Biomod/2011/SRISHTI/ArtScienceBangalore/Project
<html> <link rel="stylesheet" href="http://hackteria.org/biomod/style.css"/>
<div id="asbContent"
</html>
Project
<html> <object width="640" height="480"><param name="movie" value="http://www.youtube.com/v/UDwt_Bu3rmc?version=3&hl=en_US&rel=0"></param><param name="allowFullScreen" value="true"></param><param name="allowscriptaccess" value="always"></param><embed src="http://www.youtube.com/v/UDwt_Bu3rmc?version=3&hl=en_US&rel=0" type="application/x-shockwave-flash" width="640" height="480" allowscriptaccess="always" allowfullscreen="true"></embed></object> </html>
RASA-01: Open-Souce DNA Computing RASA-01 is a simple Do-it-yourself DNA computing kit for the general hobbyist interested in DNA computing. By collaborating with artists, designers, scientists and chefs, ArtScienceBangalore presents alternate scenarios of Human Computer Interaction with bio-molecular computers and DNA computing.
The Altair 8800 was a Do It Yourself microcomputer kit introduced in 1975. Sold through mail order advertisements, the kit, based on an Intel 8080 CPU was instrumental in sparking the micro-computer and personal computer revolution. The Altair was not an easy computer to program, the user had to toggle switches to positions corresponding to an 8080 microprocessor instruction or opcode in binary. With RASA-01 ArtScienceBangalore proposes an updated, simpler DNA version of the Altair. RASA-01 uses the Shapiro-Rothemund Unit which is a programmable two state DNA automaton. It uses a double stranded DNA as input, endonuclease and DNA ligase as main hardware and transition molecules as software.
PROJECT
This project, carried out by artists, designers and scientific collaborators, is a speculative investigation into the possibilities of using Biolomolecular design to make a simple 2-state finite machine. We prototyped an educational open-source biocomputer that takes the form of a kit which includes:
- a DIY PCR machine
- a DIY Gel electrophoresis box
- input strands of DNA
We hope this project makes the theories and methods behind bimolecular design and biomolecular computing accessible to designers, artists, enthusiasts and amateurs. We believe that burgeoning technologies have more interesting outcomes when a plurality of voices are included in their formative years.
SWEET & SOUR NANOBOTS: EDIBLE INTERFACES?
During the development of our project we imagined with a variety of interfaces. Researching the history of early kit-computing and Human-Computer-Interaction, we realized how essential rich media interfaces such as screens, keyboards, speakers and mouse were to making digital computing accessible to a wider audience. We came to the conclusion that there are many advantages and interesting design constraints by focusing on taste as the sensory interface for the nano computer. Imagining and prototyping taste as a means of human-nanocomputing-interface has forced us to ask many questions about the possibilities and limitations of design for BioMod. The focus on medicinal uses of nanotechnologies has also made us to revisit the historical relationship between medicine and food, and ask if there are any synergies in these domains for nanotechnology. From our research into the history of kit computing it appears that early design decisions in the human-computer-interfaces and peripherals can lead to lock-in effects in both thinking and actual production. By imagining the possibilities of tastable computing we hope to open unexplored areas of the solution space for computing interfaces. Our first version of an Open Source DNA Computer is called RASA - "taste" in Sanskrit.
PROJECT PROCESS
The Project involved 2 phases:
Phase1
Building the Hardware to carry out the computation- This involved building a PCR machine and a GEL BOX. The PCR we built was based on Russel Durrett's Design.
Phase2
Synthesizing the actual DNA strands and enzymes that carry out the computation
The Light Bulb PCR machine uses a light bulb and an old computer fan as its heating and cooling elements
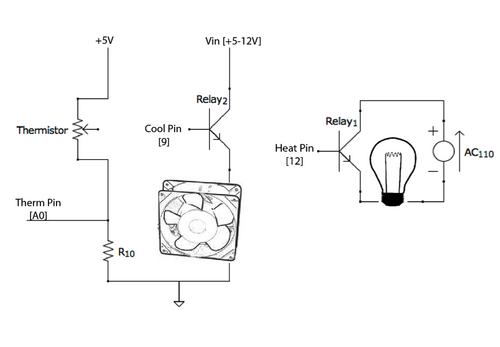
The unit is controlled by an Arduino Uno. It uses a thermistor (a resistor that allows more current to flow and more heat is available) to monitor the temperature of the samples. Here's the schematic:
The light bulb is run on240 Volt AC, so the Arduino controls a relay which turns the lamp circuit on and off. Both the Arduino and Fan can be powered by a 12 V external power supply (which provides faster cooling rates).
In this model, the tubes are heated using a 200 Watt light bulb and cooled simply by an old computer fan.
The machine first heats to 95 degrees Celsius, once the temperature is reached it is held for 5 minutes.
Then the PCR begins to cycle, each cycle consists of,
Heating to 95 degrees Celsius (DNA disassociating temperature), maintaining it for 30 seconds,
Cooling to 53 degrees Celsius (DNA primers and annealing temperature), maintaining it for 30 seconds,
Heating to 72 degrees Celsius (DNA elongating temperature ), maintaining it for 2 minutes.
The PCR cycles 30 times and then holds the temperature at 72 degrees Celsius for 5 minutes.
We built this prototype out of 4 PVC pipe for a few reasons: because there are many prefabricated attachments available, it's easily available and has consistent diameter.
There are three layers in the system all based on one 4 PVC coupling and held together with regular 4 pipe. The top layer is a 4” X 2.5” concave connector that has eight 1cm holes that holds the eppendorf tubes one of which contains the thermistor and is attached to one end of a “T” junction. The light bulb is held within the 4” ‘T’ junction. The fan is connected to another end of the ‘T’ junction and the adurino board and the 12 V adapter and the electronics are concealed within a 4” PVC cap and connected to another end on the “T” junction.
The temperature is monitored using a thermistor - basically a resistor that decreases resistance depending on how hot it gets. The way it is wired, the hotter the thermistor gets, the more potential feeds into the analog input pin on the arduino.
The arduino can be powered from an external 12V source while the lamp circuit needs to be plugged in to a 240V outlet.
Phase 2 involved building the wetware that carries out the computation
The process and the protocols are given below:- The protocols are from the paper: Programmable and autonomous computing machine made of biomolecules Yaakov Benenson, Tamar Paz-Elizur², Rivka Adar, Ehud Keinan,Zvi Livneh & Ehud Shapiro
PROTOCOL FOR BUILDING A DNA COMPUTER
Methods Synthetic DNA Double-stranded synthetic DNA molecules were prepared by annealing 2,000 pmol of commercially obtained deoxyoligonucleotides (Sigma-Genosys) in a final volume of 10ml of 10mM Tris-HCl buffer, pH8.0, containing 1mM EDTA and 50mM NaCl. The annealing was performed by heating the solution to 94 VC followed by slow cooling. The formation of a duplex was confirmed by native PAGE (20%). The oligomers were 5W-phosphorylated and PAGE-purified by the supplier and used without further purification. Input molecules These were constructed stepwise by ligating one or more synthetic DNA segments of thedesired sequence to a 1,457-bp fragment obtained by digestion of the pBluescript II SK+ plasmid (Stratagene) with FokI, followed by polymerase chain reaction (PCR) amplification of the coding segment and a 300- or 325-bp tail region. The sequences of the resulting input molecules were confirmed by sequencing.
Output-detecting molecules The output-detecting molecule for the S0 output (S0-D) was formed by ligating a synthetic adapter of 30 bp containing a FokI recognition site to a 181-bp fragment obtained by digesting the pBluescript II SK+ plasmid with FokI, PCR amplification and additional FokI digestion to form the 160-bp fragment bearing the desired sticky end. The output-detecting molecule for the S1 output (S1-D) was obtained by PCR amplification of a 285-bp fragment corresponding to positions 1,762±2,047 of the pBluescript II SK+ plasmid followed by FokI digestion of the PCR product to form a 250-bp fragment.
Computation reactions Reactions were set by mixing 2.5 pmol of the input molecule, 1.5 pmol of each output- detection molecule and 15 pmol of each transition molecule with 12 units of FokI and 120 units of T4 DNA Ligase (both from New England Biolabs) in 120 ml of NEB4 buffer supplemented with 1 mM ATP and incubating at 18 VC for 70 min. In case of multiple inputs in the same reaction, equal amounts were used, summing up to 2.5 pmol. The mixtures were purified by the Qiagen PCR purification kit and eluted using 30 ml EB buffer (Qiagen). Aliquots (10 ml) were assayed by gel electrophoresis using 3% MetaPhor agarose (FMC Bioproducts) unless indicated otherwise. The lengths of the DNA species were verified using a commercial 50-bp DNA step ladder (Promega). To further confirm that output reporting molecules were formed as expected, we amplified by PCR and sequenced the output-detection molecule/output molecule junction region in both output-reporting molecules S0-R and S1-R and found the expected sequences .
RESULTS
Our DIY PCR did not seem to give out any bands in a gel.
<html>
</html>