Artificial Skin, by Katie Geldhart and Timothy Ma
Introduction
Artificial skin is the term used to describe any kind of material designed and used to replace naturally growing skin. The primary application of such materials right now revolves around treating individuals that have suffered severe tissue loss from disease or trauma (e.g. skin cancer, burns). Alternatively, artificial skin is now being used in some places to treat patients that suffer from foot ulcers [2][11]. Labs have also developed in vitro skin samples to be used for cosmetic and medical testing. This provides a cheaper and more ethical approach for testing surface reactions to topical treatments [1][11].
The most important goals of current artificial skin technologies are to provide protection from infection, dehydration, and protein loss after severe tissue damage. Each year, over 2 million people in the United States are treated for burns [2][11]. Artificial skin is employed to seal a wound quickly to minimize the amount of fluid lost by the patient and to limit bacterial intrusion. Integra is a brand of artificial skin commonly used in medical facilities today.
Overview of the Skin
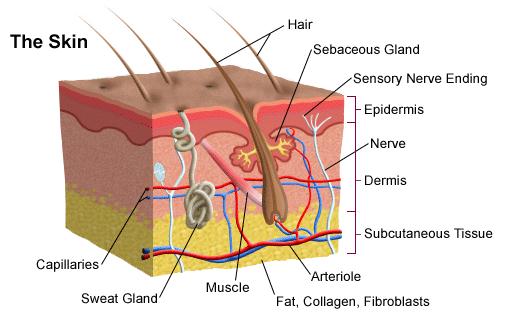
Human skin is comprised of two primary layers, the dermis and the epidermis. The epidermis is the outermost layer and comprises of keratinocytes that vary in levels of differentiation. There are no blood vessels present in the epidermis which means nutrient transport occurs from the dermis below [2][11]. The dermis contains several structures important to skin function: hair follicles sweat and oil glands, and nerves are all found within the dermis [2][11]. Epithelial keratinocytes also originate from within this layer.
History of Skin Grafts
3000-2500BC, India: Skin is allegedly transplanted by Hindus from the buttocks to repair mutilated ears and noses.
1442, Italy: An allogenous skin graft was performed by Branca de Branca who used a man’s arm skin to transplant the nose of the man’s slave onto himself.
1597, Italy: Branca’s methods were published by Tagliacozzi.
1804, Italy: First successful autograph in sheep, done by Baronio.
1823, Germany: Full thickness autologous skin grafts were successfully performed by Bunger. Done by transplanting skin from the patient’s thigh to their nose, a procedure inspired by the original Indian records.
1869, Switzerland: First epidermic pinch graft by Reverdin.
1871, England: Pollock proposes skin grafts for burn treatment.
1875, Scotland: Full-thickness skin grafts become an official clinical practice.
1886, Germany: Thin split-thickness skin grafts used by Thierch.
1970’s, United States: Cryopreserved skin used for human allografts, viable epithelial sheets grown in vitro by Rheinwalk and Green.
1979, United States: Burke and Yannas successfully test the first artificial skin (Integra) on a patient [1][11].
1981, United States: Cultured epithelial grafts used in burn patients.
1998, United States: First tissue engineered skin (Apligraf) is approved by the FDA.
2001, United States: Dermagraft, Orcel, Composite Cultured Skin are FDA approved [4][11].
2002, United States: Integra is FDA approved for treatment of severe burns [4][11].
All time points provided by reference [3]unless otherwise noted
Treatment
Extent of damage to the dermis is often an important factor in determining the skin’s ability to repair and recover from damage because it carries more functions than the epidermis. Current artificial skin techniques for burn treatment typically provide a scaffolding to promote regeneration of the dermis. An autograft is then performed to replace the epidermis. Alternatively, artificial skin has been made to replace both layers of the skin as in the case of certain small wound and foot ulcer treatments.
Skin Grafts
A patient requires a skin graft if damage to the tissue is too acute for self-repair. Most patients receiving skin grafts are burn victims. There are six degrees of burn severity but only 1st through 3rd degree burns are concerned when discussing skin grafts. A first degree burn only affects the epidermis whereas a second degree burn extends downward into the dermis. Third degree burns go through the epidermis, dermis, and the hypodermis. Burns classified as “severe” are either 2nd degree with >10% coverage or 3rd degree with >1% coverage [2]. Skin grafts are also performed on patients who experience tissue loss from cancer eradication and plastic surgery [1]. Skin tissue required for a graft can be acquired from three possible sources. It could come from the patient for an autograft, another human for an allograft, or an animal for a xenograft [2]. Allografts and xenografts can only be used as temporary wound coverings; the patient’s body will reject the grafts within a period of 7 to 10 days for an allograft and 3 to 5 days for a xenograft [2]. There are two primary types of autografts: split-thickness grafts and full-thickness. The choice between the two methods depends on the condition of the wound. Split thickness grafts take only a section of the dermis and the epidermis from the donor site to transplant to the wound site [9]. The donor site remains somewhat intact and a good amount of skin can be harvested but STSG’s suffer from a number of drawbacks. Split thickness grafts demonstrate more fragility, especially when placed over areas lacking any sort of underlying soft tissue support and contract significantly during healing [9]. There is also a noticeable discrepancy in color between the grafted skin and the surrounding tissue. Full–thickness skin grafts take out the entire dermis and epidermis layers from the donor site but they look more natural in the wound site and often provide improved skin flexibility compared to the STSG. For this reason, FTSG are commonly used for hand and facial skin graft procedures. But because FTSG also contain components of the dermis, it has higher metabolic needs and require optimal environmental conditions to survive [9]. There are several draw backs to autographs, the first of which is the removal of donor skin from another part of the body. Both STSG and FTSG procedures leave scarring in both the donor and wound sites and the patient must be placed under anesthesia [9]. Additionally, when skin is removed from one area to place in another, both areas must be treated to allow proper healing. Where autografts fail to be effective is when patients suffer significant skin loss and there are insufficient donor sites to harvest skin from. An allograft or xenograft may be used to temporarily dress the wound but a more permanent solution is required.
Synthetic Skin Treatments
Synthetic substitutes for skin may be considered as a permanent solution or as a temporary wound dressing if there is a lack of graftable skin or permanent solutions. These grafts can reduce microbial intrusion and reduce the loss of fluids and proteins [9]. Some xenografts fall under synthetic treatments. Since 2001, a total of 4 skin repair devices have been FDA approved. These include Dermagraf, Orcel, Integra, and Composite Cultured Skin [4][11]. Dermagraft and Apligraf approved in 1998 are used for the treatment of foot ulcers and contain living cells. Orcel is used to cover and aid in wound healing specifically in donor sites on burn patients. Composite Cultured Skin is specifically for children with recessive dystrophic epidermolysis bullosa and also contains living cells. Integra is the only one of these treatments currently FDA approved for treatment of severe burn areas. Integra is comprised of shark cartilage and bovine collagen [4][11]. In the most common procedures, the damaged tissue is removed and a layer of artificial skin is placed over the wound. When using Integra, the collagen matrix provides a scaffold onto which dermal cells begin to grow. Once blood vessels sufficiently vascularize the neo-dermis, the layer of silicon covering the matrix is removed and autologous epidermal cells are spread over the neo-dermis. Glycosaminoglycan within the matrix signals for cells to begin producing their own collagen and over time, a natural ECM replaces the artificial layer [2][11]. Orcel is actually a biolayered cellular matrix that contains both dermal and epidermal cells. This means that if the cells are able to vascularize and take to the area, only one surgical treatment is required. The cells within Dermagraf, Apligraf, and Orcel aid in speeding up the healing process by secreting growth factors and proteins native to skin tissue. This promotes regeneration of skin in the wound.
Complications and Failure
The two largest risks with artificial skin are the onset of infection and rejection by the immune system. Lack of blood flow to the implant can lead to cell death which facilitates breeding of bacteria. This would especially be problematic in procedures using either full-thickness skin grafts or bilayered cellular matrices as portions of these tissues remain longer without blood supply. Allografts and xenografts are often the only choice for burn patients who do not have sufficient skin remaining for an autograft. In these cases there is a high rate of immunological rejection. In addition to infection and rejection, fluid buildup between the wound site and the transplanted skin can prevent the skin from properly adhering. If this occurs, the procedure must be repeated. Skin adhesion is also a problem when using Integra if the epidermis does not fuse correctly to the implant[11]. Grafts can also fail if they are subjected to excessive stresses from movement and sheering. Movement disrupts the adherence of the graft to the patient and occurs frequently when the graft site is over flexor or extensor surfaces [9].
Ongoing Research
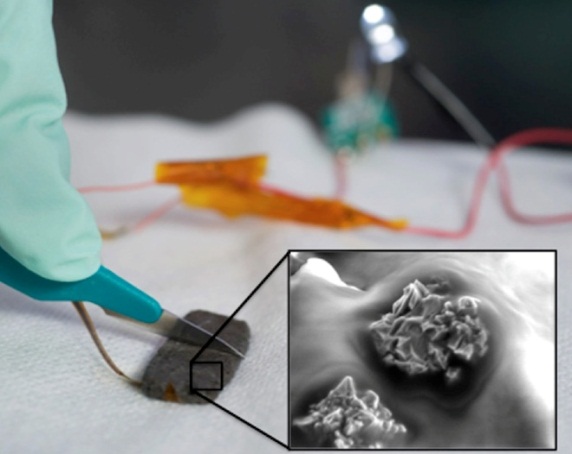
While artificial skin has aided significantly in skin regeneration, there still remain several areas for improvement. A minimum of two surgical treatments are required for severe burn patients using artificial skin. Matriderm is an upcoming treatment for burn victims similar to Integra. Unlike Integra, the dermis matrix and epidermis cells are able to be put in place in one surgical session [5][11]. Research is now also being done to attempt to develop bacteria-resistant skin cell cultures that can be used in artificial tissue. Ideally, this would allow in vitro replication of a patient’s own genetically modified skin cells. These cells could then be put into the artificial matrix for bacteria-free growth [6][11]. Other advances have developed in the application of the cells to the matrices. Dr. Fiona Woods has produced a “spray on skin” called Cellspray [7][11]. This method takes healthy cells from the patient and creates a suspension culture of the cells. The cells are then distributed by spraying them evenly across the matrix. It is believed that this method will be beneficial because the suspension cell culture can be produced much faster (5 days) than traditional methods which require formation of cell sheets (21 days). Though this procedure has not undergone clinical trials, it was controversially used on burn victims in conjunction with traditional methods in the Bali bombings in 2004 [7][11]. Further testing is required before this treatment can officially be deemed viable. Synthetic skin is being developed in hopes of enabling the sensation of touch to non-living structures. Flexible pressure transducers may eventually allow us to create an electronic “skin” with the ability to mimic touch. This currently has applications in robotics including advanced prosthetic limbs [8][11]. There is another development in synthetic skin where researchers have developed a polymer material that is flexible, electrically conductive, and self-healing. The material can be cut and heals simply by pressing the material back together [10]. Though this is a novel breakthrough, it is still a long way from becoming a viable source of artificial skin. The material is flexible but it cannot stretch the way skin does.
References
[1] Dantzer, E., Queruel, P., Salinier, L., Palmier, B., & Quinot, J. F. (2001). Integra, a new surgical alternative for the treatment of massive burns. Clinical evaluation of acute and reconstructive surgery: 39 case. Annales De Chirurgie Plastique Esthétique, 46(3), 173-189.http://www.ncbi.nlm.nih.gov/pubmed/11447623
[2] Roos, D. (2012). Skin grafts. Retrieved 02/29, 2012, fromhttp://health.howstuffworks.com/skin-care/information/anatomy/skin-graft.htm
[3] Heman, A. R. (2002). The history of skin grafts. Retrieved 02/29, 2012, fromhttp://findarticles.com/p/articles/mi_m0PDG/is_3_1/ai_110220336/
[4] Recently approved devices. (2009). Retrieved 02/29, 2012, fromhttp://www.fda.gov/MedicalDevices/ProductsandMedicalProcedures/DeviceApprovalsandClearances/Recently-ApprovedDevices/ucm083520.htm
[5] Haslik, W., Kamolz, L. -., Nathschläger, G., Andel, H., Meissl, G., & Frey, M. (2007). First experiences with the collagen-elastin matrix matriderm® as a dermal substitute in severe burn injuries of the hand. Burns, 33(3), 364-368.http://www.sciencedirect.com/science/article/pii/S0305417906002282
[6] Singer, E. (2007). A better artificial skin. Retrieved 02/29, 2012, fromhttp://www.technologyreview.com/biotech/18059/
[7] Spray on skin. Retrieved 03/01, 2012, from http://www.biotechnology-innovation.com.au/index.html
[8] Boldand, J. J. (2010). Flexible electronics: Within touch of artificial skin. Retrieved 02/29, 2012, fromhttp://www.nature.com/nmat/journal/vaop/ncurrent/full/nmat2861.html
[9] Weber, Stephen M., MD. "Split-Thickness Skin Grafts." Split-Thickness Skin Grafts. N.p., 20 Jan. 2011. Web.
[10] Wogan, Tim. "Self-Healing Synthetic ‘Skin’ Points Way to New Prosthetics."Wired.com. Conde Nast Digital, 10 Nov. 2012. Web. http://www.wired.com/wiredscience/2012/11/synthetic-skin/
[11] Geldhart, Katie. "Artificial Skin, by Katie Geldart." OpenWetWare RSS. N.p., 2012. Web. http://openwetware.org/wiki/Artificial_Skin%2C_by_Katie_Geldart
Image Sources
[A] http://www.childrenshospital.org/az/Site784/mainpageS784P0.html
[B] Wogan, Tim. "Self-Healing Synthetic ‘Skin’ Points Way to New Prosthetics."Wired.com. Conde Nast Digital, 10 Nov. 2012. Web. http://www.wired.com/wiredscience/2012/11/synthetic-skin/
