BISC110/F12: Series 3 Experiment 10 Hill Reaction Variables
Lab 10 Investigation of Factors Affecting the Hill Reaction
Introduction
In this lab, you and your partner will explore, in a self-designed, controlled experiment, one factor that may affect the rate of the Hill reaction. The following are some of the factors that you may choose to investigate:
Temperature
Consider the normal growing conditions which spinach usually tolerates. How would varying the temperature between 0°C and 45°C affect the rate of photosystem II electron transport in its thylakoid membranes? Would the temperature itself affect the behavior of DCPIP, our artificial electron acceptor? In terms of the practical aspect of this experiment there are several things to think about: you will need to equilibrate each tube for a few minutes at each temperature in a water bath before adding the thylakoids; you will have to dry the test tube between readings; therefore, you may want to read the absorbance every 20s or 30s instead of every 15s.
Light Intensity
In our study, we are interested in knowing how light intensity changes will influence the electron transport rate in photosystem II.
Would you expect a linear increase forever in electron transport rates with increasing light intensity? Why/why not? You can vary the light intensity by varying the setting of the intensity knob of your illuminator or by changing the distance between the light source and the thylakoids. Just make sure to uniformly illuminate your thylakoids and make sure that you are not also increasing the temperature of the thylakoids, a possible confounder.
Light Wavelength
Based on knowledge of the pigments that are involved in absorbing light for photosynthesis, how would the wavelength of light affect the rate of the Hill reaction?
You can vary the wavelength of light by covering the fiber-optic lens with colored (red, green, blue) polycarbonate filters (Roscolux®). Make sure to keep the light intensity constant as you vary the wavelength. Measure the % transmittance spectra of the colored filters (Appendix C, Program #6) to get a better idea of their efficacy as wavelength-selective light filters. To save time, you may do this while you are preparing your thylakoid extract.
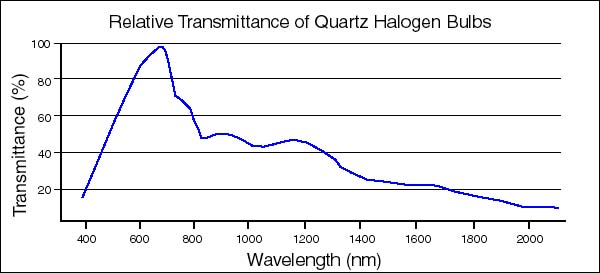
Figure 1. Relative Transmittance of Quartz Halogen Tungsten Lamps. The 150-watt projection lamps of our fiber-optic illuminators have this type of spectral energy distribution curve. The transmittance peak (%Tmax) is 675nm.
Inhibitors/Uncouplers
The inhibitors act either by binding to a component of the membrane or altering membrane structure by denaturation and/or solubilization. If the electron transport is blocked, or the membrane is disrupted, there will be a decrease in electron transport and in the reduction of DCPIP. For inhibitors, the effect is concentration-dependent. For chemicals altering membrane structure, inhibition is usually time-dependent as well as concentration-dependent.
The uncouplers act by "uncoupling" electron transport from its rate-limiting dependence on the photophosphorylation machinery. Uncouplers may have various mechanisms of action, but the end result is the dissipation of the proton gradient. We cannot measure the production of ATP, but we can measure electron transport using the dye, DCPIP. When the reactions relating to electron transport and the creation of an H+ gradient are uncoupled, electron transport proceeds at a faster rate. Therefore, the reduction and therefore color change of DCPIP occurs faster.
We shall supply stock solutions of several compounds that act as inhibitors and/or uncouplers of electron transport depending upon their concentrations:
2.0 M NH4Cl (ammonium chloride)
10–3 M DCMU (dichlorophenyldimethylurea)
(Note that DCMU can irritate the eyes, skin and respiratory tract. It is used as an herbicide, so it is advisable to wear gloves when handling DCMU.)
5% Triton-X 100 (a detergent)
You may test concentration as a variable by testing different dilutions of the stock solution of these compounds to find the concentration at which they no longer affect the Hill reaction. Remember that some compounds may act as uncouplers or inhibitors depending on their concentration. Inhibitors and uncouplers are usually added in 100µL aliquots to insure that the concentrations of all of the other ingredients in the reaction tubes are altered as little as possible. Calculate the effective concentrations of your inhibitor or uncoupler (the concentration while the reaction is occurring).
Procedure
Make table(s) and flow charts in your lab notebook for the data you will obtain from your self-designed and instructor approved experiment. Be sure you have adequate controls and replicate reactions and that all of the final volumes in your reaction tubes are the same. Calculate how to make any dilutions and know final concentration of any crucial material in the reaction before you come to lab. Set up a table in which you can keep track of the times vs. the absorbance for each reaction.
When you and your partners have collected all of your data, plot the A580 data (y-axis) versus time in seconds (x-axis) for the different treatments (you can plot several or all data sets on one graph or plot each treatment on a separate graphs). When making these graphs, do not include points where substrate is limiting. Add regression lines to each of your plots. Include the regression equations. The slopes represent the Hill reaction rate, which in this artificial system is the electron transport rate of photosystem II in photosynthesis. Plot the rates (y-axis) against your variable (x-axis) as directed by your instructor. Discuss your graphs with your partners to determine how your data answers your experimental question. Does the data support your hypothesis? If not, should you rethink your hypothesis or consider confounding variables that you didn't account for in your experiment? Show your graphs to your instructor and discuss your findings with her/him before you leave lab.
Laboratory Cleanup
- Clean all glassware with water, centrifuge tubes with ethanol and place pipettes in the wash bucket at the end of your bench.
- Dispose of any hazardous waste (reactions containing DCMU) in the container in the hood.
- Other assay reactions can be discarded in the sink.
- Place test tubes in the glass disposal container.
- Return the color filters to the instructor’s bench.
Assignment
- A complete scientific paper on your experiment today is due on the date designated in the Calendar or by your lab instructor. This assignment must be done individually. It is important that you discuss your results in terms of how the factor that you chose to investigate affects the electron transport rate and the coupling of electron transport with the photophosphorylation of ADP and/or the role of the H+ gradient in photosynthesis. Please compare your results to other investigations published in the primary literature as part of the Discussion section of your report. Refer to the Guidelines for science writing information found in the Resources section of the Wiki.
- During next week's lab, you will participate in a writing workshop focusing on the photosynthesis paper. INDIVIDUALLY, prepare an outline of your introduction and discussion. Bring hard copies to class so they can be exchanged in peer review. The outlines of the introduction and discussion sections should contain citations. A Word document list of references relating to each variable is available on your section's Sakai site, instead of the pdf files. You should search for these articles using the Wellesley College Library Research Guides or a reference database such as PubMed or Google Scholar. You are encouraged to search for additional articles that may be useful outside of those on this list. AS A GROUP OF FOUR, prepare a few PowerPoint slides that display your figures and captions. Please include (a) a representative trial from each condition graphed against time and (b) mean rates of electron transport vs. your variable. Provide your instructor with an electronic version of this PowerPoint before lab. Be prepared to describe your results informally to the class for a group peer review.
- Please come to the workshop with any questions you might have about this final paper.