst/2.0% glucose | 02:00 min | 17:00 min |
| 8 | MUT yst/0.05% glucose | 02:30 min | 17:30 min |
13. After each addition, you should vortex the tube briefly to mix the contents, then move the tube to another place in your rack in the 55°C bath to avoid accidentally adding sucrose twice to the same tube. After your sucrose additions are completed, please move away from the water bath to give other groups room to work.
14. In your lab notebook, you should set up a table like the one above that indicates the contents of each tube but you should leave room to record the absorbance readings that you will get at the end of the assays, and to record your observations about the tube's colors. You could also use this time to think about the other parts of today's lab, and begin to get those things ready.
15. Each reaction should proceed for 15min exactly. When the time is almost up, take a 5mL pipette and a green Pipetman that doesn't leak, your timer, your notebook, and the DNS reagent from your bench to the water bath. Stop each reaction by adding 3mL of DNS reagent to each tube at 30sec intervals, so that each tube will have reacted for 15min. Please use caution so that you do not add DNS reagent to someone else's tubes.
16. DNS reagent must react under alkaline conditions and the color change becomes evident only after the samples have been heated for 10min at 90–95°C. Place all your reaction tubes into one of the hot water baths that are contained in the chemical hoods in the lab. Set your timer to count down for 10min, so you will know when the time is up.
17. Carefully remove your tubes from the hot water bath. There are test tube holders near the bath to help you with the hot tubes. CAUTION: Take care not to burn your fingers in the hot water or with the steam, and please check that you are removing your own tubes from the bath.
18. Let your tubes sit on the bench for 5min until they are cool to the touch. Transfer the contents (just pour them) into a new set of tubes. Then spin tubes 5–8 for 2min in a clinical centrifuge. The cells in these tubes must pellet completely and the solution must not look cloudy after this spin. DO NOT discard the supernatant after this spin.
19. Before you measure A540nm as amount of amber color (the amount of product, reducing sugars, caused by the breakdown of sucrose to glucose and fructose, look at your rack of tubes and compare their colors. Which tubes are brown/orange rather than yellow? There should be three amber tubes, two of which are controls. Look at the table describing the contents of each tube and decide whether or not your controls worked. Does an expected set of control reactions provide confidence in your ability to evaluate the results of your experiment?
20. Wipe the outside of each test tube with a Kimwipe™. Using tube #1 to blank the spectrophotometer, read the absorbance of each reaction at 540nm and record all data in your lab notebook.
Data Analysis
Look at the absorbance (A540nm)value of tubes 5 and 6 comparatively. What can you conclude about gene regulation of invertase? Is your hypothesis supported? Does it appear from your data that the invertase gene is somewhat down-regulated by 2% glucose or is the gene completely turned off? All the other tubes serve some sort of control function. Make sure you understand what you learn from each control that allows you to use the comparison in color between tubes 5 and 6 to answer your experimental question: is invertase gene expresion in yeast under glucose repression control? Remember that we used the invertase mutant, not because we were particularly interested in showing that the invertase gene in these cells encoded a non-functional protein, but because comparing the difference in color between the mutant grown in 2% glucose vs. 0.05% glucose served as a control that let us be sure that the difference in reducing sugar level seen in tubes 5 and 6 were because of invertase gene regulation and not because of something else that we hadn't considered. Did your mutant control results (tubes' 7 and 8 A540nm values) give you that assurance? How so?
Laboratory Clean up
- Pour the DNS solutions in the waste jar in the hood. You may want to wear gloves to avoid yellow stains on your hands.
- Discard the empty test tubes from the invertase assay in the glass recycle container at the front of the lab.
Assignment
- Prepare for a Lab Practical Skills Assessment in Lab 12. You may download a set of practice problems and a key to those problems here: Media:110_extra_practice_problems1.doc and Media:110_Lab_practical_practice_key_new.doc. You may also come into the lab when there is NOT a lab going on (labs run every weekday afternoon, Wed. morning, and Thurs. evening) to practice using the microscope.
- Prepare to turn in your lab notebook for grading (10pt) in Lab 12.
- Answer the following questions (5 points) and hand them in to your instructor at the beginning of Lab 12. Download a Word document form of these questions here:Media: Lab_11_Assignment_Questions2.doc
Why is it advantageous for an organism to be able to turn particular genes on or off in response to various environmental conditions?
Name a genetic control mechanism that yeast use to regulate the production of invertase. Explain how your results for the invertase assay of the wild type yeast demonstrate this mechanism. You did the same invertase assay on yeast with a defective invertase gene (a mutant that could not make functional enzyme). What does this control assay on the mutant tell you?
The person at the bench across from you performed the invertase assays just as you did, but she forgot the second water wash of her yeast cells. The following table compares her results to yours. How might her mistake in performing the experiment lead to these results?
| Sample | Her Results A540nm |
Your Results A540nm |
|---|---|---|
| water | 0 | 0 |
| glucose | 0.84 | 0.82 |
| sucrose | 0 | 0 |
| sucrose + invertase | 0.57 | 0.6 |
| WT yst/2.0% glucose | 0.51 | 0.09 |
| WT yst/0.05% glucose | 0.61 | 0.60 |
| MUT yst/2.0% glucose | 0.50 | 0.05 |
| MUT yst/0.05% glucose | 0.05 | 0 |
Link to Labs in this Series and the Last Series
Lab 8: Photosynthetic Pigments
Lab 9: The Hill Reaction
Lab 10: Variables Affecting the Hill Reaction
Lab 11 Gene Regulation/Enzymology