BISC110/S11: Series 1 Lab 3 Dilutions & Tetrahymena Investigation
PART I: Dilutions
A helpful animation of dilution making.
Many solutions will be provided to you this semester as stock solutions. A stock solution is a solution that has been prepared with a greater concentration of solute than is needed for a biochemical procedure, so it must be diluted to the proper concentration(s) before use. Consequently, it is very important that you master the concepts and techniques of preparing dilutions early in the semester. A dilution is made by taking an aliquot (a measured portion) of a stock solution, and adding to it an aliquot of a solvent (usually water). This creates a new solution with a lower concentration of solute than was contained in the stock solution. Since the solute concentration of the stock solution is known as well as the volumes of stock and solvent, it is possible to calculate the concentration of solute in the dilute solution. The dilute solution is always less concentrated than the stock or starting solution. There are several ways to describe and prepare dilutions. Explanations are provided in this section as well as a laboratory exercise to introduce you to dilution preparation.
A. VOLUME-TO-VOLUME DILUTIONS
1. Ratio Method
This type of dilution describes the ratio of the solute to the total volume of the dilute solution. For example, to make a 1:10 dilution of a 1M NaCl solution, you would mix one part of the 1M solution with 9 parts of solvent (probably water), for a total of ten parts. If you needed 10mL of the 1:10 dilution, then you would mix 1mL of the 1M NaCl with 9mL of water yielding a total volume of 10mL. If you needed 100mL of the 1:10 dilution, then you would mix 10mL of the 1M NaCl with 90mL of water yielding a total volume of 100mL. The new concentration of NaCl in both solutions is determined by multiplying the stock concentration by the dilution ratio:
1M NaCl x 1/10 = 0.1M NaCl
2. Direct Calculation Method
Sometimes it is necessary to use an aliquot of one solution to make a specific volume of a more dilute solution. To calculate the volume of the more concentrated solution that will be needed to prepare the new solution, you can use the formula:
V1 x C1 = V2 x C2
V1= volume of more concentrated solution needed to make the new solution (usually the unknown)
C1= concentration of starting solution
V2= final volume of new solution
C2= final concentration of new solution (must be less than C1)
For example, if you have 2.5 ml of a 1M solution of NaCl and you want to make 3 ml of a 0.33M solution of NaCl, you can use the formula to calculate how much of the 1M solution you will need.
V1 x C1 = V2 x C2
(V1)(1M) = (3mL)(0.33M)
V1 = (3mL) (0.33M)/1M
V1 = 1mL
The calculations show that you will need to use 1mL of the 1M solution. Since the diluted solution is to have a final volume of 3mL, you can calculate the volume of solvent by subtracting V1 from V2 : 3mL – 1mL = 2mL. Therefore, adding 2mL of solvent to 1 ml of a 1M solution of NaCl will yield 3mL of a 0.33M NaCl solution.
3. Serial Dilutions
Serial dilutions involve the preparation of successive dilutions that vary the concentration of the solute by a constant factor, so that each successive dilution is a multiple of the previous dilution. For example, in ten-fold serial dilutions each successive dilution is one tenth of the previous dilution yielding dilution ratios of 1/10, 1/100, 1/1000, and so on. Serial dilutions may be used to prepare a series of standard concentrations for a biochemical assay, or to dilute a very dense culture of microorganisms to a concentration suitable for counting. It is very important to prepare serial dilutions carefully since each successive dilution will magnify the effect of any previous error.
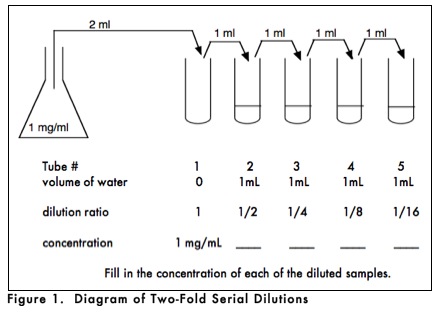
How would you prepare a dilution series? As shown in Figure 1, you could begin with a stock solution of 1 mg/ml and set up a series of dilutions in which the dilution ratio relative to the stock solution progressively changes by one-half. In this instance, the concentrations of the successively diluted solutions would be 1/2, 1/4, 1/8, and 1/16 of the original concentration. If you needed 1 ml of each dilution, you could set up a series of 5 test tubes, with the first one empty and the rest with 1mL of solvent. You would then pipette 2mL (twice the volume needed for your test) into tube 1. Then transfer 1mL of that volume to tube 2, making a 1/2 dilution since there was 1mL of water in tube 2. After mixing the contents of tube 2, half that sample (1mL) would be transferred to tube 3, making a 1/4 dilution since the 1 ml of water that was already in tube 3 will dilute the incoming 1/2 dilution by 1/2 (1/2 x 1/2 = 1/4). This procedure is repeated until the last tube is reached, which will contain 2 ml of a 1/16 dilution of the stock solution.
B. Prepare Serial Dilutions of Cobalt Chloride and Cupric Chloride Stock Solutions
Each pair will use either the 0.11M cupric chloride dihydrate (CuCl2•2H2OH20) or the 0.26M cobalt chloride hexahydrate (CoCl2•6H2OH20) stock solution prepared in Lab 1 to prepare a series of 4 dilutions. Each dilution in the series will differ from the previous dilution by 1/2, so the dilution ratio for each tube is 1/2, 1/4, 1/8 and 1/16. Calculate the concentrations of cupric chloride or cobalt chloride that the tubes will contain after the dilutions have been prepared, and record them in your lab notebook.
- Label four microcentrifuge tubes with the dilution ratios: 1/2, 1/4, 1/8 and 1/16.
- With a 200 μL micropipette and clean tip add 100 μL deionized water to each tube. Discard the tip as instructed by your instructor.
- Using a clean pipette tip, add 100 μL of stock solution to the tube labeled 1/2, close the lid, and mix by inversion (not vortexing).
- Use a clean pipette tip to add 100 μL of the 1/2 dilution to the tube labeled 1/4; mix.
- Use a clean pipette tip to add 100 μL of 1/4 dilution to the tube labeled 1/8; mix.
- Use a clean pipette tip to add 100 μL of 1/8 dilution to the tube labeled 1/16; mix.
- Using a clean pipette tip, remove 100 μL of solution from the 1/16 tube, and discard it in the sink. You now have four tubes containing 100 μL of four different concentrations of stock solution. Observe the color intensity of the four solutions and record in your lab notebook. Also, observe the relative volumes of the four solutions. If they are different from each other, then inaccurate pipetting has occurred. Save these dilutions for use in Part II, the toxicity study below.
C. Calculating Final (Effective) Concentration
It is often important to know the effective (final) concentration of a reagent in a particular reaction, such as, when you perform the toxicity assay of your heavy metal in PART II. If you find that the heavy metal is toxic to Tetrahymena at some concentrations, but not at others, you need to be able to calculate and report the concentration range of non-toxicity or toxicity. If you combine equal volumes of Tetrahymena and a dilution of your cobalt or cupric chloride solution on the slide, you have, effectively, diluted the heavy metal by 1/2, resulting in an final concentration of half the concentration in the serial dilution tube that you used in the experiment.
In order to calculate effective (final) concentration, you must know the concentration of the reagent of interest in each of the working dilutions. In the experiment described below, concentration of the working dilution would be the concentration of the cobalt or cupric chloride in each of the serial dilutions (in moles this time, but concentration could be expressed in any units of concentration, such as mg/ml or %(v/v or wt/v). You must also know the ratio of the amount of the reagent of interest (cobalt or cupric chloride) to the total volume of the final dilution. In the case of the experiment in PART II below, you are going to use 20 μL of live Tetrahymena together with 20 μL of 0.11 M copper chloride or 0.26 M cobalt chloride. The ratio is 1 part metal to two parts total, so the dilution factor is 2. What if you used only 5 μL of metal and 20 μL of Tetrahymena? In that case, there is one part metal in a total of 5 parts of final dilution so the concentration of the metal would be reduced 5 fold (multiply the conc. of the metal x 1/5).
Making a Working Dilution of a Stock Reagent When You Know the Final Concentration
Sometimes you know the final concentration of a reagent that you want in a reaction and you need to make a working dilution of a stock to achieve that final concentration in an experiment. For example, if you read a paper that said that Tetrahymena were fixed in a final concentration of formaldehyde of 0.4% (v/v). You have a bottle of stock 100% formaldehyde. You know that you are going to put 20 μL of Tetrahymena on a slide and combine them with 10 μL of a dilute formaldehyde solution. How are you going to make that dilute formaldehyde solution (working solution) from the 100% stock formaldehyde to give you a final concentration of formaldehyde of 0.4%?
Answer: There will be one part of working formaldehyde solution of unknown concentration in a total of three parts (2 parts of Tetrahymena + one part of formaldehyde) so you will be diluting the working solution of formaldehyde by a factor of 3; therefore, you want the working solution to be three times more concentrated than 0.4%, or 1.2%. Since you are only going to use 10 μL of it, you could make 1 ml of 1.2% working dilution of formaldehyde from the 100% stock. Using V1 x C1= V2 x C2, where V1 is the unknown. (V1)(100%) = (1 ml)(1.2%). Combining 12 μL of 100% formaldehyde with 888 μL of water would give you 1ml of the appropriate working dilution of formaldehyde to achieve a final concentration of formaldehyde of 0.4%.
PART II. Determine Toxicity of Cobalt Chloride or Cupric Chloride on Tetrahymena pyriformis
Each pair of students will use either the cobalt chloride or copper chloride solution to carry out a Tetrahymena toxicity study. Combine 20 μL of live Tetrahymena with 20 μL of 0.11 M copper chloride or 0.26 M cobalt chloride in a clean microfuge tube. What is the concentration of the heavy metal in moles? Wait 1 minute, and then add 20 μL of those Tetrahymena (from the microfuge tube) to a glass slide, add a cover slip, and observe, using the microscope. Is your copper chloride or cobalt chloride toxic to Tetrahymena at the concentration used? How do you define toxicity? Record your observations about behavior and morphology changes in Tetrahymenaafter exposure to heavy metal. How do you know what is normal behavior and morphology? If you decide to use lack of movement as your criterion for toxicity, how will you determine what is self-propelled movement vs. water flowing on your slide? Should you include a control? YES! What would the appropriate control(s) be? Test the remainder of the dilutions of cobalt or copper chloride solution and determine if concentration is important in the toxicity of your compound. Make a table in your lab notebook to record your data. Don't forget to calculate final concentration (in moles) of the heavy metal every time you use a different dilution and record that information in your lab notebook. Use final (effective) concentration, not dilution factor, in your data table or axis labels in a graph made from the data and in your results analysis.
PART II. Design your own experiment to test the role of a cytoskeleton element, a phosphatase, or selectivity in phagocytosis in Tetrahymena pyriformis
We should have time today for you to work with your partner to design your own experiment to answer one of several questions that may have come up from your observations or from your reading about Tetrahymena phagocytosis in the last few weeks. Your instructor will explain your options and she/he will give you a research paper or other information related to the question that you and your group chooses to explore. The information given should help you figure out an appropriate concentration of your test variable to use in your experiment. Related research papers should also help you formulate a hypothesis. Begin this project by reading the backgound information in the next lab description at Lab 4: Variable Testing in Tetrahymena. Before you leave today, have a rough draft of your experimental protocol (including showing how you will make all working solutions of reagents) approved by your instructor. If necessary, you and your group should meet with your instructor in the next few days to finalize your plan to address your question with a well designed and controlled experiment to be performed in LAB 4.
Laboratory Cleanup
- Place used serological pipettes tips down in pipette canister to soak. Place used micropipette tips in the trash.
- Rinse out all other glassware with water and invert on a paper towel on the lab bench to dry. Ordinarily, the 13mm tubes are disposable, but do not discard them in today’s lab.
- Clean the objective lens of your microscope using only lens tissue (NOT Kimwipes®) starting with the lowest power (4x) and working up to the highest. Make sure that there is NO oil on any lens.
- Rotate the 4x objective lens into the viewing position.
- The binocular head must be rotated into the storage position, to protect the ocular lenses from damage. Loosen the setscrew on the right, rotate the head 180°, then tighten the screw. Turn off the microscope light.
- Have your instructor check your microscope, before returning it to the cabinet (with its plastic cover on).
- Place the stock solutions of cobalt chloride or cupric chloride in the tray next to the sink near the instructor’s table.
- Put all used microscope slides and cover slips in the glass disposal box.
Assignments
- Finalize your experimental protocol for your self-designed experiment and have it approved by your instructor.
- Write a results section of a scientific report on your findings in the Tetrahymena toxicity experiment. You should have a narrative portion and at least 2 data displays with proper legends integrated into the narrative. (One figure should be a photomicrograph or comparative photomicrographs done as a composite single figure and the other data display could be a table or a graph). Refer to the appropriate parts of the Scientific Writing Guidelines in the Resources section of the wiki for help. The Gronlien article we discussed is a good model for how to write a results narrative and what to include in figure legends. It is also shows you how to use photomicrographs as data.
Dilution Problems:
Please solve the following dilution problems on a separate piece of paper (not in your lab notebook) to hand in at the beginning of the next lab. You can download a .doc form of these questions at Media:Lab_3_110_Dilution_Problems.doc
- You have been given a stock glucose solution with a concentration of 100mg/mL. To perform the glucose assay in lab today, you must dilute the 100mg/mL stock solution of glucose to concentrations of 80mg/mL, 60mg/mL, 40mg/mL, and 20mg/mL. You need to prepare 1mL of each dilution. Explain how you would make the 4 dilutions from the glucose stock solution.
- The molecular weight of glucose is 180g/mol. To prepare a stock solution of glucose, you dissolve 1.8g of glucose in water to a final volume of 1.0L. This stock solution is then diluted as follows: 50mL of the stock solution is added to 450mL of water, and then 1.0mL of the diluted solution is added to 99mL of water. What is the µM concentration of the final solution?
- You have been given 10 ml of a Tetrahymena culture that contains 8 x 1010 cells/ml. How would you prepare 20ml of a culture containing 4 x 105 cells/ml using a portion of the original culture and sterile media?
- You are writing a research report and you need to describe the concentration of methyl green stain on the Tetrahymena you show in a photomicrograph. The concentration of methyl green in the bottle was 5% and you added 100 μL of it to 9.9ml of an acidic buffer to make a working dilution of stain. You then combined 10 μL of that diluted stain with 40 μL of your diluted cells and put a few microliters of that mixture on the slide from which you took your photo. What was the effective (final) concentration of the stain?
- A published journal article you read says that, "To determine the prey concentration that gave optimal phagocytotic uptake,dose/response curves for both latex beads and deciliated T. thermophila were obtained (data not shown). The optimalconcentrations were 106 beads ml–1 and 105 cells ml–1".
If you wanted to do a similar experiment using 0.1ml of beads mixed with 0.9 ml of Tetrahymena, how would you dilute the stock beads (1010/ml) to make 0.2ml of an intermediate conc. of beads (working dilution) so you would achieve the appropriate final dilution of 106 beads ml–1?
Links To Other Labs in this Series
Lab 1: Boot Camp
Lab 2: Scientific Investigation in Tetrahymena
Lab 3: Dilutions & Scientific Investigation in Tetrahymena
Lab 4: Variable Testing in Tetrahymena
Lab 5: Scientific Writing & Effective Figure Design