Lab 03: Electron Diffraction
Steve Koch 03:42, 21 December 2010 (EST):Good job with this lab and persevering through the difficulties with the apparatus!
Summary
Louis de Broglie hypothesized that all particles can also behave like waves and that their wavelengths are equal to plank's constant divided by their momentums. This is called the de Broglie Relationship. One way to demonstrate the wave nature of particles is to scatter them through a diffraction grating and observe the resulting pattern to be characteristic of waves. A diffraction grating can be made out of different things, but it needs to be small enough that it provides obstacles spaced on the order of the size of the wavelength appropriate for the particle to be diffracted. When the particles encounter the diffraction grating, they spread out into a pattern of areas where many particles go or where only a few go, called the maxima and minima. In this way they act like waves spreading out after passing through a small opening.
In this lab we passed electrons through a diffraction grating made of graphite foil. Graphite is composed of layers of hexagonally bonded carbon atoms. The pattern of minima and maxima we observed were in the shape of rings created by the electrons striking a phosphorescent coating inside a vacuum chamber. We measured the diameter of the two rings and used them to find the characteristic spacings between the atoms in the graphite foil.
- In this lab I worked with Brian P. Josey.
- These are my Detailed Lab Notes.
Analysis
The diffraction grating in our experiment was a thin graphite foil with a crystalline structure. The structure is hexagonal and has two characteristic spacings of 0.123 nm and 0.213 nm. The two rings we saw correspond to these two spacings. The pattern is ring shaped instead of six hexagonal points because the electrons pass through many hexagons randomly oriented to each other in the sheets of carbon that make up graphite.
The spacing in the crystalline structure is related to voltage of the electrons and the diameter of the rings that form by
[math]\displaystyle{ d= \frac {4 \pi L \hbar c} {D \sqrt {2 e V m c^2}} }[/math]
In this equation, d is the characteristic spacing or the spacing between the atoms in the lattice, V is the acceleration voltage, D is the diameter of the ring, L=13.0 ± 0.2 cm is the distance from the foil to where we measure the ring, mc2=9.109 * 10-31 kg or 0.511 MeV is the rest mass of the electron, e=1.602 * 10-19 C is the charge of the electron, h=6.626 * 10-34 Js or 4.135 * 10-15 eVs is Plank's constant, and c=2.998*108 m/s.
Because the glass is curved where we measure the rings, the actual distance of travel for the electrons that form the rings is less than the distance from the graphite to the center of the rings. This affects the diameter of the rings.
The correction for this can be computed using the radius of curvature of the spherical tube, R, and the length of the path from the foil to the center, L.
Let the error in the distance be given by y:
[math]\displaystyle{ y = R - \sqrt{R^2-\frac{D_{obs}^2}{4}} }[/math]
It can be shown using y that:
[math]\displaystyle{ tan(\theta) = \frac\frac{D_{obs}}{2}{L-y} }[/math]
The corrected diameter using [math]\displaystyle{ tan(\theta) }[/math] is:
[math]\displaystyle{
D=2Ltan(\theta) \,
}[/math]
In Dr. Gold's Lab Manual, R is given to be 66mm.
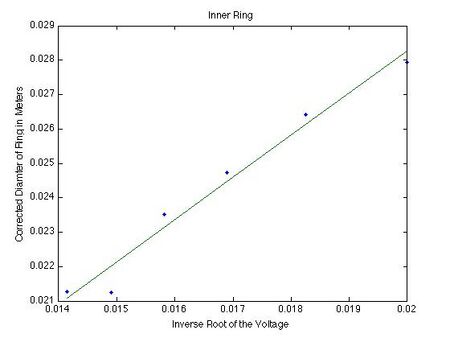
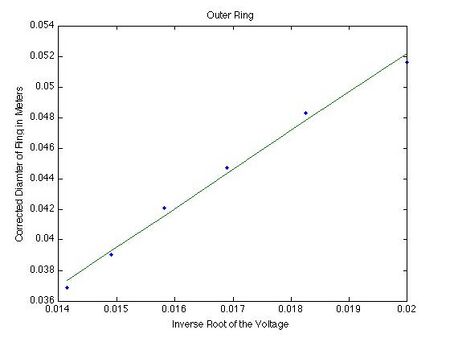
The next step in finding the characteristic spacings is to plot the corrected diameter of the ring as a function of the inverse square root of the voltage and find the slope through linear regression.
The slope for the inner ring is 1.2260 and the slope for the outer ring is 2.5405.By putting these values for the slope into the following formula, the characteristic spacings can be found.
[math]\displaystyle{ d = \frac{\frac{2hL}{\sqrt{2me}}}{Slope} }[/math]
This gives the spacing for the inner ring as 0.245 nm as the best guess. This value then has a confidence interval from 0.242 nm to 0.246 nm. For the outer ring, this gives the value of 0.139 nm for the best value of the outer ring. We have a confidence interval for this value as 0.140 nm to 0.136 nm. These intervals of confidence were determined by entering the average value plus or minus the standard error of mean into the graph and finding the new slope there.
Conclusion
In this lab I worked more with statistical analysis. I enjoyed this lab very much even though we encountered many problems and had to start over twice. I don't understand the derivations as well as I would have liked to. This lab is done mostly in complete darkness, so in the future I would suggest using a red light flashlight to read the numbers on the calipers so that the vision of the person taking measurements isn't constantly readjusting.