20.109(F12):Mod3 OrangeTR Pre-proposal
Template:20.109(F12):Mod3 OrangeTR Pre-proposal
Investigators
Rebecca Shi and Catherine Fan
TR Orange
Title of Proposed Project
20.109(F12) Pre-Proposal: Engineering Oryza sativa to Express the Proteorhodopsin Photosystem
Project Summary
As the world population and the economic prosperity of the developing world increase, there will be an increased demand for food that can only be met by a doubling of the current agricultural productivity by the end of the century. The energy efficiency of C3 photosynthesis is currently around 4.6% in terms of total available solar energy to biomass conversion, which previous efforts have attempted to improve by reducing the competing activity of photorespiration. The proposed research would take a different approach in addressing this problem by introducing a bacterial photosystem into rice plants that will absorb sunlight in the green wavelength region of light, thereby increasing the amount of solar energy available to be converted into chemical energy.
Introduction
Photosynthesis is the process behind the world’s supply of food and fiber. However, given that the world’s population is expected to plateau around 10 billion and that the increased economic prosperity of the developing will create larger demand for food by the end of the century, the current agricultural productivity will be insufficient to support the world’s population. Since it is unlikely the current amount of cropland will expand, it is predicted the current productivity will need to double in order to meet demands [1]. While more solar energy reaches the Earth’s surface every hour (4.3 x 10^20 J) than is consumed in a year (4.1 x 10^20 J) [2], much of this energy is not captured in photosynthesis. After taking into account energy losses from light that is outside the photosynthetic spectrum, photochemical inefficiency, photorespiration, and other steps in photosynthesis, it is estimated that C3 plants can convert 4.6% of incident solar energy into biomass while C4 plants have a 6% efficiency rate, though C3 plants are expected to overtake C4 plants with an increase in atmospheric CO2 concentration [3]. There have been previous attempts to increase photosynthetic efficiency by reducing the effects of photorespiration, for example by mutagenizing Rubisco or engineering E. coli metabolic pathways to divert glycolate from photorespiration [4]. We propose a separate approach to raising the theoretical conversion limit of photosynthesis by increasing the range of wavelengths available for energy capture through engineering the proteorhodopsin photosystem into Oryza sativa.
Our Idea
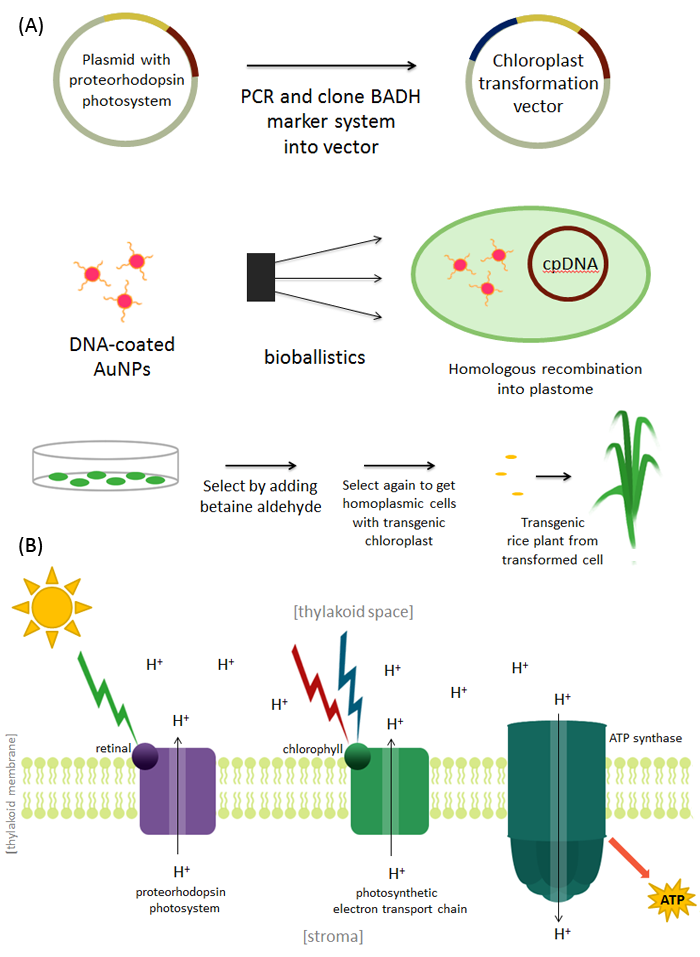
We decided to genetically engineer the organism Oryza sativa or Asian rice, a staple grain that a large part of the world’s population depends on and is the second highest produced grain after corn. It a model organism in cereal biology because it is relatively simple to genetically engineer. During regular photosynthesis the pigment chlorophyll is able to absorb red (~600-700nm) and blue light (~400-500 nm) This energy is utilized by the photosynthetic electron transport chain to pump protons across the thylakoid membrane. These protons then flow back into the stroma through ATP synthase to produce ATP. We are proposing that if the pigment retinal, which is able to absorb green light (~500-600nm), is incorporated into the chloroplast, a higher percentage of solar to chemical energy (ATP) conversion can occur in the rice plant and improve crop yield.
Dr. Justin Buck successfully incorporated the proteorhodopsin photosystem containing retinal into Pseudomonas putida and Pseudoalteromonas atlantica [5]. In order to introduce the photorhodopsin photosystem into Oryza sativa, we will add a marker, a gene that codes for betaine aldehyde dehydrogenase (BADH), into the vector coding for the proteorhodopsin photosystem. We will then coat gold nanoparticles with this DNA and use bioballistics (gene gun) to transform the chloroplasts of Oryza sativa cells [6]. We will select for transformants by adding betaine aldehyde to the media. Betaine aldehyde is normally toxic to plant cells, but cells expressing the marker BADH can degrade this molecule and survive [7]. These cells can now germinate and give us our transgenic rice plant. We should be able to visually see in the form of darker leaves if the chloroplast has incorporated this new pigment, quantify this difference by spectrophotometry to compare absorbances of leaf extracts between transgenic and normal rice. We will then use digital image analysis to model and quantify our biomass [8]. If our experiments are successful, we would expect a higher absorbance of transgenic plants in the green light (~500-600 nm) region, as well as a greater biomass in the transgenic plants. If these transgenic plants show increased biomass production, this result would suggest an increased energy capturing ability in the photosynthetic system engineered to include proteorhodopsin photosystem activity.
Figures
References
1. Chrispeels MJ, Sadava DE: Development, productivity and sustainability of crop production. In Plants, Genes and Crop Biotechnology. Edited by Chrispeels MJ, Sadava DE. Jones and Barlett Publishers; 2003:52-75.
2. Nault, Renée M. Basic Research Needs for Solar Energy Utilization. Proc. of Basic Energy Sciences Workshop on Solar Energy Utilization, Washington DC. N.p.: n.p., 2005. Print.
3. Xin-Guang Zhu, Stephen P Long, Donald R Ort, What is the maximum efficiency with which photosynthesis can convert solar energy into biomass?, Current Opinion in Biotechnology, Volume 19, Issue 2, April 2008, Pages 153-159, ISSN 0958-1669, 10.1016/j.copbio.2008.02.004. (http://www.sciencedirect.com/science/article/pii/S0958166908000165)
4. Kebeish, R. et al. 2007 Chloroplastic photorespiratory bypass increases photosynthesis and biomass production in Arabidopsis thaliana. Nat. Biotechnol. 25, 593–599. (doi:10.1038/nbt1299)
5. Buck, Justin D. "Physiological Effects of Heterologous Expression of Proteorhodopsin Photosystems." Thesis. Massachusetts Institute of Technology, 2012. Print.
6. Ramesh VM, Bingham SE, and AN Webber. 2001 A Simple Method for Chloroplast Transformation in Chlamydomonas reinhardtii. Photosynthesis Research Protocols, Methods in Molecular Biology. Edited by Carpentier R. Volume 684, DOI 10.1007/978-1-60761-925-3_23.
7. Daniell H, Muthukamar B, and SB Lee. 2001 Marker free transgenic plants: engineering the chloroplast genome without the use of antibiotic selection. Current Genetics. 39: 109-116.
8. Tackenburg O. 2007 A New Method for Non-destructive Measurement of Biomass, Growth Rates, Vertical Biomass Distribution and Dry Matter Content Based on Digital Image Analysis. Annals of Botany. 99: 777-783.