20.109(F13): Mod 1 Day 6 Lipofection and paper discussion
Lipofection
Introduction
DNA can be put into mammalian cells in a process called transfection. If you wanted to make a mouse cell fluoresce green, you could transfect it with DNA carrying the EGFP open reading frame, a promoter directing transcription of EGFP and a signal sequence for polyadenylation of the mRNA. The promoter tells the cell that the EGFP sequence should be transcribed by RNA polymerase. The polyadenylation sequence assists in the export and stability of the mRNA so that it gets translated by the ribosome. The coding sequence tells the ribosome which amino acids should be joined together.
Mammalian cells can be transiently or stably transfected. For transient transfection, DNA is put into a cell and the transgene is expressed, but eventually the DNA is degraded and transgene expression is lost (transgene is used to describe any gene that is introduced into a cell). For stable transfection, the DNA is introduced in such a way that it is maintained indefinitely. Today you will be transiently transfecting your cultures of mouse embryonic stem cells.
There are several approaches that researchers have used to introduce DNA into a cell's nucleus. At one extreme there is ballistics. In essence, a small gun is used to shoot the DNA into the cell. This is both technically difficult and inefficient, and so we won't be using this approach! More common approaches are electroporation and lipofection. During electroporation, mammalian cells are mixed with DNA and subjected to a brief pulse of electrical current within a capacitor. The current causes the membranes (which are charged in a polar fashion) to momentarily flip around, making small holes in the cell membrane that the DNA can pass through.
The most popular chemical approach for getting DNA into cells is called lipofection. With this technique, a DNA sample is coated with a special kind of lipid that is able to fuse with mammalian cell membranes. When the coated DNA is mixed with the cells, they engulf it through endocytosis. The DNA stays in the cytoplasm of the cell until the next cell division, at which time the cell’s nuclear membrane dissolves and the DNA has a chance to enter the nucleus.
Today you will lipofect several DNA samples into your mouse embryonic stem cells. As a positive control, you will transfect one sample with a plasmid encoding full-length EGFP. This plasmid will cause any transfected cells to fluoresce green. Next time we will measure fluorescence of your positive control to assess the success rate of the transfection.
You will also be transfecting two experimental plasmids, one of which you have been constructing for weeks. The EGFP coding sequence on these plasmids is truncated at either the 5’ or 3’ end of the gene. Cells expressing only one of these truncated EGFPs should not fluoresce green – but we'll check! The plasmids provide a wonderful tool for studying recombination since a cell will fluoresce green if it has been transfected with both plasmids and has recombined the genes to regenerate a full-length EGFP. Finally, you will be using the truncated EGFP plasmids to investigate the effect of double strand breaks on the frequency of recombination, as well as the effect of a DNA repair inhibitor. The inhibitor, Compound 401, specifically inhibits the NHEJ pathway, which may or may not influence the frequency of HR.
Elaborate mechanisms for coping with DNA breaks have evolved since these forms of DNA damage are so dangerous for the cell. You should (re)familiarize yourself with these mechanisms by (re)reading the excellent review by Thomas Helleday that you can find in the References section of the Module 1 frontpage. You should also check out the animations of repair mechanisms that are linked there. These animations were made by Justin Lo, a class of '08 Course 20 student and a former UROP student in Professor Engelward's laboratory.
One kind of DNA damage with particularly catastrophic consequences for the cell is double stranded breaks. The broken ends of the DNA must be correctly repaired (literally here re-paired) without loss of any encoded information. The model for such repair is called to the Szostak model, named after the person who first described it. According to this model, illustrated below, a gap in one chromosome is repaired through an interaction with its homologous chromosome. The repair first requires “homology searching” and invasion of the gapped DNA (blue) into the undamaged copy (red). The undamaged DNA serves as a template for missing sequences, which are copied and then resolved as indicated by the open triangles on the figure below. Resolution can lead to exchange of sequences flanking the original double strand break, resulting in recombination of genetic information. Alternatively the integrity of the original genetic material is preserved when the repaired strands resolve without crossover.
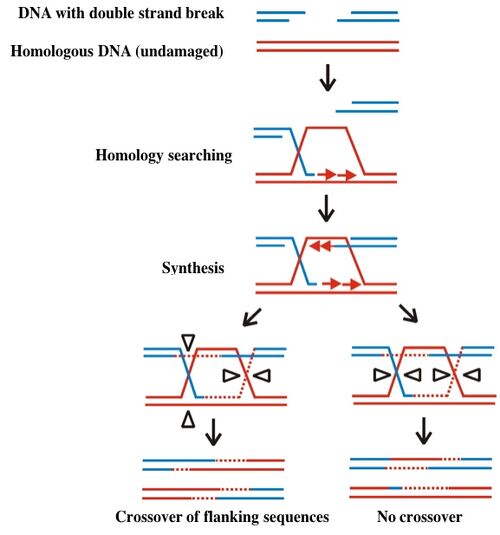
Different, and far simpler means of repair are possible if the broken ends of the DNA can be held together, either through base pairing of the overhangs or through the chromatin structure surrounding the damage. Your investigation today will assess recombination rates for different DNA lesions and conditions for homology searching.
Protocol
While half of the class works in tissue culture, the other half will have a journal article discussion.
Part 1: Lipofection
All manipulations are to be done with sterile technique in the TC facility.
Timing is important for this experiment, so calculate all dilutions and be sure of all manipulations before you begin.
In anticipation of your lipofection experiment, one of the teaching faculty plated 8x104 cells in a pregelatinized 24-well dish 24 hours ago. A special media formulation without antibiotics was used.
A schematic for your experiment is shown below.
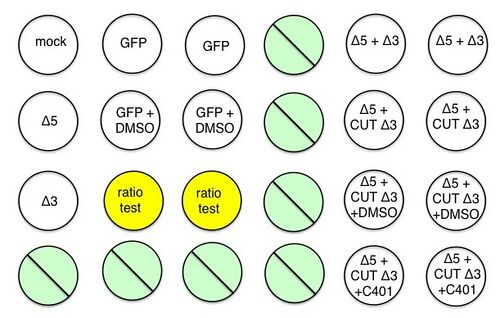
Please adhere to the schematic exactly, as some wells have been pre-treated with inhibitor C401 or its carrier solvent DMSO (as a control). The two wells marked in yellow are your opportunity to test a specific hypothesis related to template and donor DNA availability.
For each lipofection in a single well you will need
DNA: 0.1 μg of Δ3)and/or 0.1 μg of Δ5 in OptiMEM, 50 μL total Carrier: 2.5 μL Lipofectamine 2000 diluted in OptiMEM, 50 μL total Note: All DNA stocks are at ~ 0.05 μg/μL.
- 1. Dilute enough carrier for 18 lipofections. Let the dilution sit in the hood undisturbed for at least 5 minutes but not more than 30.
- 2. Three of the control lipofection conditions are completed just once. In those conditions you’ll need 50 ul of DNA + OptiMEM. Use the table below to help you calculate the appropriate volumes to use in these conditions.
| Tube | DNA/lipofection | Volume DNA | Volume OptiMEM |
|---|---|---|---|
| mock | NA | NA | 50ul |
| Δ5 | 0.1 ug | ||
| Δ3 | 0.1 ug |
- 3. For the lipofection conditions to be done in duplicate, you’ll need a total of 100-300 ul of DNA diluted in OptiMEM, depending on whether that DNA condition is repeated across different media formulations (such as with and without DMSO) or not. By making one lipofection cocktail that is later divided between replicates, you can be confident that each replicate was treated identically.
| Tube | DNA/lipofection | Number of media conditions | Volume DNA (total) | Volume OptiMEM (total) |
|---|---|---|---|---|
| GFP | 0.1 ug | 2 | 8 | 192 |
| Δ5 and Δ3 | 0.1 ug of Δ5 and 0.1 ug of Δ3 | 1 | ||
| Δ5 and digested Δ3, equal | 0.1 ug of Δ5 and 0.1 ug of digested Δ3 | 3 | ||
| Δ5 and digested Δ3, unequal | ? ug of Δ5 and ? ug of digested Δ3 | 1 |
- 4. You should now have exactly 7 eppendorf tubes, with volumes varying from 50 to 300 ul. Add an equal volume of diluted lipofectamine ('carrier') to each eppendorf (i.e., 50 ul if the tube has 50 ul). Pipet up and down to mix.
- 5. Incubate the DNA and lipofectamine cocktails at room temperature for 20 minutes. To allow the DNA/carrier complexes to form, it is important that you do not disturb the tubes during this incubation. During this time, aspirate the media from the cells in your 24-well dish, wash the wells with 0.5 ml PBS, and then put 0.5 ml of fresh media on the cells. Be sure to add the appropriate media (w/ or w/out DMSO or C401) to each well! The PBS and media can be aliquoted with a 5 ml pipet -- or a 2 ml pipet if you're feeling lucky... and agile.
- 6. After the 20 minute incubation is over, use your P200 to add 95 ul of the appropriate DNA:lipofectamine complexes to each well. Since the carrier can be toxic to the cells it’s a good idea to gently rock the plate back and forth after each addition.
- 7. Return the plate to the 37°C incubator.
- 8. Approximately 48 hours after performing the lipofection, you and your partner will collect the cells and analyze their fluorescence by flow cytometry.
Part 2: Journal article discussion
While half of the classes in tissue culture, Professor Bevin will lead a journal article discussion in the lab. Before class, please read the 1998 paper from the Takeda lab through the text associated with figure 3 (about 3.5 pages in).
Scientific papers are dense and often time-consuming to read and understand, but with practice, you will find strategies that improve your comprehension efficiency. Here's one tip to get you started: when reading newly reported results, be sure to refer to the associated figures frequently, because visual information is often easier to take in than purely verbal descriptions.
Several terms and approaches in the Takeda lab paper may be unfamiliar to you. In lecture and/or lab we will discuss topics such as Southern blotting and creating knockout mice. You should feel free to search for information about these or additional topics in advance -- even Wikipedia can be a good start! At the same time, don't feel the need to understand every detail presented in the paper. We expect you to leave the discussion knowing much more than you need to know coming into it.
Each discussion will take place in 56-711.
- First group 1:45-2:45 pm
- Second group 3:15-4:15 pm
DONE!
For next time
- Sign up for flow cytometry time here. Groups will have to stagger their arrival at lab in order to keep the number of groups and samples at the flow facility manageable.
- To prepare for your Module 1 summary, compose a page on the diagnostic digest phase of your experiment.
- Besides the diagnostic gel itself, presented in a well-labeled figure with an appropriate figure caption, you might also include a plasmid map or other schematic here.
- Below the figure(s), describe the experiment and interpret its outcomes in concise language.
- Feel free to review Logistics -- Guidelines on Formatting and Length and Content Guidelines, as needed.
- Your Module 1 methods section is due Monday, Oct 7th by 5 pm. Please review the assignment description for submission guidelines.